автордың кітабын онлайн тегін оқу The Propaganda for Reform in Proprietary Medicines, Vol. 2 of 2
Transcriber’s notes:
In this transcription a black dotted underline indicates a hyperlink to a page, illustration or footnote; hyperlinks are also marked by aqua highlighting when the mouse pointer hovers over them. A red dashed underline indicates the presence of a hidden comment that can be revealed by hovering the mouse pointer over the underlined text. Page numbers are shown in the right margin.
The text contains some uncommon characters that will not necessarily display correctly with all viewing devices. If some of the characters look abnormal, first ensure that the device’s character encoding is set to Unicode (UTF-8). The default font might also need to be changed to a Unicode font such as Arial Unicode MS, DejaVu, Segoe UI Symbol or FreeSerif. Symbols representing male and female have been replaced by M and F in the handheld version. Subscripted and superscripted characters do not always display correctly on handheld devices.
The book contained innumerable typographical errors affecting spelling, punctuation and formatting. Most spelling errors have been corrected silently, but spelling variants that mainly reflect contemporary spellings in the original quoted sources, have been left unchanged. A list of the corrections and variations can be seen at the end of the transcription. Punctuation anomalies (errors, omissions, duplications) have mostly been corrected silently, but missing apostrophes have not been corrected because of uncertainty about intended meaning. A few missing quotation marks have been inserted within curly brackets {"} and missing words inserted by the transcriber, e.g. {sic} {and} have also been enclosed within curly brackets to differentiate them from the numerous words and phrases inserted by the authors. Redundant duplicated words have been deleted. Astute readers will probably note persisting inconsistencies involving italics, fonts, hyphenation, ellipses, accents, and ligatures (ae/æ, oe/œ) that have been left unchanged because they have no impact on interpretation of the text. The Table of Contents does not correspond accurately with headings used in the text.
Footnotes are located at the end of the book and are hyperlinked in the text. In most cases a return hyperlink will take the reader back to the appropriate place in the text, except for those footnotes that are cross-referenced multiple times. In these cases the return hyperlink will take the reader back to the location where the footnote was first referenced – it is better to make use of the browser's back-arrow to return to the correct location. An error in footnote numbering was corrected silently, and a missing footnote [120] was inserted after being identified in the original source.
Several large tables have been compressed in size to enable viewing on small screens and when necessary a key has been added to identify the column headings.
THE PROPAGANDA FOR REFORM IN Proprietary Medicines
VOLUME 2
Part I
Reports of the Council
Part II
Contributions from the Laboratory
Part III
Journal Contributions: Proprietary Products
Part IV
Journal Contributions: Miscellany
PRESS OF AMERICAN MEDICAL ASSOCIATION, FIVE HUNDRED
AND THIRTY-FIVE NORTH DEARBORN STREET,—CHICAGO
1922
Copyright, 1922 by the American Medical Association
PREFACE TO VOLUME 2
There were nine editions of the first volume of The Propaganda for Reform in Proprietary Medicines. The ninth edition contained the most important reports of the Council on Pharmacy and Chemistry and of the Chemical Laboratory. It contained also those articles from The Journal of the American Medical Association (up to, and including, 1916) which dealt with the problems of proprietaryship in medicine and the furtherance of rational drug therapy.
The present volume contains similar material covering the period from January, 1917, to April, 1922, inclusive. Like Volume 1, this volume is divided into four parts:
Part I. The Council on Pharmacy and Chemistry: This section presents the principles and rules which govern the Council in the examination of medicaments, together with articles and reports bearing on the work of the Council, and the most important reports of the Council from 1917 to April, 1922, inclusive.
Part II. The A. M. A. Chemical Laboratory: This section, besides presenting the aims and objects of the Association’s Chemical Laboratory, also outlines some of the Laboratory’s work which is of particular interest to physicians.
Part III. Contributions from the Journal: Proprietary Products: This part contains articles on proprietary medicinal preparations and the methods by which they are exploited, which have appeared in The Journal A. M. A.
Part IV. Contributions from The Journal: Miscellany: In this section are articles dealing with matters of interest to the medical profession but not coming strictly under the classification of proprietary medicinal preparations.
A comparison of the material that has appeared in Volume 1 of The Propaganda for Reform with that which appears in this volume will reveal the changing conditions in the proprietary medicine field. Many of the reports in the first volume brought out the fact that medicinal preparations were at that time foisted on the profession with false claims of composition; reports of this character are less conspicuous in the present volume. Many of the reports in Volume 2 deal with unwarranted therapeutic claims, especially those advanced for animal organ preparations, serums, vaccines, preparations for intravenous medication, etc. The present volume will also be found of interest in its portrayal of the changed conditions in the proprietary medicine business brought about by the World War.
Special attention is directed to the index in this volume. It is, in effect, a bibliography, including references not only to articles in this book but also (1) to articles which appeared in Volume 1; (2) to articles on the same general subject in The Journal of the American Medical Association, and (3) to the articles appearing in the annual reports of the Council on Pharmacy and Chemistry and of the A. M. A. Chemical Laboratory, but not reprinted in either volume of the Propaganda for Reform in Proprietary Medicines.
PREFACE TO VOLUME 1: NINTH EDITION
From time to time The Journal of the American Medical Association has published the reports of the Council on Pharmacy and Chemistry and the Chemical Laboratory, as well as other matter on proprietary medicines. Repeated requests for some of the matter have led to the compilation of “The Propaganda for Reform in Proprietary Medicines,” which, in the present volume, attains its ninth edition.
The seventh, eighth and ninth editions have been compiled on slightly different principles from their predecessors. The therapeutic reform work of The Journal and of the Association’s Chemical Laboratory was at first confined almost entirely to the criticism and analysis of the so-called ethical proprietaries. This was right; the medical profession owed it to the public to combat the nostrum evil within its own ranks.
As the more flagrant evils of the “ethical proprietary” question were mitigated, the Association has turned the light on the more widespread and dangerous “patent medicine” evil. The articles devoted to “patent medicines” or quackery being naturally of greater interest to the general public than to the medical profession, the number of inquiries from laymen regarding various quacks and nostrums has steadily increased. It has been thought best, therefore, to publish separately all of the matter from The Journal relative to quackery and to those nostrums exploited only or chiefly to the public, and to include in the Propaganda for Reform practically none of the matter that is of direct interest primarily to laymen. In one or two instances in which the subjects were of equal interest to the profession and to the public, matter that has already appeared in “Nostrums and Quackery” is also given here; but as a general rule the contents of the ninth edition of “The Propaganda for Reform” are of strictly professional interest. Those physicians who are desirous of obtaining in convenient form the matter dealing with “patent medicines” should order the book “Nostrums and Quackery” or the various pamphlets on the same subjects that have been issued since “Nostrums and Quackery” came from the press.
The ninth edition of “Propaganda for Reform” contains a number of new articles, greatly increasing the size of the book. It also contains one novel feature which greatly enhances its value. The index includes references not only to articles in the book, but also to matter on proprietaries not accepted by the Council on Pharmacy and Chemistry which appeared in The Journal of the American Medical Association and elsewhere. This index makes of this edition of “Propaganda for Reform” a very full work of reference on proprietaries which are undeserving of recognition. It should be understood, however, that not all articles indexed are condemned; some are merely discussed and compared.
RESOLUTION ENDORSING THE WORK OF THE COUNCIL ON PHARMACY AND CHEMISTRY
Presented at the San Francisco Session and Signed by All the Members of the House of Delegates in Attendance
Resolved, We, Members of the House of Delegates of the American Medical Association, believe that every effort must be made to do away with the evils which result from the exploitation of the sick for the sake of gain. Earnestly believing that the continued toleration of secret, semisecret, unscientific or untruthfully advertised proprietary medicines is an evil that is inimical to medical progress and to the best interest of the public, we declare ourselves in sympathy with, endorse and by our best efforts will further, the work which has been, and is being, done by the Council on Pharmacy and Chemistry of the American Medical Association in the attempt to eliminate this evil.
TABLE OF CONTENTS
PART I: COUNCIL REPORTS
PAGE
Foreword
1Official Rules of the Council on Pharmacy and Chemistry
3The Council on Pharmacy and Chemistry, Present and Future
12“Accepted by the Council on Pharmacy and Chemistry”
19Helping the Council
20Delays in Passing on Products
20Cooperation of the Pharmaceutical Houses
21Budwell’s Emulsion of Cod-Liver Oil, Nos. 1 and 2
22Rheumalgine
23Gray’s Glycerine Tonic
24Tongaline and Ponca Compound
26Alfatone
28Uricsol
30Jubol
31Urodonal
32Formamint
33Hydragogin
41Filudine
41Lactopeptine and Elixir Lactopeptine
43Iodum-Miller and Iod-Izd-Oil (Miller’s)
49Elixir Iodo-Bromide of Calcium Comp. “Without Mercury” and “With Mercury”
52Lecithin Preparations Omitted from N. N. R.
53Proprietary Names for Liquid Petrolatum
55Seng
55Frosst’s Blaud Capsules
56Tyree’s Elixir of Buchu and Hyoscyamus Compound
57Hydroleine
58Curative Vaccine, Bruschettini
58Stearn’s Wine
59Protonuclein and Protonuclein Beta
59Hydropsin
61Digitalysatum
63So-Called Secretin Preparations
64Has Secretin a Therapeutic Value?
65Radio-Rem
79Olio-Phlogosis
79The Hypophosphite Fallacy
80Pulvoids Calcylates
85Sulfuryl Monal
86Mark White Goiter Serum and Mark White Iodinized Oil
87Kora-Konia
92The Therapeutic Value of the Glycerophosphates
93Hydras
96Bromin-Iodin Compound
97Ammonium Hypophosphite Omitted from N. N. R.
98Alphozone Omitted from N. N. R.
99Calcium Glycerophosphate and Sodium Glycerophosphate Omitted from N. N. R.
99Gardner’s Syrup of Ammonium Hypophosphite Omitted from N. N. R.
100Gluten Products Made by the Kellogg Food Company
100Iodo-Mangan Omitted from N. N. R.
106Liquid Albolene
106Naphey’s Medicated Uterine Wafers
107Nujol
108Pulvoids Natrium Compound
108Saloform
110Secretogen
110Iron Citrate Green
115Aspirin
116Pil. Cascara Compound-Robins
117Casta-Flora
118Firwein
119Firolyptol Plain and Firolyptol with Kreosote
120Biniodol
121 Comparative Symptoms Resulting from the Use of Several Oily Suspensions ofRed Mercuric Iodid (Mercury Biniodid)
123Corpora Lutea (Soluble Extract), Parke, Davis & Co.
128Wheeler’s Tissue Phosphates
129The Claimed Galactagogue Effects of Nutrolactis and Goat’s Rue Not Substantiated
131The Alleged Galactagogue Action of Galega and Nutrolactis
131The Russell Emulsion and the Russell Prepared Green Bone
134Brom-I-Phos
136Creosote-Delson and Creofos
137Triner’s American Elixir of Bitter Wine
139Trimethol
140Ferrivine, Intramine and Collosol Iodine
144Eskay’s Neuro Phosphates
146K-Y Lubricating Jelly
147Ziratol
148Gonosan
150Alcresta Ipecac
153Iodeol and Iodagol
154Capsules Bismuth Resorcinol Compound Not Admitted to N. N. R.
157Dixon’s Tubercle Bacilli Extract and Dixon’s Suspension of Dead Tubercle Bacilli
158Formosol
158Iodolene, a Solution of Iodin in Liquid Petrolatum, Inadmissible to N. N. R.
159Kalak Water
160Minson’s Soluble Iodin “Kelpidine” Not Admitted to N. N. R.
161Nutone
162Tri-Arsenole, L. O. Compound No. 1 and L. O. Compound No. 2
163Unctol
166V-E-M (Schoonmaker Laboratories, Inc.)
166Hemo-Therapin
168Venosal
169Secretin-Beveridge and the U. S. Patent Law
170The Question of the Stability of Secretin
171Need for Patent Law Revision
177Surgodine
180Medeol Suppositories
181Guaiodine
183Several “Mixed” Vaccines Not Admitted to N. N. R.
184Ophthalmol-Lindemann
189Silvol Ineligible for N.N.R
189Katharmon
191Iodinized Emulsion (Scott) and Creosotonic (Scott)
192Campetrodin and Campetrodin No. 2
193Carminzym
194Phillips’ Phospho-Muriate of Quinine Comp
197B. Iodine and B. Oleum Iodine
198B. Iodine Products
199 Antithyroid Preparations (Antithyroidin-Moebius and Thyreoidectin) Omittedfrom N. N. R.
202 Cephaelin and Syrup Cephaelin-Lilly Omitted from N. N. R. and Syrup Emetic-LillyNot Accepted
203Colalin Omitted from N. N. R.
203Foral
204Granular Effervescent Bromide and Acetanilid Compound-Mulford
206Holadin and Bile Salt Mixtures
207Liquor Santaiva, S. & D., Omitted from N. N. R.
211 Maltzyme, Maltzyme with Cascara Sagrada, Maltzyme with Cod Liver Oil, MaltzymeFerrated and Maltzyme with Yerba Santa Omitted from N. N. R.
211Methaform Omitted from N. N. R.
212 Pineal Gland, Red Bone-Marrow and Thymus Gland and Their PreparationsOmitted from N. N. R.
213Piperazine and Lycetol Omitted from N. N. R.
214Stanolind Liquid Paraffin Omitted from N. N. R.
214Westerfield’s Digitalis Tablets
215Xeroform-Heyden and Bismuth Tribromphenate-Merck Omitted from N. N. R.
216Cream of Mustard Refused Recognition
218“Pluriglandular” Mixtures
218Cerelene Not Admitted to N. N. R.
219Collosol Cocaine Not Admitted to N. N. R.
221Cuprase Not Admitted to N. N. R.
222Collosol Preparations
223Pulvoids Calcylates Compound
226Proteogens of the Wm. S. Merrell Company
227“Arsenoven S. S.” and “Arseno-Meth-Hyd”
231Hormotone and Hormotone Without Post-Pituitary
234Formaldehyde Lozenges
235Lavoris
237Medinal
239Omission of Cotarnin Salts (Stypticin and Styptol) from N. N. R.
240Micajah’s Wafers and Micajah’s Suppositories
241Alkalithia
242Arhovin Omitted from N. N. R.
243Chloron, Chlorax and Number “3”
244Elarson Omitted from N. N. R.
248Iodiphos
249Mervenol and Armervenol Not Admitted to N. N. R.
249 Normal Phenol Serum (Cano) and Methyl-Phenol Serum (Cano) Not Acceptedfor N. N. R.
251Soamin Omitted from N. N. R.
253Some Mixed Vaccines Not Admitted to N. N. R.
254Somnoform
255Tablets Formothalates
256Triple Arsenates with Nuclein
256“Anti-Pneumococcic Oil” and the Use of Camphor in Pneumonia
257Dial “Ciba”
259Apothesine
260Eumictine
262Platt’s Chlorides
263 Anti-Tuberculous Lymph Compound (Sweeny) and Anti-Syphilitic Compound(Sweeny)
266Syrup Leptinol (Formerly Syrup Balsamea)
268Formitol Tablets, II
271Sukro-Serum and Aphlegmatol
273Supsalvs Not Admitted to N. N. R.
274 Hypodermic Solution No. 13, Iron, Arsenic and Phosphorus Compound NotAccepted for N. N. R.
275Parathesin Not Admitted to N. N. R.
276Chlorlyptus
277Aquazone (Oxygen Water)
290Coagulen-Ciba Omitted from N. N. R.
290Ferric Cacodylate Omitted from New and Non-Official Remedies
292Libradol
293Helmitol Omitted from N. N. R.
295Spirocide Not Admitted to N. N. R.
296Digifolin-Ciba Not Admitted to N. N. R.
298Some of Loesser’s Intravenous Solutions
299“National Iodine Solution” Not Admitted to N. N. R.
300Mon-Arsone Not Admitted to N. N. R.
302Oxyl-Iodide Not Admitted to N. N. R.
304Quassia Compound Tablets
306Toxicide
307Pil. Mixed Treatment (Chichester)
310Atophan Omitted from N. N. R.
313Urotropin Omitted from N. N. R.
316Styptysate Not Admitted to N. N. R.
318Lipoidal Substances (Horovitz) Not Admitted to N. N. R.
320Yeast Preparations and Vitamin B Concentrates
321PART II: CONTRIBUTIONS FROM THE A. M. A. CHEMICAL LABORATORY
The Chemical Laboratory of the American Medical Association
322The Work of the American Medical Association Chemical Laboratory
322Lead in “Akoz”
328Sodium Acetate in Warming Bottles
329Anti-Syphilitic Compound (Sweeny)
330“Ambrine” and Paraffin Films
330The Stability of Iodine Ointments
337Iodolene and the Solubility of Iodin in Liquid Petrolatum
344American-Made Synthetic Drugs—I
344Standardization of Commercial Bismuth Tribromphenate
348Standardization of Procain and Examination of the Market Supply
355Deterioration of Sodium Hypochlorite Solutions
358Syphilodol
359Cerelene
362Dr. De Sanctis’ Rheumatic and Gout Pills
363Iodex and Liquid Iodex
365PART III: CONTRIBUTIONS FROM THE JOURNAL:
PROPRIETARY PRODUCTS
Iodin in Liquid Petrolatum
367American-Made Synthetic Drugs—II
369Nostrums in Retrospect
379Bell-Ans (Pa-Pay-Ans Bell)
380Anasarcin and Anedemin
383Pepto-Mangan
387Cactina Pillets
391Ammonol and Phenalgin
393Fellows’ Syrup, and Other Preparations of the Hypophosphites
395Shotgun Nostrums
398Tyree’s Antiseptic and Aseptinol
401Neurosine and the Original Package Evil
404Anasarcin Advertising
407Antimeristem-Schmidt
408Antiphlogistine
409“Auto-Hemic Serum”
409“Autolysin” Advertising
413“Basic Cancer Research” and “Cosmopolitan Cancer Research Society”
414Seleni-Bascca
416Bell-Ans (Papayans, Bell)
418Campho-Phenique
418“Cinchophen”: Formerly “Atophan”
419“Collosols”: An Uncritical English Endorsement
420Cotton Process Ether
421Dionol
422The Eli Products of Eli H. Dunn
424Glover’s Cancer Serum
425Glyco-Thymoline and Poliomyelitis
427Glykeron: Cold Storage Testimonials
428Gray’s Glycerine Tonic: “Whose Bread I Eat His Song I Sing”
429Hagee’s Cordial of Cod Liver Oil
429Hypno-Bromic Compound
430Intravenous Compound (Loffler)
430Intravenous Specialties
435Iodex
436The William F. Koch Cancer Remedy
437The Lucas Laboratories’ Products
440“Phylacogens”
441Pineoleum Advertising Methods
442“Proteal Therapy” and Henry Smith Williams
443Proteogens
445Pulvane
450Sal Hepatica
451Salicon
453So-Called Secretin Preparations
454Succus Cineraria Maritima
455Tekarkin
458Tyree’s Antiseptic Powder Again
462Wheeler’s Tissue Phosphates
463Briefer Paragraphs
465PART IV: CONTRIBUTIONS FROM THE JOURNAL:
MISCELLANY
Albert Abrams, A.M., M.D., LL.D., F.R.M.S.
472Acetylsalicylic Acid, Not Aspirin
480The Allied Medical Associations of America
486“Arsenicals”
491Beer and Cancer Cures
494Biologic Therapeutics and Its Commercial Domination
496Capell’s Uroluetic Test
497Chemotherapy and Tumors
499The Direct Sales Company
510Discoveries and Discoverers
511“Drug Reform”
513Drug Therapy: The Fallibility of Textbooks
515Thomas Webster Edgar
515Glycerophosphates
520Influenza Vaccine
520Intravenous Therapy
522Iodin Fumes
523Italian Physico-Chemical Company
524What is Liquid Petrolatum?
526The Lowenthal Postgraduate Course
527Medical Society of the United States
531The National Formulary—A Review of the Fourth Edition
535Nonspecific Protein Therapy
536Willard Ealon Ogden
538“Patents”
542Pharmaceutical Barnums
545The Pharmacopeia
546Physician’s Stock in Prescription Products
548Pituitary Gland Preparations
549Proprietorship in Medicine
550Philip Rahtjen and His Discoveries
553Sodium Cacodylate in Syphilis
555Tablets: Dependability of Dosage
556Therapeutic Evidence: Its Crucial Test
557“Vaccines in Toxic Conditions”
560Vitamins: Their Distribution
561The William A. Webster Co. and the Direct Pharmaceutical Co.
564Yeast
566Briefer Paragraphs
570We seem to have wandered from the topic in our caption, but not so in reality, because the burden of our thought is to lend our influence to the spread of the motto of the Advertising Clubs of the World, namely, “Truth in Advertising.” It is our purpose to stimulate a larger degree of enthusiasm for the work of the Council on Pharmacy and Chemistry and the Chemical Laboratory, a more generous flow of inquiries concerning articles unfamiliar to the physician, and particularly to urge that the words “Accepted by the Council on Pharmacy and Chemistry of the American Medical Association” be printed on the label and on all advertising circulars of proprietary articles that have been admitted to New and Nonofficial Remedies. Then, when pamphlets and circulars are received by physicians they will read the statements of manufacturers with sympathetic understanding and with full confidence in the verity of the declarations. The importance of creating just that sort of receptivity in the mind of the prospective buyer is so well known to the astute publicity expert that it is needless for us to dwell on its advantages. Every proprietary article advertised in our journal, in The Journal of the American Medical Association, and in the other state association journals, as well as in several well-edited privately owned journals, does in effect say to the reader that the articles so advertised are accepted by the Council because only proprietary articles so accepted are accepted by us. The fact is further acknowledged when these firms are permitted to exhibit their goods at our annual sessions for again the rule is enforced that only proprietary articles which have been approved by the Council may be placed on display.
Unfortunately, but very naturally, there are many cases in which the information available at the time the product is submitted is not sufficient to justify the Council in coming to definite conclusions for or against the preparation. In some cases the manufacturer possesses the required information, but to obtain it from him takes time; in other cases the manufacturer does not possess the information—perhaps he did not realize the inadequacy of his evidence until the subject was brought to his attention by the Council.
“Cooperation of the Pharmaceutical Houses: At the opening meeting of the House of Delegates last year, President Arthur Dean Bevan suggested the desirability of greater cooperation between the large pharmaceutical houses and the Council on Pharmacy and Chemistry. The need of such cooperation has been recognized by the Council from the first. In no one direction has the Council made greater effort than in its endeavor to secure the fullest cooperation of the various pharmaceutical houses. The difficulty has been, and always must be, the fundamental antagonism between objectives that are largely commercial on the one hand and purely scientific on the other. Nevertheless, the Council has always believed—and has acted on the belief—that there is a possible middle ground wherein the interests of therapeutics would not be injured but would go hand in hand with a commercial development based on enlightened self-interest.
“The profits to be made by a pharmaceutical house from the sale of a staple drug—a pharmacopeial, National Formulary, or nonproprietary preparation—which enters into free competition with other drugs of the same kind, are moderate; the profits to be made from the sale of a proprietary medicine on which the manufacturer holds a monopoly are usually large—sometimes enormous. There are, broadly, two kinds of proprietary preparations advertised to physicians: One represents laborious research ending in the production of a new medicinal chemical; this product can be patented and the manufacturer can obtain a seventeen-year monopoly on its manufacture and sale. The other represents no research but comprises simple mixtures—frequently of the “shotgun” variety—of well known pharmaceuticals, or biologic products sold under trade names. As these do not represent anything new or original the manufacturer is unable to obtain a patent, but by means of the trade name he can and does obtain a perpetual monopoly. This, from a business standpoint, is more valuable than the limited monopoly granted by a patent. It is not surprising that proprietary remedies of the latter type flourish so long as physicians unthinkingly accept and prescribe them solely on the manufacturer’s valuation.
“The Council has practically the undivided support of manufacturers of medicinal chemicals; that is, of proprietaries of the first mentioned type. But pharmaceutical firms which have found it profitable to promote proprietaries of the second type—“specialties,” unscientific or ordinary mixtures of pharmaceuticals or biologic products sold under trade names—have not supported the Council.
“When the Council was organized, it was hoped and believed that all the large pharmaceutical houses would find it possible and desirable, if not actually more profitable, to shape their business methods so as to make their proprietary and other articles conform to those conservative standards on which the Council bases its rules, and thus render such articles acceptable for New and Nonofficial Remedies. It soon developed, however, that the methods of the pseudochemical companies, whose sales propaganda in the interest of unscientific nostrums with its attending damage to scientific medicine had led to the establishment of the Council, had found their lodgment in most of the pharmaceutical houses. It was a genuine disappointment to the Council to find that some of the large and old-established firms were not only unwilling to cooperate with the Council, but in many instances exhibited a definite antagonism to the Council’s work.
“The object—and duty—of the officers of pharmaceutical houses is primarily to pay dividends to their stockholders. Through skilful advertising or the persuasiveness of “detail men,” they are able to induce physicians to prescribe their controlled products, on which there are large profits, even though such products have not only not been accepted by the Council, but in many instances, have been disapproved. Is it any wonder that concerns which put out such products are indifferent or openly antagonistic to the work of the Council? The matter is largely one of business policy. When the medical profession as a unit will support the Council in its work, then such firms will find it good business policy to accede to Dr. Bevan’s suggestion—but not before.”
[Editorial Note.—An old practice in hospitals—happily now practically obsolete—was to have certain stock mixtures prepared in bulk. Among these there was usually a so-called tonic mixture, used in a more or less haphazard manner when nothing in particular seemed indicated. Such a stock mixture was used in the State Hospital for the Insane at Utica, N. Y., during the many years that Dr. John P. Gray was superintendent (from the early fifties to the early eighties), although it is very doubtful whether he originated the mixture. After the death of Dr. Gray—so the story runs—one of his sons, with a partner, formed the firm of Purdue Frederick Company, and began the exploitation of the elder Dr. Gray’s name, in connection, presumably, with this stock preparation. As indicated in the Council’s report, Gray’s Glycerine Tonic Comp.—and what an absurd name!—is simply a mixture of ordinary drugs, requiring no skill whatever in compounding. If there is a physician living who cannot write a prescription offhand as good as this formula, that physician should either go back to a medical school or change his vocation. There is, and can be, no excuse for prescribing such a ready-made mixture, for every cross-roads drugstore has the ingredients and any pharmacist worthy of the name could compound it. Among the scores of nostrums that disgrace the medical profession of this country, none is more typical of all that is inimical to scientific medicine, to the medical profession and above all to the public—for, after all is said, it is the public that ultimately is humbugged.]—(From The Journal A. M. A., July 10, 1915.)
Alfatone is a worthless alcoholic cordial. The audacity of the attempt to promote its sale by a discourse on the merits of a well-known fodder plant is the sole reason for devoting any attention to it. It is recommended that Alfatone be held ineligible for New and Nonofficial Remedies, and that this report be published.
6. That Formamint conflicts with the rules of the Council. False statements are made with regard to its composition (Rule 1); grossly unwarranted claims are made for its therapeutic properties (Rule 6), and therefore its exploitation to the public (Rules 3 and 4) is a public danger.
The amount of lactic acid or “loosely combined HCl” present in Lactopeptine is very small, since the total acid which may be titrated by sodium hydroxid and phenolphthalein is measured by 0.5 c.c. of the normal hydroxid for 1 gm. of the Lactopeptine powder, in the mean. In different samples examined the range was found to be from 0.41 c.c. to 0.6 c.c. Tests with methyl orange, methyl red and other indicators showed that the free acidity is but trifling; if the whole of this acid, as measured by phenolphthalein, were calculated to HCl, the amount would be too small to have any appreciable physiologic activity, in view of the daily dose recommended, 10 to 20 grains of the powder.
Actions and Uses.—The lecithin preparations have been recommended in many pathologic conditions, especially in malnutrition and sexual debility. Moderate doses are said to bring about a marked retention of nitrogen and phosphorus, but satisfactory proof of this is lacking. It is extremely unlikely that the small doses which have been recommended in pill or tablet form or in emulsions can have any perceptible action, in view of the fact that many of our natural foods contain much greater weights of available lecithins than the medicinal doses provide. There is no good basis for the statement that the free lecithin has a greater food value or is more readily assimilated than is the substance as found in eggs or tissue. The reverse proposition is much more likely to be true, especially when it is considered that the commercial preparations are usually somewhat altered or decomposed in the process of separation.
It is further claimed that the elixir is a “specific” for “Inflammation of the Bladder, Bright’s Disease, Renal Colic, Suppurative Nephritis, Acute Cystitis, Urethritis, Catarrh of the Bladder [it would be interesting to know what distinction the manufacturer draws between ‘Inflammation of the Bladder,’ ‘Cystitis’ and ‘Catarrh of the Bladder’], Acidemia, Edema, Vesical Catarrh of Old Age, Lithemia” and that ascites and anasarca “can be reduced greatly to the satisfaction of the patient, and honor of the physician” by using a mixture of Tyree’s Elixir and infusion of digitalis. Such claims as these do not merit serious discussion, for they carry their own refutation.
The composition claimed for Hydropsin brands it as an irrational mixture in which potent drugs are combined with, and more or less covered up by, others that are obsolete and inefficient. The name, instead of indicating its composition, suggests diseases in which it may be thoughtlessly and indiscriminately used. The claim that the danger of toxic or cumulative action has been removed, if accepted by physicians, tends to uncritical use with possible disastrous results. Hydropsin is ineligible for New and Nonofficial Remedies because of conflict with Rules 1, 2, 6, 8 and 10.—(From The Journal A. M. A., Jan. 8, 1916.)
Since firms other than the G. W. Carnrick Company are manufacturing alleged secretin preparations, and since recommendations for the use of secretin preparations in gastro-intestinal diseases have even crept into textbooks, it seemed desirable to obtain further information on certain points. The Council therefore requested Prof. A. J. Carlson of the University of Chicago to check the results of previous investigators with regard to the action of secretin administered by mouth or directly into the intestine, and, in addition, to investigate the secretin content of certain alleged secretin preparations.
But there remains the alleged favorable effect from secretin therapy by mouth in various diseases in man. It is, perhaps, impertinent for laboratory men to comment on these clinical results. The ordinary “testimonials” need not be considered, but we should like to ask the serious worker who thinks he has actually obtained good results from secretin therapy how certain he is of the causal relation between the giving of secretin or alleged secretin and the abatement of the disease.
These preparations are now described in the appendix to New and Nonofficial Remedies. Borcherdt’s Malt Olive with Hypophosphites (Borcherdt Malt Extract Company, Chicago) is said to contain in each 100 c.c., 0.64 gm. each of calcium and sodium hypophosphites, with malt extract, olive oil and glycerine. Maltzyme with Hypophosphites (Malt-Diastase Company, New York) is said to contain, in each 100 c.c., 0.4 gm. each of calcium, sodium and potassium hypophosphites and 0.005 gm. each of iron and manganese hypophosphites, with maltzyme. Maltine with Hypophosphites (Maltine Company, Brooklyn, N. Y.) is said to contain in each 100 c.c., 0.64 gm. each of calcium and sodium hypophosphites and 0.42 gm. of iron hypophosphite, with maltine. Maltine with Olive Oil and Hypophosphites (Maltine Company, Brooklyn, N. Y.) is said to contain, in each 100 c.c., 0.6 gm. each of calcium and sodium hypophosphites, with maltine and olive oil. In general, no therapeutic claims are made for these mixtures so far as the hypophosphites are concerned. The addition of hypophosphites to such mixtures is irrational and, since it tends to perpetuate the hypophosphite fallacy, detrimental to sound therapeutics.
As there is no evidence to show that strontium salicylate, calcium salicylate or a mixture of the two salts has any advantage over sodium salicylate, these claims cannot be accepted. The name and the statement of composition are objectionable in that they do not reveal the identity of the drugs in “Calcylates” and in suggesting that this preparation possesses radical advantages over salicylates in other forms.
On Dec. 9, 1915, Rachel Watkins, M. D., of Chicago, read a paper entitled “A Serum Treatment for Physiologically Defective Thyroids, With Clinical Reports” before the Stock Yards Branch of the Chicago Medical Society. The “serum treatment” discussed was Mark White’s “Goitreine” which, in the course of its checkered career, had lost its original name by the wayside. This paper appeared in the December, 1915, issue of the Illinois Medical Journal.
Kora-Konia is a white powder, slightly greasy to the touch. Qualitative tests showed the presence of boric acid, zinc, magnesium, a solid fatty acid and material insoluble in hydrochloric acid containing magnesium and aluminum. Starch was not found. Quantitative determinations gave the following results:
The value of hydrastis in the treatment of the diseases and conditions mentioned is problematical at best, and the small amount present in Hydras is wholly useless. As for the other constituents, cramp bark (Viburnum opulus), helonias (false unicorn—Chamælirium luteum or Helonias dioica) and scutellaria (skullcap—Scutellaria lateriflora) are drugs which are practically ignored by most writers on materia medica and therapeutics.99 Dogwood (Cornus florida) is a mildly astringent aromatic bitter for the use of which there is no scientific evidence.100
Calcium glycerophosphate and sodium glycerophosphate were accepted for New and Nonofficial Remedies chiefly in order that these products might be standardized. These mixtures now being defined in the new edition of the U. S. Pharmacopeia, this reason for including them in N. N. R. no longer exists. The report of Marshall (The Journal, Feb. 13, 1915, p. 573) which has the endorsement of the Council (The Journal, Sept. 30, 1916, p. 1033) shows that organic phosphorus compounds are split up into inorganic phosphates before absorption, that the animal organism can synthesize its complex organic phosphorus constituents from inorganic phosphates and consequently that the glycerophosphates, so far as their phosphorus value is concerned, are not superior to other phosphates. In fact, sodium and phosphate are more effectively administered as neutral or acid phosphate. It is evident that sodium glycerophosphate is a superfluous pharmaceutical preparation, particularly when the difficulty of obtaining a pure product and its high price is considered. So far as its calcium value is concerned, calcium glycerophosphate has no advantages over such calcium salts as the carbonate, phosphate, lactate, or chlorid. In view of the foregoing, the Council directed that sodium glycerophosphate and calcium glycerophosphate be omitted from New and Nonofficial Remedies.—(From Reports of Council on Pharmacy and Chemistry, 1916, p. 52.)
The report having been sent to Naphey & Co., the manufacturer offered, on condition that the preparation be accepted, to revise the advertising matter in minor particulars, to remove disease names from the trade package and to adopt the name Naphey’s Wafers or Naphey’s Tablets. The Council advised Naphey & Co. that the proposed names do not conform to the requirements for acceptance in New and Nonofficial Remedies because they do not indicate the composition of this pharmaceutical mixture, and moreover, that the routine use of a complex formula such as that of these tablets is irrational.—(From Reports of Council on Pharmacy and Chemistry, 1916, p. 66.)
It is obvious that any drug which would destroy the tubercle bacilli in the human lungs without exerting a toxic action on the patient would be a great contribution to medicine. But although elecampane may have been used for centuries it has proved to have little, if any, merit, and even the National Standard Dispensatory, p. 848, says: “Elecampane was formerly employed as a tonic, stimulant, diuretic, diaphoretic, expectorant, and emmenagogue, but has now largely fallen into disuse.” One looks in vain in the standard textbooks on therapeutics for a description of the uses of inula (or elecampane), and of its so-called “active principle,” helenin.
[Editorial Comment.—There are always those who are ready to exploit the unfortunate tuberculous. It is, unfortunately, a fact that many physicians accept as true, statements clothed with obscure and voluminous quasi-scientific verbiage. Such men would laugh at the bald claim that the moon is made of green cheese; when, however, one plausibly and with due solemnity, affirms that the nocturnal luminous earthly satellite is composed of an infinite aggregation of molecules of bewildering and awe-compelling complexity, built up from the recently discovered polypeptids, the whole being of a verdant tint, the person addressed looks impressed and opines that it sounds reasonable! The advertising for The Russell Emulsion and The Russell Prepared Green Bone is dangerous because it appeals to the thoughtless—layman and physician, alike.]—(From The Journal A. M. A., June 23, 1917.)
Creofos: The composition of Creosote-Delson not having been furnished, the statement concerning the composition of Creofos is also unsatisfactory (conflict with Rule 1). The therapeutic claims are unsubstantiated and grossly exaggerated (conflict with Rule 6). The name is not descriptive of its composition as is required for pharmaceutical mixtures (conflict with Rule 8). There is no evidence that hypophosphites prevent decomposition of creosote (if this occurs). Hence the inclusion of hypophosphites must be considered irrational (conflict with Rule 10).
The Council adopted the recommendation of the Committee on Pharmacology to the effect that the claims made for Trimethol are unsupported by acceptable evidence. Accordingly, Trimethol and the pharmaceutical preparations said to contain it—Trimethol Syrup, Trimethol Capsules, and Trimethol Tablets—were held ineligible for New and Nonofficial Remedies.—(From The Journal A. M. A., Aug. 11, 1917.)
While from these cases the obvious conclusion was drawn that Intramine and Ferrivine “have no specific effect on early syphilis,” these authors subsequently treated a case of tertiary syphilis with the drugs. An Intramine injection caused pain for several days but did not stop the progress of the disease. Ferrivine was then administered “not without a feeling of grave responsibility” in view of their previous experiences. They state that “the reaction which resulted in this instance was the most severe” they ever experienced after an intravenous injection of any of the antisyphilitic remedies with which they had previously worked. It is stated that “for a period of some minutes there was grave doubt as to the patient’s survival.” After resuscitation the patient passed a disturbed night, and rigors which ensued lasted until the following afternoon. The author’s report that in this case also no clinical improvement occurred and that the Intramine-Ferrivine treatment was replaced by a course consisting of salvarsan, potassium iodid and mercurial inunction.
The phosphorus content of 1⁄64 grain of strychnin glycerophosphate is ridiculously small. Yet it is asserted that this strychnin salt is of superior value because it combines the effects of strychnin with a “food-like form of phosphorus.” Eskay’s Neuro Phosphates has an acid reaction which is capitalized, thus:
“For a period of at least three years in my hospital, dispensary and private practice, I conscientiously tried out most of the balsamics on the market (including Gonosan, which I favored for some time) both alone, and combined with local injections. As a result of this study, I have come to the conclusion that the balsamics have little, if any value in the treatment of gonorrhea. During the past few years I have relied almost entirely on local therapy, and seldom prescribed any of the balsams in my private practice, certainly in not more than 5 per cent. of the cases. My results I find are just as satisfactory, and my patients appreciate the fact that they are not loaded up with disagreeable medication. Instead of the balsamics, I am using sodium bicarbonate more and more, and feel convinced that the proper use of this drug is of more value than all of them combined.”
“Responding to your letter of March 10th, we beg to suggest that literature covering the different matters at issue are readily available to your referee, and all statements emanating from us are made advisedly.
“If you cannot satisfy yourselves that this preparation is a scientific product, ethically advertised, and a desirable advance in therapeutics, you can only delete it from your next issue of New and Nonofficial Remedies.”
A consideration of the negligible food value of NuTone as well as of the inefficiency of the other components and the claim that it is indicated in “malnutrition,” “wasting diseases” and “incipient phthisis” classes NuTone with that large group of shotgun mixtures which do harm in that dependence is placed on them in conditions in which the patient will probably be restored to health if proper medical and hygienic measures are adopted in time.
Venosal is recommended for the treatment of “rheumatism,” meaning, the context would indicate, infectious rheumatic fever. As colchicum has no special action on this disease and as there is no apparent reason for the employment of the trace of iron present, these additions in fixed proportions are unscientific, if not absurd. According to the advertising matter:
Subsequently, A. J. Carlson and his co-workers, at the request of the Council, studied the question of the stability of secretin and demonstrated (The Journal A. M. A., Jan. 15, 1916, pp. 178 and 208) that commercial secretin preparations contained no secretin and, further, that secretin given both by the mouth and even in enormous doses directly into the intestine is entirely inactive.
Surgodine (Sharp and Dohme, Baltimore, Md.), according to an advertising pamphlet, is a solution of 21⁄4 per cent. of iodin in alcohol, containing no alkaline iodid, but miscible with water in all proportions. The A. M. A. Chemical Laboratory reports that Surgodine is an alcoholic liquid (containing 91.8 per cent. alcohol by volume) containing free iodin, combined iodin and free acid, probably hydrogen iodid (hydriodic acid). Quantitative estimations gave 2.51 gm. free iodin per 100 c.c. and 1.78 gm. combined iodin (the greater part apparently was present as hydrogen iodid).
In those cases of hemorrhoids in which palliative measures may be expected to enable the patient to avoid surgical interference and afford relief from attacks, the object should be to secure cleanliness, to avoid irritation, whether it be by friction or irritating fecal matter, to reduce inflammation by astringents and, when necessary, to relieve pain by analgesics. If an antiseptic dusting powder is desired, boracic acid in impalpable powder with talc may be employed; if an astringent, finely powdered oxid of zinc may be added; if a local analgesic is necessary, a little extract of belladonna may be incorporated with petrolatum or other ointment base. The main reliance, in any event, should be to effect normal bowel movements by regulating the diet rather than by the use of purgatives; the use of warm water to insure cleanliness; the avoidance of irritation, especially that caused by friction and secretions; a mild astringent to reduce inflammation.—(From The Journal A. M. A., March 9, 1918)
The Intravenous Products Company claims that Guaiodine is made by an “electro-chemical process of preparing colloidal iodine,” discovered by one E. B. Page, and that by this process the tendency of iodin to produce iodism has been “overcome.” It is said to be “pre-eminently an antiseptic and germicide.” Guaiodine is a dark brown, oily liquid with a specific gravity of 0.9845 at 15.6 C. and an odor suggestive of guaiacol. Its solubilities were those of a fat. Free iodin was absent in the recently purchased specimen (traces were present in an older one). Steam distillation indicated that the product consisted of volatile and nonvolatile constituents. The volatile matter was concluded to consist, in the main, of guaiacol or some guaiacol-like body, and the nonvolatile matter to be an iodized fatty oil. Quantitative determinations indicated that Guaiodine contained about 7.25 per cent. of iodin in combination, and that it is composed approximately of 3 per cent. volatile matter and 97 per cent. nonvolatile matter. Hence Guaiodine appears to be an iodized fatty oil to which a small amount of guaiacol or some guaiacol-like substance has been added.
On the recommendation of the Committee on Serums and Vaccines, the Council declared “Sherman’s Mixed Vaccine No. 40” ineligible to New and Nonofficial Remedies because the therapeutic claims made for it are unwarranted (Rule 6) and because the combination, in view of its complexity, is irrational and detrimental to sound therapy (Rule 10).—(From The Journal A. M. A., June 23, 1918.)
Katharmon is in conflict with Rules 1 and 4 of the Council on Pharmacy and Chemistry because of its indefinite and secret composition and the method of advertising it indirectly to the public; it is in conflict with Rules 10, 6 and 8, in that it is an irrational shotgun mixture sold under unwarranted therapeutic claims and under a name nondescriptive of its composition.—(From The Journal A. M. A., Aug. 10, 1918.)
Iodinized Emulsion (Scott) is not a “pharmaceutical triumph”; it is an irrational mixture—a reminder of a decadent polypharmacy—sold under misleading and unwarranted claims. It is inadmissible to New and Nonofficial Remedies for conflict with Rules 1, 6, 8 and 10.
The action of such a mixture as a whole is practically that of the sum of the actions of its constituents. The therapeutic action of strychnin and quinin are described in every text-book of therapeutics, but it is necessary to distinguish carefully between the various conditions in which these alkaloids have been used without discrimination, and those conditions in which they have been proved to be of value. While both have been widely used in a great variety of conditions, neither is of proved value in more than a distinctly limited range of diseases. The manufacturers of Phillips’ Phospho-Muriate of Quinine Comp. seem to appeal to the less discriminating who use these alkaloids without any definite conception of exactly what they seek to accomplish with them. Quinin, although used by the uncritical in a host of diseases, has a definite field of usefulness in the treatment of malaria, both prophylactic and curative, but the required dose in the treatment of malaria is many times larger than that recommended in the Phillips’ preparation. The claim that the “strychnin and quinin in this preparation are so well balanced that they produce a mild, buoyant effect, so advantageous, instead of alcoholic stimulation, to relieve depression and fatigue from mental or physical exertion” is nonsensical, if, indeed, it is not mendacious balderdash.
No reply to this letter was received, and hence a copy of the letter was sent to Schieffelin and Company and also to Rufus Crowell and Company with the explanation that unless the statements in the Colalin advertising which the referee had questioned were substantiated by satisfactory evidence, were suitably revised, or else the advertising matter withdrawn pending revision, the referee would be obliged to recommend to the Council that Colalin be omitted from New and Nonofficial Remedies.
To determine if “one and all” of those who had used Foral were still using the preparation, four of the testimonials, appearing in an advertising pamphlet, were investigated. The pharmacist of the hospital from which the first of these testimonials was stated to have emanated replied that the person whose name appeared in connection with it had left the hospital about ten years ago and that no depilatory preparation has been used in this hospital for some time. So far as he knew, depilatories were not now in use in the surgical wards of the hospital. In regard to the second testimonial, the pharmacist of this hospital wrote that the hospital had not bought the preparation, but that some of it had been obtained for an elderly deaconness, who had personal use for a depilatory. The physician signing the third testimonial replied that the preparation was effectual for the removal of hair from the scalp, but that “... we have gotten out of the habit of using it.” In the case of the fourth testimonial, its asserted author wrote “... if it is applied in too large a quantity or too concentrated, or permitted to remain on too long, it will vesicate. It was for this reason chiefly that I discontinued its use. It is a very bad smelling mixture and patients complain of it very bitterly.”—(From Reports of Council on Pharmacy and Chemistry, 1918, p. 55.)
In 1916, the Council voted to omit Maltzyme with Hypophosphites, and Maltzyme with Phosphate of Iron, Quinine and Strychnine. At that time the labels used on the Maltzyme preparations still in New and Nonofficial Remedies contained a list of Maltzyme combinations which included those which had been dismissed. As the Council does not permit an accepted article to be used as a means of advertising an article not accepted, it voted to continue the following preparations for a period of three years on condition that reference to the deleted articles be omitted from the labels when those then in stock had been used up: Maltzyme, Maltzyme with Cascara Sagrada, Maltzyme with Cod Liver Oil, Maltzyme Ferrated and Maltzyme with Yerba Santa. While the Maltzyme Company made no definite agreement to revise its advertising propaganda in accordance with the Council’s requirements, the Maltzyme preparations were retained in the belief that in due time the required revision of the labels would be made.
As explained in the report which follows, “Stanolind Liquid Paraffin” was omitted from New and Nonofficial Remedies at the request of the proprietors. Announcement of this omission was made in the preface to New and Nonofficial Remedies, 1918, but publication of the Council’s report was postponed pending actual conflict with the rules. The Council now authorizes publication of the report because a circular indirectly advertising the product to the public was found enclosed with the trade package of Stanolind Liquid Paraffin.
If these quotations mean anything, they imply that these tablets present a distinct advance in digitalis therapy. There is no warrant for such a claim. The statement with reference to the occurrence of an oil in digitalis is partly false and partly misleading. Tincture of digitalis, which the tablets are claimed to represent, is fat free; the fixed oil that is present in the drug is not soluble in 70 per cent. alcohol, the menstruum used for the preparation of the official tincture of digitalis. Furthermore, a fairly large amount of this oil (such as is present in 100 therapeutic doses of the drug) is incapable of causing gastric disturbance. Gastric disturbance is a side action that is inseparable from slight overdosage with all true digitalis bodies and is not in any way due to local gastric action. The claim that such action is prevented by the use of enteric pills or tablets is obviously false and misleading.
When the Laboratory’s findings with regard to Xeroform-Heyden and the action of the Council deleting the article from New and Nonofficial Remedies was reported to the Heyden Chemical Works, the firm expressed regret that efforts to produce a product equal to that formerly obtained from Germany had so far not been successful and announced that it had decided to withdraw Xeroform-Heyden from the market for the present.
With the offer “to supply you with as much literature as may be necessary and as little of the actual remedies as may be desired” if “the prospects for the inclusion of these formulas in N. N. R. are good,” Henry R. Harrower sent the Council a booklet descriptive of his preparations and labels for the following mixtures:
“To sum up, Cuprase has given positive results in about 94 per cent. of the cases in which it has been employed for a sufficiently long period, and some brilliant results in about 20 per cent. of these cases. Therefore, it may be affirmed, that among the internal remedies for cancer, Cuprase is the one which has produced the most successful results, and can, under certain circumstances, compete with surgical methods, even, so far as the rapidity of their results are concerned.”
“Pulvoids Calcylates Compound (Sugar coated orange color)” is advertised (Medical Times, January, 1919) as being “Analgesic-Antipyretic and Diuretic,” and is included in the preparations designated by the advertiser as “Approved Remedies for LaGrippe and ‘Flu.’ ” The claim that “Their tolerance is remarkable” refers not to the physicians who tolerate such products, but to the alleged fact that Pulvoids Calcylates are tolerated remarkably well. The advertisement continues:
Rheumatism:—Proteogen No. 2.—The Doctor has one case being treated with No. 2. She has improved so rapidly she cannot express her pleasure, and will continue for some time on the treatments. She is a patient who was confined during the time she suffered from a rheumatic illness, and it seemed to affect her mental condition. This condition is clearing up also, very much to the pleasure of both patient and doctor.—November 27, 1918.
Influenza:—Proteogen No. 12.—First day, temperature 102, gave 1 c.c. Proteogen No. 12; second day, temperature 100, gave 1 c.c. Proteogen No. 12; third day, temperature 98.8, gave 1 c.c. Proteogen No. 12, and then discharged the case as recovered.—October 31, 1918.
Asthma:—Proteogen No. 4.—Splendid results obtained from a sample of Proteogen No. 4. Three ampoules affected [effected?] complete recovery.—October 9, 1918.
Cancer:—Proteogen No. 1.—Mrs. B. pronounced recovered from Cancer by Dr. O. W. A., of Catlin, after having injections of Proteogen No. 1 for some time.—October 4, 1918.
Eczema:—Proteogen No. 5.—Tried No. 5 on a patient with eczema, and with happy results. Have not done anything for him for about five months—and he is now at his business. Proteogen No. 5 also RELIEVED HIM OF CONSTIPATION AND WHAT HE CLAIMED A TRAUMATIC STRICTURE OF THE LOWER PORTION OF SIGMOID FLEXURE. He is sure pleased and recommending them to his friends. (Proteogens).—February 17, 1919.
Syphilis:—Proteogen No. 10.—I am getting such excellent results with the No. 10 Proteogen for Syphilis that I am badly in need of more, as I am treating so many cases. Please send me four dozen C. O. D.—October 9, 1918.
Enlarged Prostate:—Proteogen No. 1.—Have used Plantex in four cases, with good results in each case. One of them his father, an elderly man.—April 25, 1918.
Lobar Pneumonia:—Proteogen No. 12.—The only case I have used Proteogen No. 12, was a man who had Lobar Pneumonia of left lung following Influenza. After crisis came, patient continued to have slight rise in temperature, cough, and after using 10 doses of your Proteogen No. 12, temperature was normal, cough very much better, patient began to take on flesh and is still improving.—December 26, 1918.
Tuberculosis:—Proteogen No. 3.—The Doctor writes: The Proteogen No. 3 sent me worked wonders in my patient. The case came under my care when he was too far gone for anything to benefit him a great deal, but the Proteogen did for him more than anyone could have expected, yet he died leaving me with a few ampoules to try on the next patient.—September 20, 1918.
Gonorrheal Cystitis:—Proteogen No. 11.—My patient has taken two boxes of your Proteogen No. 11 given for gonorrheal cystitis of probably two years’ standing and at this writing I consider her almost, if not entirely, cured which I think speaks very highly of your remedy. I expect to use more of your preparations in the future.—April 12, 1919. [This testimonial, either by clerical error, or because the results were considered remarkable, was repeated elsewhere in the material submitted by the Merrell Company.]
Acute Gonorrhea:—Proteogen No. 11.—Mr. A. E. R., age 65, weight 140 pounds. First attack. Had had no previous treatment. Came to me January 2, 1919. Had discharge, all acute symptoms, burning, etc. Gave seventeen injections of Proteogen No. 11, also mild antiseptic urethral wash. Discharged on February 15, 1919, clinically cured.—April 11, 1919.
Epithelioma of Buttock.—Proteogen No. 1.—I used Proteogen No. 1 on an epithelioma of buttock some six months ago with favorable results and no return of symptoms as yet.—April 13, 1919.
So much for the facts; yet the physician is asked to jump from this region of solid fact into a sea of hypothesis; to believe that small amounts of the well-known drugs thyroid and pituitary, plus an unknown amount of unknown hormones of the testes and ovary are of great value in conditions that in themselves are often purely hypothetical. He is asked to believe that this combination has virtues in such conditions as “hypofunction of the adrenal system,” neurasthenia, the “fatigue syndrome,” amenorrhea, dysmenorrhea, “natural and artificial menopause,” sexual neuroses, cold extremities, cardiac asthenia, low blood pressure, infantilism, sterility, melancholic conditions, obesity, anorexia, anemia, slow metabolism, constipation, psychasthenia, lowered virility and the sexual neuroses of the unmarried, hysteria following functional exhaustion of the nerve centers, frigidity, etc., etc., especially if he guesses that the trouble is due to a “pluriglandular disturbance,” “glandular hypofunction,” an “adreno-pituitary deficiency,” suboxidation, etc.
Direct Application to Wounds.—The widely quoted results of the gynecologist K. Abel, on the footpad of cats, were found to be quite unreliable. When the experiment is properly controlled, the results are either negative or the bleeding may be increased. Quantitative experiments on wounds of the footpad of dogs showed that cotarnin invariably increased the bleeding. Equally negative or unfavorable results were obtained with wounds to the comb of roosters, and to the liver and spleen.
In addition to “Micajah’s Medicated Wafers,” Micajah & Co. also put out “Micajah’s Suppositories for Hemorrhoids.” These have been examined in the A. M. A. Chemical Laboratory and, like the “Medicated Wafers” have been found to contain alum, boric acid and borax—and these substances practically alone—incorporated in cocoa butter. The company claims that “to these have been added Ammonii Ichthyosulphonate, Balsam of Peru, Ext. Belladonae.” The A. M. A. chemists report, however, that if extract of belladonna is present at all it is in amounts too small to be detected by the method commonly employed in the chemical examination of alkaloidal drugs. The chemists report further that while ammonium ichthyosulphonate and balsam of Peru both have a decided odor and are dark in color, the suppositories have but little color and the odor of the cocoa butter that forms their base is not covered by these drugs; obviously, therefore, if ammonium ichthyosulphonate and balsam of Peru are present at all it is in amounts utterly insufficient to exert any therapeutic effect.
The A. M. A. Chemical Laboratory reports that Alkalithia is an effervescent mixture which contains alkaline carbonates and bicarbonates together with caffein, free tartaric acid and free citric acid. The major portion of the alkali carbonates and bicarbonates is converted into citrates and tartrates when the preparation is dissolved in water—as is done before it is taken. An excess of alkali is present, however, as the solution has an alkaline reaction. Each “heaping teaspoonful” (which was found to be about 4.85 Gm.) contains about 0.044 Gm. of caffein (the manufacturers claim 0.0648 Gm. per heaping teaspoonful). As taken, Alkalithia, therefore, represents caffein in a solution of alkali tartrate, citrate and bicarbonate containing free carbonic acid. If it is assumed that all of the tartrate and citrate in Alkalithia is converted into carbonate in the organism, a “heaping teaspoonful” of Alkalithia would represent about 2.9 Gm. of sodium bicarbonate. This assumption is, however, not correct, for it is known that tartrates are not completely converted into carbonates in the organism.
The Council declared “Chloron,” “Chlorax” and “Number ‘3’ ” in conflict with the rules governing admission to New and Nonofficial Remedies. All are of unreliable composition (conflict with Rule 1). The therapeutic claims made for the preparations are not substantiated by acceptable evidence and are unwarranted and misleading. Chloron is inferior as an antiseptic to the well-known surgical solution of chlorinated soda on account of its low chlorin content and uncontrolled reaction. There is no warrant for the claim that Chlorax is useful in the treatment of “Kidney Conditions,” “Diabetes,” “Acute Infections,” “Blood Dicrasias,” “Lithemias and Rheumatism,” and “Nervous Conditions,” nor is there warrant for the claim that “Number ‘3’ ” is a remedy for the purification of the blood or a “Syphilis remedy” (conflict with Rule 6).
As a matter of fact, Joachimoglu found that very little arsenic was absorbed when Elarson was given to dogs and rabbits; most of it was recovered from the feces; only traces were found in the liver and kidneys and none in the blood and brain. The absence from the latter organs shows that the lipoid solubility does not obtain in the body. It is claimed in the circular that Elarson has the advantage over Fowler’s solution “in that it is free from any irritating action upon the gastro-intestinal tract”; it is stated that as many as sixty tablets have been given to dogs daily without any toxic effects. Joachimoglu, on the other hand, found powdered Elarson to be very irritating to the gastro-intestinal tract; also that the dog could not stand sixty tablets at all (gar nicht vertragen), such doses causing vomiting, diarrhea and intestinal hemorrhages; on repeated administration the symptoms became progressively more severe. Joachimoglu also found that, compared on the basis of arsenic content, Elarson, given intravenously, is from ten to twelve times as poisonous as arsenic trioxid. Elarson is recommended for the class of cases in which Fowler’s solution is used.
Soamin is the name under which the firm of Burroughs Wellcome and Company sells its brand of sodium arsanilate. The Council directed the omission of Soamin from New and Nonofficial Remedies and authorized publication of the report which appears below after the proprietors of the product had declined to withdraw or suitably revise the unwarranted therapeutic claims which it made.
Mr. Lackenbach states that these bacterial mixtures were prepared for him by E. R. Squibb & Sons. Their sale in interstate commerce is permitted under the license granted to the latter firm by the U. S. Treasury Department. However, no evidence of any kind was presented to the Council proving the therapeutic efficacy of the several mixed vaccines. As a mixture of two or more kinds of organisms is accepted for New and Nonofficial Remedies only if there is satisfactory evidence that its therapeutic use is rational, the Council declared the several vaccine mixtures ineligible for New and Nonofficial Remedies (Rule 10).—(From Reports of Council on Pharmacy and Chemistry, 1919, p. 90.)
The quantities of quinin, iron and nuclein in the doses represented in these mixtures are negligible; thus, one tablet of Triple Arsenates with Nuclein containing 1⁄64 grain of quinin arsenate contains only about 1⁄90 grain of anhydrous quinin; the tablet containing 1⁄64 grain of iron arsenate contains 1⁄210 grain of iron; 4 minims of the nuclein solution (assuming it to be the “Nuclein Solution-Abbott”) would contain but 2⁄5 of a grain of nuclein—a substance which even in large doses is of questionable therapeutic value. The amounts of iron and nuclein contained in doses of this preparation are insignificant in comparison with the amounts present in ordinary foods. The only substances present in even small therapeutic doses are strychnin and arsenic. The effects of arsenic and strychnin are very different and there are comparatively few conditions in which they should be prescribed at the same time. Hence a preparation containing these two in fixed proportions is illogical.—(From Reports of Council on Pharmacy and Chemistry, 1919, p. 92.)
A year later, a circular letter sent out by A. Klipstein and Company reiterated the claim that the asserted decomposition of Dial “Ciba” in the body prevents after-effects, the drug being still contrasted with barbital (“veronal”). In view of the reiteration of this highly improbable claim, the referee undertook to study the comparative action of Dial “Ciba” as compared with other hypnotics. It was found that the actions of Dial “Ciba” are not distinguishable, qualitatively, from those of barbital, there being no perceptible difference in the after-effects or in the nature of the side actions. In toxic doses, both caused profound depression with the temperature falling to that of the room (or about one degree above), the respiration being extraordinarily slow and shallow as one would expect with lowering of the temperature. There were also the same evidences of nausea that are so frequently seen after toxic doses of the various hypnotics of this group. In view of these results, the Council declared that it is unwarranted to claim freedom from after-effects for Dial “Ciba.”
It has been shown that when toxicity tests of local anesthetics are made on cats these animals seem to respond to the drugs in a manner more closely approximating humans and it is a suggestive fact that the more toxic of local anesthetics, as shown by tests on cats, have been found the most dangerous in clinical use. The absolute toxicity of Apothesine has been measured by Eggleston and Hatcher132 by the intravenous injection in cats. The fatal doses, in terms of milligrams per kilogram ranged as follows:
It is unscientific (Rule 10). Eumictine is composed of hexamethylenamin, salol and sanalol in fixed proportions. Hexamethylenamin may serve a useful purpose in some forms of infection of the urinary tract, but neither it nor salol is of any considerable value in gonorrhea. It is now known that the balsamic preparations, formerly so widely used, do not have the curative effects in gonorrhea and associated conditions that used to be ascribed to them. To combine three substances, none of which has any distinct therapeutic value in the conditions for which Eumictine is proposed, does not enhance their value. There is nothing original in the combination used in Eumictine, or in the manner of dispensing it.
Supsalvs are advertised by the Anglo-French Drug Company as “stable suppositories of ‘606’ (of French manufacture)” with the claim that by the rectal administration of these suppositories the effects of arsphenamine may be obtained. The asserted efficacy of Supsalvs medication is based in part on the claim that for these suppositories an excipient was found which mixes with the cocoa butter base “to form an assimilable emulsion.”
Object: To test the claims of thromboplastic and hemostatic activities.
Claims: Coagulen is alleged to be a “physiological styptic prepared from the natural coagulants of animal food contained in the blood platelets. It has the characteristics of a lipoid.” (If cephalin is meant it is difficult to understand why platelets should be selected in preference to other abundantly supplied organs such as brains).
“Coagulen is indicated in all cases of external and internal hemorrhage due to a deficiency of the coagulating power of the blood: epistaxis, hemophilia, hemorrhage from gastric or duodenal ulcer, melaena neonatorum, hemorrhage from the gums, the lungs, the bladder, the uterus, hemorrhage during or after operations (turbinectomy, tonsillectomy). It has also been used as a prophylactic before operations, likely to produce severe hemorrhage.”
“In cases of true hemophilia one application of 5 grains of coagulen usually suffices to control the hemorrhage.” “In gastric and intestinal hemorrhage the internal administration of coagulen will be found effective.” “In bonegrafting, plastic surgery, dentistry and nose and throat surgery the application of a 10 per cent. solution of Coagulen will be found to be of valuable assistance in controlling hemorrhage and oozing.”
“It is a non-toxic and non-irritating powder to which a certain amount of sugar has been added, with a view to ensuring its prompt solution in water or physiological sodium chloride solution.”
Description: “Coagulen is a yellowish granular powder with but slight odor, a sweet taste and is readily soluble in water or a normal salt solution.” The dry Coagulen obtained corresponds to the description claimed. Old specimens show the presence of dark brown particles. Coagulen is marketed in 3 forms: (1) as dry powder containing lactose, which, it is claimed, facilitates solution in water; (2) as 3 per cent. sterile solution in ampoules;137 (3) tablets.
Methods of Study: The alleged thromboplastic activity was tested by the method of Howell and a modification of this method by Fenger as described in “New and Nonofficial Remedies.” In the Howell method dog or cat blood is used, while beef blood at body temperature is used in Fenger’s method. In other respects the methods are essentially the same. Briefly these consist of noting the acceleration of coagulation time in a mixture of equal parts of serum and the thromboplastic agent to which about an equal part of oxalate plasma is added. Under these conditions cephalin causes clotting in about 1 minute or even less as compared with 20 to 30 minutes or more of the control.
The effects were compared with freshly prepared cephalin and other thromboplastic agents, using saline (0.9 per cent. NaCl) as control. The effect of different concentrations was also studied.
The literature of the manufacturers claims that Coagulen is harmless. This was tested by making intravenous and subcutaneous injections into guinea-pigs, using saline and cephalin as controls.
Bloods of 4 different species were used, namely, cat, dog, beef and human. Dog’s peptonized blood and plasma were also tried.
The 15 different tests that were made in vitro were carried out with 3 different samples of fresh dry Coagulen (from manufacturer), 2 old samples (one from Council on Pharmacy and Chemistry and one of our own), 3 fresh specimens of sterile solution in ampoules (from manufacturer), one old specimen and 4 small ampoules (Council on Pharmacy and Chemistry).
The tablets were not tested since these are made from dry Coagulen and the results would hardly be expected to show anything different.
Results: The results obtained may be briefly summarized as follows: (1) 0.1 per cent. to 5 per cent. Coagulen did not accelerate the coagulation time of blood and oxalate plasmas in the majority of tests any more than the controls of saline, while 0.1 per cent. cephalin was found to shorten the coagulation time from 1⁄3 to 1⁄2.
(2) There was no difference between the behavior of old and fresh specimens.
(3) No acceleration of coagulation in vitro was observed even with the highest concentrations tried, namely 25 and 50 per cent.
(4) Irrigations made with fresh dry coagulen in solution and sterile solution in ampoules on superficial bleeding from the foot-pads of 3 normal and peptonized dogs and local application to hemorrhages from dissected femoral arteries and bone and liver wounds of 3 dogs showed that coagulen was no more active than normal saline.
Toxicity: Subcutaneous and intravenous injections of different doses of Coagulen solutions (fresh ampoules) and dry Coagulen in solution in 8 guinea-pigs produced definite anaphylactoid symptoms with injury to the circulatory and respiratory systems as indicated by cardiac dilatation, abdominal congestion and pulmonary hemorrhages, congestion, distention and sometimes thrombi. On the other hand, the control animals injected with saline and cephalin remained practically unharmed.
Conclusions: The results obtained justify the following conclusions:
(1) Coagulen is entirely inactive as a thromboplastic and hemostatic agent.
(2) Coagulen is distinctly injurious when injected systemically.
(3) The claims of hemostatic efficiency and harmlessness for Coagulen by the manufacturer appear exaggerated and unjustified.
Recommendations: Because of its uncertain composition, the possible dangers when injected systemically, and its inactivity as a thromboplastic and hemostatic agent when tested by several different methods, Coagulen merits no recognition as a therapeutic agent for inclusion in New and Nonofficial Remedies.
The detail evidences used as the basis of this brief report concerning Coagulen will be published shortly in the Journal of Pharmacology,138 together with the results with other thromboplastic agents.
“As far as the Referee knows, the only claim that Iron Cacodylate has as a therapeutic agent is that it forms a convenient method for the administration of Iron and Cacodylate (while there is no reason why a drug should not be given by mouth, usually intramuscularly, and apparently it has recently been given intravenously). The effects to be expected from its use are those of iron and arsenic.
“Granted that iron and arsenic are valuable therapeutic agents, Iron Cacodylate is not a satisfactory preparation in which to administer these drugs for the following reasons:
“1. It would appear that Cacodylates are not the best form in which to administer arsenic. Cacodylates in therapeutic doses exert but a feeble action. Small quantities may be reduced to cacodyl (CH2)4As2, and varying amounts to inorganic arsenic. The amount transformed to arsenic is apparently unknown and probably varies in different individuals. On these grounds alone the use of the cacodylates where an arsenic effect is desired seems dubious.
“2. The amounts of iron and cacodylates contained in the doses recommended are small when compared with the usual doses of either iron or cacodylate. The amount of iron in the Iron Cacodylate preparations is small, about .0036 gram per dose, while the preparations admitted to ‘Useful Drugs’ contain much larger amounts per dose recommended. The list follows:
Massa Ferri Carbonates
Fe per dose
.042 gm.
Pilulae Ferri Carbonates
"
.058 gm.
Tinctura Ferri Chloride
"
.022 gm.
Ferri et Ammonii Citrae
"
.042 gm.
“The approximate amount of arsenic in Iron Cacodylate in the commonly recommended doses varies from .012 gm. to 0.024 gm., while the amount of arsenic in Sodium Cacodylate in the recommended doses varies between .021 and .35 gms. It would seem that a much more rational method of administration of these two drugs would be separately, in which case a better control over the dosage is possible.
“3. The Referee has been unable to secure reliable clinical evidence that Iron Cacodylate is a serviceable preparation. A search of the available literature for the past fifteen years has been made, also Drs. Edsall, Longcope, Stengel, Hoover, Phillips and Miller have been consulted. These physicians know nothing of its use.
“4. In view of the above, it appears to the Referee that Iron Cacodylate is an irrational and useless method of the administration of iron and arsenic.”
Granting, for the sake of argument, that carefully controlled experimental clinical evidence were available to substantiate this statement with reference to a single case of pain, the statement would be misleading when considered as a general explanation of the preparation’s relieving pain by removing the cause of pain when taken in connection with the conditions for which it is recommended and in which pain is even a minor symptom. Still, if pain were relieved in these cases by removing the cause, the patient would be cured of the conditions which give rise to the pain, and these include: “Acute pain in the chest;... acute inflammation in the chest;... persistent local pain;...” (This might be interpreted as including tuberculosis; pneumonia; cancer, and appendicitis.) “lumbago; sciatica; articular rheumatism” (gonorrheal infections?).
The Council’s two consultants were also asked whether or not, in their opinion, the administration of mercury by inhalation is a method which the Council should endorse to the extent of recognizing a preparation based on this principle. This inquiry was also sent to the members of the editorial board of the Archives of Dermatology and Syphilology. Five replies were received. One advised a thorough study of the different methods of administering mercury by inhalation. The other four were opposed to such recognition on the ground that as the dosage is not exact the effects, therefore, are not certain.
This claim scarcely needs comment, since it is well established that the chief “disadvantages” of digitalis are inherent in the principles which produce the desired effects of digitalis and may be avoided to a large extent by a carefully regulated dosage of any digitalis preparation. In short, the advertising for Digifolin asserts that this digitalis preparation has all the advantages of digitalis itself, but none of its disadvantages. This claim has been refuted so frequently that manufacturers must be aware that it is untenable. Further the claims now made for Digifolin are essentially those made nearly four years ago at which time the attention of the American agent was called to their unwarranted character.
The Council has recognized intravenous preparations which satisfied these requirements. It is evident, however, that hexamethylenamin, sodium iodid and sodium salicylate do not. When given orally they do not undergo material decomposition in the digestive tract, they are rapidly absorbed, they cause no direct local reaction, and in the conditions in which they are used the hour or so which is required for absorption is immaterial, especially as they are used continuously for some time. Mercuric chlorid does indeed produce some local irritation, but there is as yet no convincing evidence that its intravenous injection causes less injury than oral administration. More experience under controlled conditions is needed before the intravenous use of mercuric chlorid can be approved. Especially objectionable are the fixed proportion mixtures of sodium iodid with sodium salicylate and with hexamethylenamin. The dosage of all three drugs has to be adapted to individual conditions. This is impossible when giving them in fixed proportions.
These inconclusive opinions certainly do not agree with the favorable impression which other portions of the manufacturer’s literature create. If the factor of natural recovery in the conditions represented by these seven cases is given due weight, little, if anything, is left to the credit of “oxyl-iodide.” Such clinical evidence as is supplied by the manufacturer indicates that the therapeutic efficiency of “oxyl-iodide” is doubtful, and not an improvement over either cinchophen or iodid.
Further, Atophan is recommended “In Pyorrhea Alveolaris as a systemic support to local and specific measures.” Atophan is not indicated here. Pyorrhea requires local medication, if anything at all. It could exert no local beneficial effects in this condition; indeed, the employment of Atophan might lead to irritation. Good dental treatment is more essential than medication.
It was found that the pamphlet contained a number of unwarranted statements. Particularly objectionable are the claims made for the use of Urotropin as an antiseptic in body fluids that are alkaline, such as the cerebrospinal fluid, bile, aqueous humor of the eye, saliva, the excretions caused by middle ear infection and other excretions of the nasal, bronchial, laryngeal and mucous membranes. The lack of efficacy of hexamethylenamin in alkaline secretions is generally admitted and the clinical references to the use of hexamethylenamin in the pamphlet are obsolete. In the introduction to the pamphlet, Schering and Glatz state that they are well acquainted with the scientific research work discrediting the efficiency of hexamethylenamin in nonacid mediums, but that they feel that the accumulated evidence for its efficacy in such conditions should not be “brushed aside.” However, the pamphlet is not made up of quotations, but of unqualified statements. With one exception, all references to the antiseptic properties of the drug in alkaline mediums are previous to 1913, that is, before the importance of reaction of the medium was fully appreciated. To quote these earlier articles without regard to the later work, which in most eyes discredited them, constitutes in effect an exploitation of this brand of hexamethylenamin under unwarranted therapeutic claims.
While, as stated a moment ago, the laboratory has encouraged the sending of inquiries and has earnestly striven to furnish the information asked for, it is obvious that the amount of chemical work which can be done is limited. The small size of the laboratory force, consisting of three chemists engaged in actual analytical work, makes it necessary to select for investigation those problems which shall be of general interest to the medical profession. As the American Medical Association is national in its scope, the laboratory has held that it can do analytical work only when such work will be of general interest to physicians and of value both to the medical profession and the public. In view of this it has refrained from undertaking analyses which would benefit only the physician making the inquiry and possibly his patient. The laboratory further has not felt justified in undertaking work of merely local interest; instead it has used its endeavors to secure the investigation of such local problems by municipal or state authorities.—(From The Journal A. M. A., Nov. 25, 1916.)
Inasmuch as it has been demonstrated by Carlson and Woelfel (Carlson, A. J., and Woelfel, A.: Solubility of Lead Sulphate and Basic Lead Carbonate in Human Gastric Juice.... In Hygiene of the Painter’s Trade by Alice Hamilton, Bull. of U. S. Bureau of Labor Statistics No. 120, May 13, 1913, pp. 22–32) that even small quantities of lead sulphate when taken into the system for a long time, have produced lead poisoning, the laboratory deemed it important that the products be examined for lead.
The bottle is not flexible and weighs 31⁄2 pounds. The contents consisted essentially of sodium acetate. This salt melts when heated. When it cools the temperature inside the bottle is relatively constant, as it will remain at the “freezing point” until all of the sodium acetate has solidified. The duration of the time that it remains warm when well wrapped is simply in inverse proportion to the conductivity of the surrounding environment. When two ordinary towels were carefully arranged about it, the air between the bottle and the wrappings was maintained at a temperature of 40–50 C. (104–122 F.) for a period of eight hours.
At the time the specimens were received from the Mulford Company, the firm reported experiments which were under way to determine the keeping qualities of the solution. These experiments indicated marked deterioration of the specimens, which had become red from permanganate formation, and also that one specimen, which had not become red, had lost 5 per cent. of its available chlorin in one month. The Mulford Company explained that when sufficient data had been accumulated, a decision would be made either as to placing a time limit on the solution or making a claim as to the rate of deterioration. When the extreme deterioration found by this laboratory was reported to the Mulford Company, the firm replied that this was a much greater loss than the average deterioration found in its chemical laboratory, namely, an average of 10 or 12 per cent. per year. It advised that because of the instability of concentrated solution of sodium hypochlorite, its manufacture had been discontinued.—(From Reports A. M. A. Chemical Laboratory, 1918, p. 81.)
III. Arsenic and Antimony.—About 3 gm. of the powdered specimen was digested with sulphuric acid in a Kjeldahl flask. One-half portion (which was evaporated almost to dryness and treated with 5 c.c. of concentrated hydrochloric acid) was submitted to treatment with hydrogen sulphid, diluted, and saturated with hydrogen sulphid. The precipitate was treated in the usual manner of the group separation with warm ammonium sulphid solution. The filtrate from this treatment was acidulated with hydrochloric acid, the precipitate removed, and treated with concentrated hydrochloric acid. The substance insoluble in hydrochloric acid was treated with more concentrated hydrochloric acid and a crystal of potassium chlorate. The solution was tested after the Gutzeit method of the Pharmacopeia IX, for arsenic. A very small amount was indicated. The hydrogen sulphid test was not indicative. The solution which might contain the antimony was tested with hydrogen sulphid. In one case only was a slight orange coloration produced. No antimony was deposited on platinum foil in the presence of granulated zinc. These tests were run in triplicate.
Cerelene is another compound wax for the treatment of burns. According to the work of Sollmann (J. A. M. A. 68:1799, 1917) it is highly improbable that compound mixtures have any advantage over simple paraffin of low melting point. Cerelene must therefore be considered as an unessential modification of paraffin, and as in conflict with Rule 10, unless definite evidence of superiority be submitted. Cerelene mixtures containing medicinal ingredients also appear unscientific since the evidence that the ingredients do not leave the wax has not been successfully contradicted. Finally, the claims made for Cerelene are rather extreme, and would need some revision before they could be accepted.
From this examination we conclude that DeSanctis’ pills contain powdered colchicum seed, benzoic acid, and sugar of milk. There is also present fatty material which resembles the fat of colchicum seed, but may be, in part, added fatty acid. The percentage of colchicin found (0.50) is about that of a good quality of colchicum seed, the U. S. Pharmacopeial standard being not less than 0.45 per cent. Since the pills contain material other than colchicum seed, this assay would indicate a colchicum seed of high alkaloidal content, or the possible reinforcement of the pills with colchicum extract or colchicin.
Liquid Iodex.—This is sold by Menley & James, Ltd., the firm selling Iodex Ointment. According to a circular in a trade package “the valuable properties of Free Iodine are available in Liquid ‘Iodex’ in a state of greatly enhanced activity; but the irritating, corrosive and hardening drawbacks of ordinary solutions of the drug are absent.” The label on a bottle reads as follows: “Liquid ‘Iodex’ (Liq. Iodi. M. & J.). A nonirritant preparation of iodine (21⁄2%) ... This product contains Free Iodine....”
Bell-ans is now advertised directly to the public—but it is no less valuable on that account. True, it is a “patent medicine” in the commonly accepted sense of the term, but it is no more a “patent medicine” today than it was fifteen years ago when it reached the public, not through the direct medium of the newspapers but the more indirect route of the medical journals and undiscriminating physicians. It is true that, in view of the serious nature of many conditions which are loosely spoken of by the public as “indigestion,” its present method of exploitation is likely to make it just that much more dangerous because of the larger publicity that will be given. The point to be borne in mind is that Bell-ans is now in fact what it has always been in essence, a “patent medicine.”
We are not aware of a single publication in which a careful, detailed clinical study of Anasarcin has been reported. The claims made for Anasarcin, past and present, indicate either a deliberate purpose to mislead or crass ignorance of the rudiments of pharmacology and therapeutics. The exploiters of the nostrum claim that thousands of physicians have found Anasarcin tablets of unsurpassed remedial value in the treatment of disorders of the circulatory system and of ascitic conditions.244 It must be admitted that too many physicians have prescribed Anasarcin, otherwise the manufacturers would not have continued to spend thousands of dollars in advertising it in medical journals during a period of more than ten years.
The same form of iron is available in the compound iron mixture, formerly official, which Hunter says is exceedingly good. In this country the compound solution of iron and ammonium acetate, Basham’s mixture, so called, has long enjoyed a wide reputation as causing very little disturbance of the stomach, and the homely tincture of ferric chlorid is probably useful in a large majority of cases in which the stomach is not especially irritable.
In other words, the literature does not afford a report of a single piece of careful painstaking work the results of which lend support to the claims made for Cactina Pillets as stated above, for it is obvious that if Cactus grandiflorus contains no active principle, no active principle can be extracted from it. Some time after the report of the Council was published, Hatcher and Bailey secured genuine Cactus grandiflorus directly from a competent botanist, Dr. C. A. Purpus, of Vera Cruz, Mexico, and studied it experimentally. They reported (J. A. M. A. 56:26 [Jan. 7] 1911) in part as follows:
The claim made by many proprietary medicine manufacturers that they are “strictly ethical” because they advertise only to physicians is mere verbal camouflage. There may be no more certain way of insuring the continued use of a nostrum by the public than to have it prescribed by physicians; and none know this better than the makers of nostrums. A proprietary individuality is obtained by giving some special form to the tablets and package or a special coloring to the capsules (“Specify ‘Phenalgin Pink Top Capsules’ ”) so as to indicate the identity of the products in such a way that the patient may in the future procure them without the advice or warning of the physician. When a proprietary preparation with the name or initials stamped on it or attached to it is prescribed, the patient immediately is aware of the fact, and his respect for the physician’s intelligence and wisdom is naturally lessened.
The case seems to stand about like this: A nostrum maker spends thousands of dollars to tell physicians that his cloudy preparation is not like other preparations, and physicians are expected to accept that as convincing evidence that they should prescribe and their patients, perforce, take it. This too, in spite of the evidence gained by careful scientific investigators that the hypophosphites in fairly large doses contain less available phosphorus than half a glass of milk, and that there is no evidence available that they exert any therapeutic effects at all.
Perhaps you may have seen temporary relief follow the administration of Neurosine in chorea, and may argue that theorizing is of little value in the face of personal experience. We shall not deny that some may have had that experience, for Osborne calls attention to the fact that the success of any medicinal treatment must be judged in the light of the fact that chorea is self-limited, and the intensity of the symptoms will abate in from two to four weeks. In view of this, we would hardly dispute the claim that one may administer narcotics, such as those contained in Neurosine, and the symptoms of chorea may abate in spite of such mistreatment. In all the years that Neurosine has been exploited to physicians with such remarkable claims, we have never seen a report of a careful clinical study in which the product has been used under the conditions which scientific investigation demands. Would you prescribe any nonproprietary preparations which had never been studied clinically, if a horse-shoer or grocer’s boy told you it would cure epilepsy or malaria?
To the Editor:—As an old Fellow of the A. M. A. I beg to present the following facts to you, and to ask if anything can be done by you to expose the methods of these people: A concern calling itself “The Anasarcin Chem. Co.” of Winchester, Tenn., has caused to be sent to physicians a chart on the subject of “Diagnostics of Renal Diseases.” This chart contains eighteen plates, which were all taken without knowledge or permission of either myself or my publishers, William Wood & Co., from the third edition of my book on “Urinary Analysis and Diagnosis.” The plates are partly composite plates, but mostly portions of plates, exactly reproduced from my book. I at once caused my publishers to write to the Anasarcin Company; and a few days ago I received a letter from a Dr. H. Elliott Bates of 118 East Twenty-Eighth Street, New York, whose letterhead says, “Medical Advertising.” In this letter the writer says that it was he who suggested the sending of such a chart, and admits that all the plates were taken from my book. In this letter he offers to have a letter sent to every physician of the country “in which it is explicitly stated that the cuts on the chart were taken from your book, and that complete information regarding the matters treated on the chart can be found in your book.” In other words he offers to advertise my book free of cost to me, so that I should take no further steps in the matter. I consider this entire matter an outrage, and thought it best to write to you for advice, since my publishers seem to think that in spite of the violation of the copyright nothing can be done.
Antimeristem-Schmidt was rather widely exploited some six or seven years ago. As was explained in The Journal, March 8, 1913, p. 766, it is a preparation claimed to be useful in the treatment of inoperable cancer and as a supplementary treatment after operations for cancer. The treatment is founded on a theory advanced by one O. Schmidt that the cause of cancer is found in a fungus, Mucor racemosus, which, Schmidt at first asserted, carried a protozoon which he regarded as the real cause of the disease. The vaccine is said to be prepared from cultures from this fungus. While Schmidt claims that he has been able to produce cancer by means of the organism, scientific research has not verified his claims. Extensive clinical trials have shown the treatment to be without effect. The Journal also advised its readers on April 19, 1913, that no license for the sale of Antimeristem-Schmidt had been granted by the Treasury Department and, therefore, its importation into this country was prohibited. Neither the therapeutic nor the legal status of the product has been changed since then.—(From The Journal A.M.A., Dec. 6, 1919.)
In this connection it is worth noting that the American Newspaper Publishers’ Association, in a special bulletin issued in 1909, published a very complete list of press agents and the interests these agents represented. This list contains the name D. E. Woolley, who then was sending out press notices for the National Association of Piano Dealers of America. Is this the gentleman who is now acting as press agent for the Basic Chemical Corporation of America? If it is, it may be that the slump in the piano trade has caused Mr. Woolley to turn from musical instruments to cancer cures.—(From The Journal A. M. A., Sept. 3, 1921.)
It will be remembered that the Federal Trade Commission adopted the names arsphenamin and neoarsphenamin for the drugs first introduced as “salvarsan” and “neosalvarsan,” respectively; the terms barbital and barbital sodium for the substances first introduced as “veronal” and “veronal sodium,” and the word procain as the name for the compound first marketed as “novocain.” In issuing licenses for the use of the patents on these drugs, the commission stipulated that the drugs should be sold under the new American title unless the firm desired to use a new trade designation, in which case the titles chosen by the commission should be given equal prominence. The Council on Pharmacy and Chemistry has cooperated with the Federal Trade Commission and has adopted the new names as the descriptive names which appear in New and Nonofficial Remedies. The Chemical Foundation, Inc., which has purchased some 4,500 German-owned patents, many of them for synthetic drugs, proposes to continue the wise policy of the Federal Trade Commission by requiring that those who receive licenses for the use of patents for synthetic drugs must use a common designation for each drug selected by the foundation. “Cinchophen” has been selected as the designation for the substance introduced as “atophan” (also described in the U. S. Pharmacopeia under “phenylcinchoninic acid”). In consideration of this action on the part of the Chemical Foundation, and also because physicians found it difficult to use the pharmacopeia name “phenylcinchoninic acid,” the Council on Pharmacy and Chemistry has recognized the contracted term “cinchophen” as a name for the drug introduced as “atophan.” It is hoped that physicians will support this simplified and nonproprietary nomenclature in the same spirit with which they adopted the terms “arsphenamin,” “barbital” and “procain.”—(Editorial from The Journal A.M.A., Aug. 9, 1919.)
In the United States the medical profession has created a means whereby physicians need not be misled by such “high” authorities as evidently has been the case with our English confrères. Once more the value of the Council on Pharmacy and Chemistry is strikingly manifested. What are the facts about “Collosols”? The Council has reported that a number of the “Collosol” preparations were not colloids at all, and “if ... injected intravenously as directed, death might result, making the physician morally if not legally liable”;249 that in the cases in which the therapeutic claims were examined, the claims were found to be either exceedingly improbable or exaggerated; furthermore, that the A. M. A. Chemical Laboratory found “Collosol Cocaine,” on analysis, to contain only 40 per cent. of the claimed amount of cocain.250
The Du Pont Ether and the claims made for it are seemingly based on the work of one man, “James H. Cotton, M.A., M.D., Toronto, Canada,” who published an article on “Cotton Process Ether and Ether Analgesia,” in the American Journal of Surgery for April, 1919. However, Cotton did not give the composition of the “new” ether nor, so far as we are aware, has his work been corroborated. In view of the inquiries received, the Secretary of the Council on Pharmacy and Chemistry asked the Du Pont Chemical Works for the composition of the new ether. From the firm’s reply we quote one paragraph:
A “Special Note” in the “Confidential Guide” advises physicians who “have to deal with Hysteria” to “write the Author of this Guide, who will explain by personal letter a method of cooperation by which such Convulsions may be At Once and forever stopped.... There will be $100 for You from every case treated.” One physician wrote to the “Author of this Guide”—Eli H. Dunn, M.D.—asking for further information on this treatment for hysteria. He received in reply two letters both signed Eli H. Dunn; one was to be shown to the patient, the other was for the doctor’s own information. The letter for the patient to see described the marvelous effects of “Dunn’s Intravenous and Restorative Treatment” in hysteria and recommended it “with the utmost confidence in every case able to pay you the fee commensurate with the service you render.” Then followed these two paragraphs:
There has now come to hand a report just published by a special committee appointed by the council of the Academy of Medicine, Toronto, to investigate the Glover Serum. The report of this committee may be summed up by one of its closing paragraphs, which reads:
At present the public is much exercised over the epidemic of infantile paralysis. Anticipating that the nostrum fraternity would attempt to reap a golden harvest from the public distress, the federal officials issued a bulletin of warning on the subject. Naturally, the bulletin was addressed to the lay public, the government assuming that physicians knew enough to avoid being misled by any such advertising campaigns. Apparently, the assumption is too broad. At any rate, the manufacturers of “Glyco-Thymoline” are circularizing physicians, one of whom writes as follows:
The law which limits the length of time that food products may be kept in cold storage could with advantage have its scope extended to include “patent medicine” testimonials. Physicians recently received through the mails—at a time when the mails were frightfully congested with Christmas business—a sixteen page pamphlet sent out in a plain envelop as first class matter. The caption of the pamphlet reads: “Cough and Its Treatment in Pulmonary and Laryngeal Tuberculosis: By Henry Levien, M.D., While Medical Director and Physician-in-Charge of the Liberty Sanitarium, Liberty, N. Y. From the Buffalo Medical Journal.” The pamphlet is devoted to the alleged virtues of that dangerous and widely advertised nostrum, “Glyco-Heroin (Smith),” whose more recent and less descriptive name is now “Glykeron.” Physicians might assume, and doubtless will assume, from the pamphlet that this reprint represents a recent pronouncement on the subject with which it deals. The facts are that the “Liberty Sanitarium” has, apparently, been out of existence for at least fifteen years, while the article itself originally appeared more than eighteen years ago—September, 1901. One of many physicians who sent in the copies received called attention to the fact that he had left the address to which the pamphlet was directed, more than six years ago. Even at that, the mailing lists of the concern that sells this heroin-containing nostrum are more than twelve years ahead of its “clinical reports.”—(Editorial from The Journal A. M. A., Jan. 17, 1920.)
“At first sight one might question whether a cod liver oil product which contains absolutely no cod liver oil had not suffered the loss of essential elements. But a closer reading discloses a significant qualification, namely, the phrase, ‘those elements which make the crude oil such a high-class reconstructive.’
Intravenous Compound (Loffler) stands revealed as a nostrum of secret composition which physicians are asked to inject into the veins of their patients. It must be purchased in connection with some supplementary material, “a complete set of apparatus,” sold by the same concern. Its successful administration is said to depend on following a technic detailed either in a booklet sent out by Loffler or given by Loffler in a “Post-graduate Course” which costs physicians $50 unless they have purchased six dollars’ worth of another nostrum, “Thymozene.”
“ ‘Luvein’ Arsans (Plain)” is said to be: “Di hypo sodio calcio phosphite hydroxy arseno mercuric iodid.” The first part of this “formula” might stand for sodium and calcium hypophosphite. The remainder is meaningless except that it suggests (but does not insure) the presence of arsenic and mercury iodide.
“Pneumonia Phylacogen has been found to be a dependable means of preventing and treating pneumonic complications of Influenza. In one large city it became a routine measure to give all persons affected with Influenza an injection of Pneumonia Phylacogen as a prophylactic of pneumonia. The results were remarkable. Not only did the cases improve rapidly, but in a great majority of them the pneumonia did not occur.”
This form of advertising is done with intent to deceive and did deceive the doctor in Oklahoma. It was therefore a successful falsehood, its success depending on the false use of the name of the President of the American Medical Association to bolster up the sale of the product.
In May, 1917, physicians in the West received a letter from the “Ellison-White Chautauqua System” informing them that Dr. Henry Smith Williams was to lecture at “your Chautauqua” and reminding them that “he has recently issued two volumes, ‘The Autolysin Treatment of Cancer’ which he believes will be his greatest contribution to medical science.” The present “Proteal” treatment appears to be a modification of the “Autolysin” treatment. Dr. Williams, in attempting to justify the use of his “Proteal” in tuberculosis, cancer, rheumatism, etc., takes advantage of certain investigations bearing on the nonspecific reactions resulting from the parental injection of foreign proteins. So far as we can discover, there is no scientific evidence to indicate that the “Proteal” treatment expounded by Williams is of value in the treatment of cancer, tuberculosis or the other numerous diseases for which the “Proteals” are recommended.
The first two statements quoted above are obviously false. The third statement might have been true although it seemed unlikely. A letter was, therefore, written to the Department of Public Health of the Commonwealth of Massachusetts and the claim of the K. A. Hughes Company relative to the adoption of Salicon in all the state camps by the “state medical authorities” was brought to their attention. The reply of the department on this point was emphatic:
In China the administration of powdered tiger-bone is—or was—a favorite form of treatment in cases of supposed cardiac weakness. The theory is, presumably, that the cardiac strength of the tiger would be a good thing for the patient to acquire. Since many patients have recovered after taking tiger-bone, and no one has proved that they might not have died had they failed to take it, “clinical experience” stands back of the treatment; and where is the skeptic so rash as to challenge that? The Chinese physician believes in his tiger-bone therapy, and, with the best interests of his patient at heart, insists on obtaining absolutely true and authentic tiger-bone. Not satisfied with the assertions of the dealers, the conscientious Chinese physician subjects his tiger-bone to a kind of physiologic standardization. He offers the bone in question to a dog! If it is an ox-bone—a frequent form of substitution—the dog will seize and eagerly gnaw it, whereas, according to all the teachings of Chinese pharmacognosy, if it is a tiger-bone the dog will depart hurriedly with his tail between his legs. Very foolish? Yes! But before we smile superciliously at the Chinese medical man, let us turn to the report of the Council on Pharmacy and Chemistry on “So-Called Secretin Preparations.”257 After reading this report let us put to ourselves, squarely and honestly, the question: Has the attitude toward secretin therapy, of a certain portion of those who represent Western modern medicine, really been much more scientific than that of the Chinese medical profession toward tiger-bone therapy? On the basis of a hypothesis scarcely less crude and unsubstantiated than that which assumes that tiger-bone is of value in heart disease, it has been assumed that secretin is of value in gastro-intestinal diseases. On the ground of “clinical evidence” scarcely more critical than that exhibited by our confrères in the antipodes, it has been asserted that alleged secretin preparations actually are efficious. Indeed, in one respect the methods of the Chinese physician appear more scientific than those of his Western brethren. To the best of his ability, the Oriental at least makes sure that he is administering genuine tiger-bone; he does not rely on the unverified word of his dealer alone. The American physician has not been making the least effort to ascertain whether his supposed secretin preparations are truly such; and, as a matter of fact, scientific investigation seems to indicate that some of these products contained no secretin at all! Whatever one may think of the validity of his test, the Chinese physician does his best according to his lights. As to “clinical experience,” Dr. Jacobi has well said that some people make the same mistake a hundred times and call it “experience.”—(Editorial from The Journal A. M. A., Jan. 15, 1916.)
With the best brains of the world at work on the problem of cancer, it is reasonable to assume that any man who has found out even a little more than has previously been discovered or is able to accomplish even a little better results than the average in the treatment of this dreaded disease, would be well known to scientific medicine.
The “memorandum” is a four-page leaflet of which three pages are devoted to “Tyree’s Antiseptic Powder.” One of these three pages is a reproduction of a letter on the stationery of the Surgeon General’s Office of the War Department, and signed “W. M. Gray, M.D., Microscopist, Army Medical Museum; Pathologist to Providence Hospital.” The letter describes a series of “bacteriological and comparative tests” made by Dr. Gray with Tyree’s Antiseptic Powder. The entire second page of the circular is given over to the results of these bacteriologic tests which compare various strengths of Tyree’s Antiseptic Powder with “mercuric bichlorid,” phenol and formaldehyde.
Mr. Sinclair says that he has spent time in Dr. Abrams’ clinic and is wonderfully impressed with Dr. Abrams’ achievements. So is the small boy impressed with the marvelous facility with which the magician extracts the white rabbit from the silk hat. Mr. Sinclair is convinced “that Albert Abrams has discovered the great secret of the diagnosis and cure of all the major diseases.” The small boy is equally convinced that the prestidigitator has solved the mystery of producing snow white bunnies from airy nothings.
After the Sterling Products Company had acquired the pharmaceutical end of the business, the Winthrop Chemical Co. was incorporated in the state of New York. This concern seemingly secured control of all the Bayer pharmaceutical specialties except “Aspirin.” The Bayer Co., it was announced, had been merged with the Sterling Products Co., and “Aspirin-Bayer” added to the latter firm’s list of “patent medicines”: Cascarets, Danderine, Pape’s Diapepsin, California Syrup of Figs, Neuralgine and Dodson’s Livertone. The business is apparently a paying one financially as witness the following excerpt from a recent announcement in a drug journal:
George Starr White, M.D., F.S.Sc., Lond., Los Angeles, Calif.—A letter received by a physician a few days before the recent convention of the Allied Medical Associations, held out as an inducement to be present the fact that “Geo. S. White will show you how to diagnose disease by means of dif. colored lights and the reaction of the body to the magnetic meridian.” Dr. George Starr White was the “Second Vice-President” of the Allied Medical Associations in 1918. White, according to our records, was graduated in 1908 when he was forty-two years old, by the New York Homeopathic Medical College and Hospital. He was licensed in New York in 1908, in California, Connecticut and Nevada in 1913, and in Michigan in 1916. He seems to have been one of the proponents of “spondylotherapy,” “zonetherapy,” etc., and in 1915 it was announced that he would give one week courses in “Spondylotherapy” in Chicago, Kansas City and Denver, respectively. In his advertisement he emphasized that he was a Fellow of the American Medical Association, which, while true at the time, is no longer true, as on Feb. 4, 1916, he was expelled from membership in the Los Angeles County Medical Association. In May, 1915, White was arrested in Chicago and fined $100 and costs for practicing medicine without a license. Dr. White’s specialty seems to be what is ponderously known as “Bio-Dynamo-Chromatic Diagnosis.” This has been described by one of its enthusiastic adherents as “Diagnosis by Sympathetic Vagal-Reflex.” To obtain the “Sympathetic Vagal-Reflex” it seems the patient must face east or west and have his bare abdomen percussed until a dull area is located. The patient is then faced north or south and again percussed. Then, it seems, different colored lights are thrown on the patient, the location of the areas of dullness being determined meanwhile. A combination of ruby and blue lights “will cause a reflex in cases of gonorrhea,” a “green light will cause a reflex in cases of liver or gallbladder trouble,” while the color for carcinoma is orange red! During the height of the influenza epidemic last winter, White seems to have put on the market “Valens Essential Oil Tablets” which were for “Gripping the Flu out of Influenza,” and were also said greatly to benefit or cure incipient tuberculosis, hay-fever, asthma, and “catar.” The letters “F.S.Sc., Lond.” after Dr. White’s name look well, sound well, and have an air of erudition and mystery that is well worth what they cost. They mean “Fellow of the Incorporated Society of Science, Letters and Arts of London, Ltd.” The “Fellowship” costs one guinea. Not a few “patent medicine” exploiters in the United States carry these mystic letters after their names. The society in question was a seriocomic concern that was exposed by London Truth some years ago and was also dealt with in The Journal of May 29, 1909, in connection with the “Aicsol Consumption Cure” exposé.
The Adamson Bill, known as the “trading with the enemy act,” has recently been passed by the House of Representatives, is now before the Senate, and will doubtless be enacted into a law. One of its clauses confers authority on the Federal Trade Commission to grant licenses to citizens of this country to operate patents owned by enemy aliens. Physicians are interested in the bill primarily because it includes the salvarsan situation. The manner in which salvarsan has been supplied in this country has been so arbitrary and the prices charged so tremendously above the actual cost, that we should not be satisfied unless the monopoly is ended so that the drug can be supplied at least at a fairly moderate figure, and the old methods eliminated. It is to be hoped, therefore, that the Federal Trade Commission will not grant exclusive control—that is, exclusive license—to any one person or firm. To do so would simply perpetuate the old monopoly and the old conditions. England has adopted a law, which, in principle, is similar to the Adamson Bill, and there several concerns have been licensed to manufacture the product. The same should be done here. The Dermatologic Research Laboratories of Philadelphia announce that they can supply arsenobenzol at $1.50 a tube, and that there is immediately available a supply sufficient for any demand that may be made. The same laboratories have announced also that in a few months they will be able to supply hospitals for $1.00 a tube. Considerable responsibility rests on the Federal Trade Commission in this matter, for it is not only a question of monopoly, but also a question of scientific qualifications and ability to make the product on the part of some who may make application. Undoubtedly the commission will secure the cooperation of the United States Public Health Service, under whose supervision these drugs should be manufactured no matter who shall be licensed to make the product.—(Editorial from The Journal A. M. A., July 21, 1917.)
The danger of commercialized therapeutics has been enormously increased by the introduction of biologic products. These substances offer a rich field for the commercially minded, first, because of the remarkable results which seem to have followed the use of certain products of this type; second, because the field is new and the mode of action of these substances not readily understood and, third—and most important—because, by the very nature of the problems involved, few physicians are well informed concerning them. The influenza epidemic of last year was widespread and fatal in character. It stimulated earnest research in methods of prevention and cure. We were all in a frame of mind to grasp at any straw. Here and there some worker would cry “Eureka”—only to be disappointed when his product was actually put to the test. However, there were more than enough manufacturers ready to place any product on the market with specious claims that could not be positively denied. Vaccines, serums, proteins—all were advanced with such glowing statements as to their properties that only those physicians who kept their feet firmly on solid ground could resist the appeal. Now we have had another epidemic—mild, it is true—but the memories of last year make the average physician ready to accept anything which promises hope, and the manufacturers “make hay while the sun shines.” Physicians have been and are being deluged with literature on the prophylaxis and treatment of influenza. So far as we know, few publications have contained any word of warning on these matters. One exception has just come to notice: the Medico-Military Review, a semimonthly mimeographed publication sent to medical officers of the Army by the Surgeon General’s Office. This says:
Not only must there be unquestionable evidence, however, of the diminution in size of the tumor, but this diminution must be of a kind not ordinarily attributable to the natural evolution of the tumor.... It is safe to say that multiple tumors offer enormous difficulties in the matter of interpreting therapeutic results. At present we have in the wards of the hospital a patient with multiple metastatic carcinomas of the skin. For several months we have at intervals made accurate measurements of certain of these tumors and have found that some have undergone retrogression, others have entirely disappeared, while still others have continued to grow steadily. In the case which afforded the ascitic fluid used in Hodenpyl’s experiments, many of the lymphatic metastases underwent complete retrogression, while the metastatic process in the liver, as was demonstrated at necropsy, increased progressively, and ultimately almost destroyed that organ. Thus, in multiple carcinosis, the retrogression of individual nodules is no indication that therapeutic intervention has produced an improvement.
These are some of the reasons why physicians oppose patent medicines as they are now exploited; and for these reasons, physicians should take an absolutely uncompromising attitude, and use every opportunity to educate the public. The patent medicine interests naturally try to obscure the issue. By the art in which they are so skilful, they aim to suggest to the public that physicians are opposed to patent medicines, in order to drive patients to their offices. They “forget” to mention that physicians have never conducted a “campaign” against really efficient preventive public-health measures, no matter how many prospective patients were involved. No physician has ever refused to give diphtheria antitoxin because this would diminish the number of his visits. A short memory is a very convenient asset for self-interested persons. It is not so convenient for the public—but it is all too frequent. Physicians must, therefore, make it plain that their stand is not against patent medicines, but for the protection of the health of the public.—(From The Journal A. M. A., March 4, 1916.)
As to serum treatment, the only noteworthy new method so far is the injection in severe cases of influenzal pneumonia of the serum of patients who have recovered from such pneumonia.292 The principle of this method is rational; analogous procedures have given seemingly good results in scarlet fever and other diseases; and the results reported in influenzal pneumonia appear promising. Further trial of this treatment under proper conditions consequently seems to be warranted. It should be borne in mind, however, that McGuire and Redden292 made their observations in the declining phase of the epidemic when the organism or organisms concerned appeared to be losing virulence. For this and other reasons, the expectations as to what may be accomplished by this method must be kept within reasonable bounds. Influenza is a self-limited disease with variable complications and of variable severity in different places, thus offering great difficulties in the way of evaluation of different methods of treatment.
These factors apply particularly in the case of substances like iodin, arsenic, mercury or the biologic products in which the mode of administration radically modifies the action. For some time, manufacturers have urged substitutes for tincture of iodin, claiming that their substitutes were free from the undesirable properties of the tincture, and, at the same time, possessed special virtues which the tincture could not possess. More recently, attention has been directed to the administration of iodin in the form of vapor. The diffusing and penetrating powers of gases have particularly attracted the attention of therapeutists, since by this method drugs may be applied to rather inaccessible portions of the body, such as the lining of the lungs, the throat and the mucous membranes of the genito-urinary tract. Furthermore, it has been asserted that iodin in the form of fumes has increased combining powers, and is thus far more potent in effect than iodin administered by any other route. There do not seem to have been any adequate scientific investigations of the subject, however, until the recently published results of Luckhardt and his collaborators293 at the University of Chicago. In their experiments, both on man and on animals, accurately determined quantities of iodin were vaporized in a special device, and the fumes applied to the skin. At the same time, the tincture was applied to the skin of other persons as a control. Iodin was also applied to the skin of dogs with hyperplastic thyroid glands; and the effects on the gland, before and after administration, studied. Dogs were also used to determine whether iodin fumes were absorbed from the lungs. As a result of these investigations, which are reported in great detail, it was found that iodin, when deposited on the skin in the form of fumes, is absorbed. More iodin was recovered from the urine, following the application of the tincture, than was recovered following the use of the fumes. This result is explained by the authors on the ground that probably more iodin was actually applied, and that the iodin so deposited was held in combination with the protein during the process of coagulation of the latter by the alcohol of the tincture, leading to a state of continuous absorption. It is probable, furthermore, that the iodin deposited on the skin in the form of fumes is revaporized to some extent by the heat of the body.
About four years ago when the “Academy” seemed to be making a particularly heavy bid for American dollars the member of The Journal staff in charge of the Propaganda Department wrote to the “Academy,” on his personal stationery, asking about the cost of membership in the “Academy” and asking also for a copy of the “prospectus.” And that was all. In reply he received a letter stating that “in consideration of” his “many dignities and great learning” he had been nominated “an officer of this academy” and had been awarded “la médaille de première classe” for humanitarian work and scientific merit. In order to obtain these tokens of the “Academy’s” regard it would be necessary to inform the “Academy” of acceptance “in conformity with Section 19 and 22 ...” As the Propaganda Department did not consider the diploma and gilt medal worth $15 even as exhibit for its museum of fakes, the “Form of Acceptance” was not filled in and returned “in accordance with Section 19 and 22.”
The use of liquid petrolatum in chronic constipation, which has recently become the vogue, has naturally been commercialized; as a result, also naturally, claims of superiority of one brand over another have been made. Some of these claims may have been well founded; others certainly are not. Some have claimed superiority for those products made from Russian oil over those made from American oils. As naphthene hydrocarbons predominate in Russian crude petroleums, and paraffin hydrocarbons in many or most American crude petroleums, it was assumed that the petrolatums derived from these sources differed from one another in like manner. Both the naphthenes and the paraffins are chemically inert; but some unexplained therapeutic superiority has been assumed to reside in the naphthenes. Consequently, it has been urged that the American liquid petrolatums should not be used internally. So far these claims and counterclaims have been based on much theory and little fact. The Journal publishes this week a contribution by Benjamin T. Brooks, Senior Fellow in charge of petroleum investigations at Mellon Institute, Pittsburgh. Brooks calls attention to the fact that Marcusson, in 1913, pointed out that most of the so-called “mineral oils” used for therapeutic purposes contain no paraffin hydrocarbons whatever; that they consist solely of naphthenes and polynaphthenes. Brooks confirms this statement so far as American liquid petrolatums are concerned. He states that many American petroleums, such as most of those from the Gulf region, are like the Russian in containing no paraffin; and that, in the case of those petroleums that do contain it, the customary refinery method of removing paraffin is sufficient to produce true naphthene and polynaphthene petrolatums. “The claim that only Russian oils belong in this class,” he says, “has no basis in fact and has been advanced presumably for business reasons.” The name “paraffin oil” applied to these liquid petrolatums, then, is a misnomer. The new name, “white naphthene oils,” suggested by Brooks, seems superfluous, however, since the pharmacopeial title, “liquid petrolatum,” is subject to no such objection.—(Editorial from The Journal A. M. A., Jan. 1, 1916.)
Part I of the volume contains formulas, good, bad and indifferent, including the equivalents of a large number of shotgun proprietaries. Part II contains descriptions of drugs. This is a new feature. The purpose is to provide standards for those drugs not described in the Pharmacopeia but used in N. F. preparations. Many of these drugs were described in the U. S. Pharmacopeia VIII, but have not been included in the ninth revision. Practically all are either worthless or superfluous. Part III contains descriptions of special tests and reagents.
The booklet further states that “THE OGDEN METHOD has entirely eliminated the use of cautery, the ligature or any injections, in the treatment of hemorrhoids,” but that “the use of the electric current has proved to be the very correct method in such cases, as will be demonstrated at the clinic.” The booklet reiterates the statement that Ogden’s association with the Burleson and Burleson concern at Grand Rapids makes him “eminently well qualified to instruct members of the medical profession in this important branch of the medical science!”
In the past, therapeutic agents and apparatus have been controlled by patents and trademarks for profit. If there have been exceptions, they have been rare. The Principles of Medical Ethics of the American Medical Association contain this statement: “It is unprofessional to receive remuneration from patents for surgical instruments or medicines.” This does not mean that the patenting is wrong in itself; there are occasions when it is wise, if not necessary, to obtain a patent in the interest of the public, and, in the case of surgical instruments and medicines, of the medical profession. In certain instances it is absolutely necessary that the article produced shall maintain a definite standard of quality and purity—and, it may be added, shall be sold at a reasonable price. Enterprising pharmaceutical manufacturers have usually been ready to appropriate the results of scientific research by investigators or therapeutic measures suggested by practicing physicians. Not infrequently, in such instances, the desire for financial gain has caused the marketing of such products with extravagant, if not false, claims as to their value. Yet the patent laws may be used so as to protect and benefit the public and the medical profession. In research laboratories, work is being carried on resulting in the production of new therapeutic agents. It is important that these agents shall be so controlled that they may be made available without subordination to commercial interests. It has become practically necessary, therefore, for research workers to protect their products in the interest of the public welfare and scientific medicine. It has not been an easy matter to decide how best to bring about the desired results. This question has been before the Board of Trustees of the American Medical Association; and, in 1914, the House of Delegates passed a resolution authorizing the board to accept at its discretion patents for medical and surgical instruments and appliances; as trustees, for the benefit of the profession and the public, provided that neither the Association nor the patentee should receive remuneration from these patents. The Rockefeller Institute for Medical Research has solved the problem in a similar manner. In connection with the report of the discovery of several new arsenic compounds, Jacobs and Heidelberger,302 working in the Rockefeller Institute, say:
Does the public love to be humbugged? We doubt it. That we, whether sage or fool, are humbugged is undeniable. We are humbugged just to the extent that we are ignorant. There lies one of the most powerful factors operating to the advantage of the “patent medicine” maker and the quack. The layman’s ignorance of the possibilities and limitations of drugs is wide and deep. Hence the ease with which he is fooled on this subject. A seeming frankness in advertising being the order of the day, the nostrum maker makes a pretense of telling what is in his stuff without disclosing any facts that will tend to lift the veil of mystery and thus destroy his greatest asset. So the exploiter of nostrums to the medical profession, realizing that at least a pretense must be made of giving the composition of medicaments offered to the physician, declares that his clay poultice has for its base “anhydrous and levigated argillaceous mineral.” This sounds much more imposing than dry and finely powdered clay, and satisfies by its very sonorousness. Now comes a product exploited chiefly to members of the dental profession but also, it seems, to physicians. Tablets, “activated tablets,” if you please! They are “an anodyne, analgesic febrifuge sedative, exorcising [sic!] antineuralgic and antirheumatic action.” And their composition? Simply “an activated, balanced combination of the mono-acetyl-derivative of para-amidophenetol together with a feebly basic substance in the alkaloidal state from the Thea-Sinensis.” As clear as the Missouri River! Some day some dentist or physician is going to investigate and find that this awe-inspiring, polysyllabic example of exuberant verbosity means nothing more mysterious than our old friends acetphenetidin (phenacetin) and caffein. In the meantime, the exploiters may smile softly and murmur, “Barnum was right!”—(Editorial from The Journal A. M. A., Jan. 1, 1921.)
The efforts of the medical members of the committee, however, have not been entirely fruitless. Of the articles described in the U. S. Pharmacopeia VIII, 243 have been deleted; sixty-seven new articles have been added. The loss of 167 titles may be set down as a gain. Moreover, most of the new substances give promise of therapeutic usefulness. Thirty-six are taken over from New and Nonofficial Remedies; nineteen are substances which are in the edition of Useful Drugs now in the press. It cannot be said, however; that all of the additions have been judiciously selected. It is an infelicitous time to add calcium and sodium glycerophosphate just when grave doubts of their therapeutic efficiency are being felt. The addition of the extracts of aconite, hydrastis and viburnum prunifolium is likewise unfortunate. All are superfluous preparations, the first because a drug so powerful that an average dose of the extract is only 10 mg. or 1⁄6 grain is better given in the form of tincture; the second because hydrastis is a drug of uncertain value, already represented by three preparations, and the third because viburnum prunifolium has been discarded and discredited by the best therapeutic authorities. It must be accounted clear gain, on the other hand, that the deletions include many inert, obsolete or superfluous substances like bismuth citrate, kaolin cataplasm, pipsissewa, coca leaves, ladyslipper, wahoo, cotton root bark, compound acetanilid powder and compound syrup of hypophosphites, not to mention nine salts of iron and thirty-eight fluidextracts of various drugs. Wines, unmedicated and medicated, whisky and brandy are also among the articles dropped.
The company to which our correspondent refers put out a proprietary product prescribed by physicians and used by the public. Some years ago the company in question advertised its product in The Journal until its stock-selling scheme was brought to the attention of The Journal; the advertisements were then rejected. Some years later, on evidence that the company had discontinued its stock-selling methods to physicians, its product was again admitted to the advertising pages of The Journal. Our correspondent says that he believes that the physicians who hold stock in this company “are entitled to a square deal.” What about the public? Is it getting a square deal when physicians are financially interested in the products that they may be called on to prescribe? Is the average layman’s confidence in the medical profession likely to be enhanced when he learns that the physician to whom he went for treatment has a financial interest in the therapeutic agent which was prescribed? Our correspondent’s complaint against the company seems to be, not that the company sold stock to physicians, but that “the dividend checks have been few and far between,” the assumption being that had the dividends come regularly, there would have been no complaint. It cannot be too often emphasized that it is against public interest and scientific medicine for physicians to be financially interested in the sale of products which they may be called on to prescribe for the sick. It is perfectly true that there are many physicians who would not consciously permit financial considerations to warp their judgment; but it is not humanly possible to remain unbiased in cases of this sort. It is conceivable that a judge on the bench might make every effort to dispense impartial justice in a suit in which one of the parties was a firm in which he, the judge, had financial interest. Nevertheless, it would be obviously improper for such a judge to try a case of this kind. Yet, in this supposititious case the only harm that could result would be of a financial nature. In the case of the physician, the harm is not to the public’s purse but to the public’s health.—(From The Journal A. M. A., Dec. 11, 1920.)
To the Editor:—Dr. William G. Ward’s letter (The Journal, Feb. 3, 1917, p. 390), and the recent admirable article by Dr. Harold N. Cole (The Journal, Dec. 30, 1916, p. 2012) recall to mind Dr. J. B. Murphy’s clinical note on the use of sodium cacodylate in the treatment of syphilis (The Journal, Sept. 24, 1910, p. 1113), and the experimental work of Cap. H. J. Nichols, U. S. Army (The Journal, Feb. 18, 1911, p. 492). The results of Nichols’ work conclusively proved, at least from a laboratory standpoint, that this drug was of very little value as a spirocheticide in combating syphilis. Prior to the publication of Dr. Murphy’s letter I had employed sodium cacodylate extensively as a remedy in psoriasis, and I still continue to use it in selected cases of the disease.
The public “demands” vitamins in pill form! Why? For the same reason that the public, lay or medical, demands many things today that it does not need—because the whole trend of modern advertising is toward creating demands, rather than supplying needs. Vitamin concentrates are being “demanded” by the public because shrewd and forward-looking “patent medicine” exploiters are using all the subtle arts of modern advertising to convince the public that it is in serious danger of vitamin starvation, and that the only hope lies in buying these alleged concentrates to make up a hypothetical deficiency. It seems inconceivable that a rational man would pay a tremendously high price for certain food factors which are already present in his ordinary diet. But he will; and advertising is the reason. Advertising campaigns such as these of the vitamins constitute a vicious circle; an artificial demand is created and then the manufacturer excuses his business on the ground that he is merely supplying a demand! As our British contemporary says, “ordinary fresh foods are the simplest, cheapest and richest sources of vitamins.”—(Editorial from The Journal A. M. A., March 18, 1922.)
In a similar bulletin issued August, 1914, there were recorded several more cases of adulteration and misbranding charged against the William A. Webster Company. Some “Wine Coca Leaves” was held adulterated in that the amount of alcohol present was wrongly declared on the label; it was held misbranded in that while it contained cocain, the label failed to bear any statement regarding the quantity of proportion of this drug. Tablets of “Acetanilid and Sodium Bromid Compound” were found deficient in strength. “Anti-Vomit Tablets,” “Aspirin Tablets,” “Bismuth and Calomel Tablets,” “Quinin Laxative Tablets,” “Salol Tablets,” “Sodium Salicylate Tablets,” “Neuralgic Tablets,” “Diarrhea Calomel Pills” and “Morphin Sulphate Hypodermic Tablets” were also misbranded in that the amount of certain ingredients found in them failed to tally with the amount declared on the label. In all of these cases also the William A. Webster Company pleaded guilty and was fined.
THE PROPAGANDA FOR REFORM IN PROPRIETARY MEDICINES
PART I
REPORTS OF THE COUNCIL ON PHARMACY AND CHEMISTRY
FOREWORD
THE COUNCIL ON PHARMACY AND CHEMISTRY
The Council on Pharmacy and Chemistry was established by the American Medical Association primarily for the purpose of gathering and disseminating such information as would protect the medical profession—and thus the public—in the prescribing of proprietary medicinal articles.
The Council consists of sixteen members, fifteen appointed for a term of five years without pay, and the sixteenth, a secretary, who is also the director of the Chemical Laboratory of the American Medical Association (see Part II).
At the present time (1921) the membership is:
C. L. Alsberg, A.M., M.D., Chief of the Bureau of Chemistry, U. S. Department of Agriculture, Washington, D. C.
C. W. Edmunds, M.D., Professor of Materia Medica and Therapeutics, University of Michigan Medical School, Ann Arbor.
R. A. Hatcher, Ph.G., M.D., Professor of Pharmacology, Cornell University Medical College, New York City.
A. W. Hewlett, M.D., Professor of Medicine, Leland Stanford Junior University School of Medicine, San Francisco.
John Howland, M.D., Professor of Pediatrics, Johns Hopkins University Medical Department, Baltimore.
Reid Hunt, M.D., Professor of Pharmacology, Medical School, Harvard University, Boston.
W. T. Longcope, A.B., M.D., New York.
G. W. McCoy, M.D., Director of the Hygienic Laboratory, U. S. Public Health Service, Washington, D. C.
Lafayette B. Mendel, Ph.D., Sc.D., Professor of Physiological Chemistry, Sheffield Scientific School, Yale University, New Haven.
F. G. Novy, Sc.D., M.D., Professor of Bacteriology, University of Michigan Medical School, Ann Arbor.
W. W. Palmer, B.S., M.D., Bard Professor of Medicine, Columbia University College of Physicians and Surgeons, New York.
W. A. Puckner, Phar.D., Secretary of the Council, Director of the Chemical Laboratory of the American Medical Association, Chicago.
L. G. Rowntree, M.D., Sc.D., Professor of Medicine, Mayo Foundation, Rochester.
G. H. Simmons, M.D., LL.D., Chairman of the Council, Editor of The Journal of the American Medical Association, Chicago.
Torald Sollmann, M.D., Professor of Pharmacology and Materia Medica, Western Reserve University School of Medicine, Cleveland.
Julius Stieglitz, Ph.D., Sc.D., Chem.D., Professor of Chemistry, University of Chicago, Vice-Chairman of the Council, Chicago.
At its first meeting in 1905, the Council began examining the proprietary and nonofficial medicinal preparations offered to physicians of the United States, and authorized the publication of a book (New and Nonofficial Remedies) containing descriptions of those preparations which were deemed worthy of the consideration of physicians. It also issued reports (Reports of the Council on Pharmacy and Chemistry) to the medical profession on those preparations which were not eligible. The Council adopted a set of rules by which to measure the eligibility of each preparation for admission to New and Nonofficial Remedies. These rules were designed primarily to protect the public—through the medical profession—against fraud, undesirable secrecy and objectionable advertising in connection with proprietary medicinal articles. The rules originally adopted have been subjected to revision from time to time to meet changing conditions. For the information of those who wish to familiarize themselves with the work of the Council the rules which are now in force (1921) follow this introduction. A summary is also to be found in the article, “The Work of the Council on Pharmacy and Chemistry, Present and Future,” page 12.
Since 1906, the Council has issued New and Nonofficial Remedies annually. In each issue are listed and described the articles that stand accepted on January 1 of the year of publication. The book describes proprietary medicinal articles on the American market that are found eligible under the rules, and also such nonproprietary, nonofficial articles as give promise of therapeutic usefulness, listing the acceptable brands. Articles of a similar character are grouped together, and each group is preceded by a general discussion for the purpose of comparison.
Since 1908, the Council has also issued an annual volume, “Reports of the Council on Pharmacy and Chemistry,” which contains reports on proprietary medicines that were found inadmissible to New and Nonofficial Remedies. The reports issued prior to 1916—and deemed of sufficient interest to physicians—were reprinted in the Propaganda for Reform in Proprietary Medicines, ninth edition (1916). The more important reports issued from 1916 to 1921, inclusive, are in this volume.
While it is the chief function of the Council to investigate and report on proprietary medicinal preparations, its work has broadened so that the Council’s work may now be characterized as a propaganda for the rational use of drugs. Thus, its Committee on Therapeutic Research encourages the investigation of questions concerning the actions of drugs. These investigations are brought together in the “Annual Reports of the Therapeutic Research Committee.” The Council also has a committee on medical teaching which has issued the publication “Useful Drugs,” a concise, but thorough and up-to-date, discussion of the more important drugs. In addition, the Council appointed a committee to prepare an “Epitome of the U. S. Pharmacopeia and National Formulary,” in which are presented those portions of the United States Pharmacopeia and the National Formulary that are of interest to physicians and in which is given a concise statement of the therapeutic usefulness of such drugs and preparations.
OFFICIAL RULES OF THE COUNCIL ON PHARMACY AND CHEMISTRY
[May 1, 1921]
Introduction
The following rules have been adopted by the Council primarily with the object of protecting the medical profession and the public against fraud, undesirable secrecy and objectionable advertising in connection with proprietary medicinal articles.
New and Nonofficial Remedies.—The book New and Nonofficial Remedies contains a description of proprietary articles which have been accepted as conforming to the rules of the Council; and of such simple nonproprietary and nonofficial substances as seem of sufficient importance to warrant their inclusion.
Mixtures.—For admission to N. N. R., proprietary pharmaceutical mixtures must comply with the rules; and, to determine such compliance, they will be investigated by the Council. The Council, however, endorses the principle that prescriptions should be written on the basis of the therapeutic effects of the individual ingredients. For this reason, it includes in this book only those mixtures that present some real advantage. There is also an appendix in which are included those proprietary articles which, so far as known to the Council, comply with the rules, but which do not possess sufficient originality to be admitted to the body of the book.
Rules Governing the Admission of Proprietary Articles to the Book New and Nonofficial Remedies
Definition of Proprietary Articles.—The term “proprietary article,” in this place, shall mean any chemical, drug or similar preparation used in the treatment of diseases, if such article is protected against free competition, as to name, product, composition or process of manufacture, by secrecy, patent, copyright, or by any other means.
Rule 1.—Composition.—No article will be accepted for inclusion in the book New and Nonofficial Remedies, or retained therein, unless its composition be furnished to the Council for publication. For simple substances, the scientific name and the chemical formula, rational or structural, if known, should be supplied. For mixtures, the amount of each active medicinal ingredient in a given quantity of the article must be stated. The general composition of the vehicle, its alcoholic percentage, and the identity of the preservatives must be furnished.
Rule 2.—Identification.—No article will be accepted or retained unless suitable tests for determining its composition are furnished to the Council. In the case of chemical compounds, these shall consist of tests for identity and purity. In the case of mixtures, description of methods for determining the amount and active strength of the potent ingredients shall be furnished, if practicable.
Rule 3.—Direct Advertising.—No article that is advertised to the public will be accepted or retained; but this rule shall not apply to: (a) disinfectants, germicides and antiseptics, provided the advertising is limited to conservative recommendations for their use as prophylactic applications to superficial cuts and abrasions of the skin and to the mucous surfaces of the mouth, pharynx and nose (but not to those of the eye, and the gastro-intestinal and genito-urinary tracts) and provided they are not advertised as curative agents (see comments to Rule 3); and (b) nonmedicinal food preparations, except when advertised in an objectionable manner.
Rule 4.—Indirect Advertising.—No article will be accepted or retained if the label, package or circular accompanying the package contains the names of diseases in the treatment of which the article is said to be indicated. The therapeutic indications and properties may be stated, provided such statements do not suggest self-medication. Dosage may be indicated. (This rule shall not apply to remedies with which self-medication is altogether improbable, to vaccines and antitoxins or to directions for administering or applying remedies when similar immediate, heroic treatment is indicated.)
Rule 5.—False Claims as to Origin.—No article will be accepted or retained concerning which the manufacturer or his agents make false or misleading statements as to source, raw material from which made, or method of collection or preparation.
Rule 6.—Unwarranted Therapeutic Claims.—No article will be accepted or retained concerning which the manufacturer or his agents make unwarranted, exaggerated or misleading statements as to the therapeutic value.
Rule 7.—Poisonous Substances.—The principal label on an article containing “poisonous” or “potent” substances must state plainly the amount of each of such ingredients in a given quantity of the product.
Rule 8.—Objectionable Names.—Proprietary names for medicinal articles will be recognized only when the Council shall deem the use of such exclusive names to be in the interest of public welfare. Names which are misleading or which suggest diseases, pathologic conditions or therapeutic indications will not be recognized (the provision against therapeutically suggestive names does not apply to serums, vaccines and antitoxins, or to foods). In the case of pharmaceutical preparations or mixtures, the name must be so framed as to indicate the most potent ingredients.
Rule 9.—Patented Products and Protected Names.—If the article is patented—either process or product, or both—the number of such patent or patents must be furnished to the Council. Furthermore, if the name of an article is registered, or the label copyrighted, the registration (trademark) number and a copy of the protected label should be furnished the Council. In case of registration in foreign countries, the name under which the article is registered should be supplied.
Rule 10.—Unscientific and Useless Articles.—No article will be accepted or retained which, because of its unscientific composition, is useless or inimical to the best interests of the public or of the medical profession.
Explanatory Comments on the Rules
Introduction.—The Council on Pharmacy and Chemistry was established in February, 1905, by the American Medical Association, primarily for the purpose of gathering and disseminating such information as will protect the medical profession in the prescribing of proprietary medicinal articles. In pursuance of this object, the Council examines the articles on the market as to their compliance with definite rules designed to prevent fraud, undesirable secrecy and the abuses which arise from advertising directly or indirectly to the laity. Such articles as appear to conform to the rules are accepted; and their essential features are described in the annual publication of the Council, New and Nonofficial Remedies, if they come within the scope of this book. These descriptions are based in part on investigations made by, or under, the direction of the Council, but in part also on evidence or information supplied by the manufacturer or his agents. Such interested statements are examined critically, and are admitted only if they appear to be in conformity with the evidence. It is, however, manifestly impossible for the Council to investigate the composition of every complex pharmaceutical mixture, or to check thoroughly every therapeutic claim; it can give only an unbiased judgment on the available evidence. Criticisms and corrections of the descriptions which may aid in the revision of the matter will be appreciated. The Council judges an article entirely by the facts in evidence at the time of its admission. Previous noncompliance with the rules (short of intentional fraud) does not prevent the favorable consideration of an article which is in accord with existing rules. Infringements of the rules after acceptance of an article for New and Nonofficial Remedies, or the discovery that the Council’s information was incorrect, will cause the acceptance to be reconsidered. An article is accepted for New and Nonofficial Remedies, and will continue to be included in the book, with the understanding that serious violations of the rules, after acceptance, will be followed by the omission of the article and publication of the reasons for such omission. The Council desires physicians to understand that the admission of an article does not imply a recommendation. Acceptance simply means that no conflict with the rules has been found by the Council.
Duration of Acceptance.—Unless an agreement to the contrary is made at the time of acceptance, articles admitted to New and Nonofficial Remedies will be retained for a period of three years, provided that during that period they comply with the rules and regulations which were in force at the time of their acceptance. At the end of this period all articles will be carefully reexamined for compliance with existing rules. Particular weight will be given to the question as to whether recent evidence has substantiated claims as to the therapeutic value of any preparation, this evidence to consist partly of recent statements in the literature and partly of the general esteem in which the preparation is held by clinical consultants of the Council. The reacceptance of articles after such reexamination shall be for three years unless a shorter period is specified. Any amendments to the rules, by specific requirements or by interpretation, which may be made after the acceptance of an article, shall not apply to such article until the period of acceptance has elapsed. At the end of this period the article, if it is not eligible under the amended rules, will be omitted.
The Scope of New and Nonofficial Remedies and Appendix.—To aid physicians and manufacturers in deciding what articles come within the scope of this book, or, in other words, to enable physicians to recognize whether an article which is not described in New and Nonofficial Remedies has been omitted because it does not need admission or because it has been rejected, the Council furnishes the following more detailed definitions:
Official Articles.—Articles official in the U. S. P. or N. F. do not require consideration by the Council if they are marketed under the official name and if no unestablished therapeutic claims are made for them.
These do not require consideration by the Council, since standards for them are provided in these books, and enforced under the provisions of the federal Food and Drugs Act, except that they may be mentioned for information. Consideration by the Council becomes necessary if a U. S. P. or N. F. product is offered for sale under a name other than that, or the synonyms, under which the product is described in one of these books of standards, or if the proprietors or their agents advance claims that the product possesses therapeutic properties other than those commonly accredited to it.
Modifications of U. S. P. and N. F. Products.—A pharmacopeial or National Formulary product which is marketed under the official title or synonym, but with well-founded claims that its purity, permanence, palatability or other physical properties excel the official standard, may, if no extraordinary therapeutic properties are asserted, be considered as an official article and held not to be within the scope of New and Nonofficial Remedies. When such products are marketed under the claim that they possess therapeutic properties other than those commonly accredited to the U. S. P. or N. F. products of which they are modifications, they shall be subject to the consideration of the Council.
Specifically Exempted Preparations.—Foods, in general, unless marketed with the claim that they possess therapeutic properties shall not, at the present time, be considered by the Council. Mechanical appliances, at the present time, shall not be considered by the Council. Mineral waters (natural), at the present time, shall not be considered by the Council. With these exceptions, products which in the judgment of the Council are manufactured and marketed in conformity to the principles underlying the rules of the Council may be accepted for N. N. R. Products which are manufactured and marketed in a manner which does not conform to the principles underlying the rules of the Council shall not be accepted for N. N. R. The burden of proof in establishing claims for therapeutic properties of products considered by the Council shall lie with the proprietor or, when a foreign made product, with the agent who markets the product in the United States. To avoid confusion with nonofficial substances marketed under similar names, the Council recommends that official substances be prescribed by their official titles, followed by the abbreviation “U. S. P.” or “N. F.”; thus: Tinctura Nucis Vomicae, U. S. P.; Elixir Gentianae, N. F.
Substances Described in New and Nonofficial Remedies.—In the body of the book will be described simple proprietary substances and their preparations; proprietary mixtures if they have originality or other important qualities which, in the judgment of the Council, entitle them to such place, and important, nonproprietary, unofficial articles. The Council recommends that when the latter are prescribed, they be indicated by the abbreviation, “N. N. R.,” thus insuring to the prescriber the quality of these articles laid down in the book.
Proprietary Mixtures.—A mixture will be considered as proprietary, and therefore requiring consideration by the Council for admission to the book or appendix, if it contains any proprietary article; if it is marketed under a name which is in any way protected, or if its manufacturer claims for it any unusual therapeutic qualities. Proprietary mixtures which are marketed in conformity with the rules are listed in the appendix of the book under the names of the respective manufacturers. Such proprietary mixtures are not admitted to the body of the book, save in the exceptional cases cited in the preceding paragraph.
Nonproprietary Mixtures of Official Substances.—Since the ingredients of such mixtures do not require consideration by the Council, and since the mixtures are not open to the proprietary abuses which call for the work of the Council, it is not necessary that they should be investigated by the Council. The physician must judge whether such mixtures should be directed to be prepared by the pharmacist, or whether he is justified in ordering a ready-made preparation. If he decides to use a ready-made, nonproprietary preparation, he must judge for himself whether it is marketed in accordance with the rules. It should, however, be remembered that the application of a trade name to any substance makes it proprietary.
Explanation of Rule 1: Composition
Secrecy Objectionable.—It is not only the right but also the duty of the physician to know the essential composition of what he prescribes; the Council cannot compromise on this proposition.
Vehicles and Preservatives.—In the case of mixtures, not only the potent ingredient, but also the general character of the vehicle, the presence of alcohol, and the identity of preservatives, or of any other substance, whether added or present as an impurity, must be stated if these can under any circumstances affect the therapeutic action of the article. This, as a rule, does not mean the publication of trade secrets, such as flavors or the details of the working formula.
Trade Secrets.—Furthermore, trade secrets will not be received as confidential by the Council, since it accepts information only with the distinct understanding that this may be freely published, at its discretion.
Inspection of Factories.—The Council does not accept invitations to inspect factories; its concern is with the finished products.
On the other hand, the Council requires that the information be complete and accurate as to medicinal ingredients.
Unofficial Constituents.—Unofficial constituents of proprietary mixtures must be presented by the manufacturer in the regular way and must be acted on by the Council before the preparations containing them can be accepted.
Fraud.—When it appears that a manufacturer has made a deliberately false statement concerning a product, he is asked to furnish an explanation; and if this is not satisfactory, the product will not be accepted, even if the false statement is subsequently corrected or omitted.
Testimonials.—The foregoing paragraph applies not only to statements made to the Council, but also to statements furnished to physicians by the manufacturer or his agents, even when these statements are in the guise of testimonials.
Explanation of Rule 2: Identification
In order to avoid errors in the case of chemical compounds, and to guard against adulterations, lack of potency or strength, and the mistaking of one chemical for another, it is necessary to have at hand suitable tests.
Tests, etc.—If these facts have appeared in the literature, or in standard textbooks, reference to them will be sufficient; but with new chemicals, especially synthetics, the manufacturer or his representatives will be required to supply such tests for publication, as will assure an intelligent opinion of these products.
Physiologic Standardization.—In cases in which chemical methods of identification are unknown or unreliable, physiologic standardization should be employed. The Council considers the phrase “physiologically standardized” or “assayed” as misleading unless the standard and method are published in sufficient detail to permit of their control by independent investigators. It is evident that when no standard is published, it is impossible to know whether the quality is high or low, and the conscientious manufacturer who sets for himself a high standard is placed on a level with the dishonest or careless one who adopts a low standard. Again, if the process of standardization is not published, it is impossible to learn, without actual trial, the relative value of one preparation as compared with that of another manufacturer, or to confirm or disprove the statements of the manufacturer as to the quality of his product.
Standardization of Disinfectants and Germicides.—No disinfectant or germicide of the phenol type will be accepted for New and Nonofficial Remedies whose phenol coefficient, determined according to the method of the Hygienic Laboratory, U. S. P. H. S., is not stated on the label of the preparation.
Explanation of Rule 3: Direct Advertising
Lay Advertising.—The impossibility of controlling the irresponsible claims which are usually made in advertisements to the public, the well-known dangers of suggesting by descriptions of symptoms to the minds of the people that they are suffering from the many diseases described, the dangers of the unconscious and innocent formation of a drug habit, and the evils of harmful self-medication, including the dangers of the spread of many infectious and contagious diseases when hidden from the physician, and similar well-known considerations, are the reasons for discouraging, in the interest, and for the safety, of the public, this reprehensible form of exploitation. Advertising in medical journals, etc., distributed solely to physicians, does not come within the scope of this rule.
Exceptions.—In the case of subjects on which the public should be instructed, as the use of disinfectants, germicides, antiseptics and foods, advertisements to the public, if not in objectionable forms, are considered admissible. In no case shall such advertisements include recommendations for use as curative agents, nor shall the names of any diseases be mentioned in exploitation. If the preparation is sufficiently toxic to require caution in its use to prevent poisoning, this fact shall be stated on the label. On account of the deplorable results which would follow any abuse of this privilege, the conscientious cooperation of manufacturers and their agents in adhering strictly to the limitations laid down is asked; and for the same reason the acceptance of an article which is so advertised as to infringe on these limitations in any essential way (as by naming diseases or by making false and exaggerated claims) shall be summarily rescinded, and the reasons for such action may be published without notice to manufacturer or agent. A disinfectant, germicide or antiseptic will be accepted for description in New and Nonofficial Remedies, and an article of this class which has already been accepted will continue to be included in New and Nonofficial Remedies only on the explicit understanding by the manufacturer and agent that such infringements of the rule will be followed by deletion of the article and by publication of the facts as described.
Foods.—We may divide the foods into three groups. The first group contains the ordinary foods, including the well-known breakfast foods. These do not come under the supervision of the Council in any way. The second group includes a large and important class of manufactured products, such as invalid and infant foods, which in a sense stand between the first and third groups. The public has the same interest in these foods that the physician has, and usually is supplied with full information concerning them. While the primary recommendation of these articles should naturally come from the physician, it cannot be expected that their continued use should depend on repeated prescriptions. Information concerning this group of foods would come naturally and properly from a physician, and the collection and dissemination of this information may very properly be included in the work of this Council. As the products in this class are used extensively, it is not proper to limit their advertising to medical journals, but the advertising should be permitted in the lay press so long as it is conducted in a manner compatible with the rules of the Council. The third group includes medicinal foods proper, such as predigested foods. These have a relatively low food value and are characterized by a high alcohol or preservative content. They frequently contain strictly medicinal substances, or food substances for which distinct therapeutic properties are claimed. These products should be used only on the advice of the physician, and the advertisements should be restricted as in the case of ordinary medicines.
Advertisements in Foreign Countries.—The Council deals primarily, in the interest of the public and of the medical profession, with articles proposed for admission to New and Nonofficial Remedies, and, in determining the status of any article, must take into consideration any statements made regarding it or any method of advertising it employed by the manufacturer or his authorized agents or representatives, whether in this country or abroad. The Council will not regard as within its scope, however, questions concerning the marketing of articles (except the matter of direct advertising to the laity and unwarranted claims or misrepresentations) in any country which has a public body corresponding to this Council.
Explanation of Rule 4: Indirect Advertising
Matter Distributed Solely to Physicians.—It should be remembered that the sole intent of this rule is to protect the physician, so that in prescribing a proprietary medicine he shall not unconsciously advertise proprietary preparations. The rule imposes no restriction on the legitimate methods of bringing a remedy to the attention of the profession, such as advertising in medical journals, circulars and other printed matter distributed solely to physicians. The rule applies only to the package as it may reach the patient.
Naming Diseases on Labels.—The naming of diseases on the label or package is not necessary, as is shown by the very large number of proprietary products which have been successfully introduced without resorting to this expedient. This method of popularizing a proprietary remedy with the laity is most objectionable, and should not be tolerated in any form. In general, therapeutic indications should be omitted from the label and package. The Council will not insist on this point, however, when such indications are so given as not to promote self-medication, particularly in diseases which require expert diagnosis and supervision. It will be considered an infringement of the rule if an article be marketed in bottles which have the name of the article blown into the glass, or if otherwise the name or initials or other distinctive mark of the article is permanently stamped on the container, on the article itself, or is on the stoppers or seals. Articles which are marketed in any of these ways are not accepted for New and Nonofficial Remedies. Readily removable labels are not objectionable, nor is the permanent affixing of the firm’s initials or name to the trade package if such initials or name is not suggestive of the article. The Council does not countenance the use of an accepted article for advertising other articles which have not been accepted by the Council.
Explanation of Rule 5: False Claims as to Origin
Source.—No false or misleading statement in regard to an article can be permitted concerning the source of material from which it is made, or the persons by whom it is made. Some glaring frauds of this nature have been perpetrated in the past, and this rule is intended to prevent such imposition.
Explanation of Rule 6: Unwarranted Therapeutic Claims
Therapeutic Questions.—This rule insists that the claims of manufacturers or agents concerning the therapeutic properties of their products must be compatible with demonstrable facts. Manufacturers will be held responsible for all statements made or quoted in their advertising “literature” regarding their products. Recognizing the existence of honest differences of opinion on many therapeutic questions, the Council desires to be liberal in the application of this rule. It is natural that a manufacturer should be partial toward his own product, and a moderate degree of emphasis in advertising may not be objectionable. The Council, however, will not admit claims which are neither in harmony with already accepted facts nor supported by acceptable evidence. In doubtful cases the Council considers these questions with the advice and cooperation of its staff of clinical consultants.
Clinical Evidence.—To be acceptable, the clinical evidence must offer objective data with such citation of authority as will enable the Council to confirm the facts and establish the scientific value of the conclusions drawn. Clinical data are worthless when the author is not cited. The facts on which claims with regard to the value of a remedy are based must have been rendered accessible for investigation and confirmation by disinterested observers, either through publication or through the records of a hospital or other institution.
Explanation of Rule 7: Poisonous Substances
Poisons.—For the information of the pharmacist or dispenser, and to enable him to safeguard the interests of the patient and the physician, all articles containing such potent agents as the poisonous alkaloids and other organic substances and the salts of some of the metals should have the exact amount of these ingredients which is contained in the average adult dose stated on the label.
Explanation of Rule 8: Objectionable Names
“Coined” Names.—Many of the abuses connected with proprietary medicines arise from “coined” proprietary trade names. Such names will not be recognized by the Council unless in particular instances the Council shall deem their use to be in the interest of public welfare. In every such exception the burden of proof, both for establishing and for continuing the exception, lies with those who market the product.
Proprietary (“Trade”) Names When Permitted.—In consideration of the benefits which may come from the discovery of a therapeutic agent, the Council concedes to the person or firm which, by right of discovery, controls such a product the right to name it. The Council will offer no opposition to an arbitrary name for such a new product, provided it is not misleading, therapeutically suggestive, or otherwise subversive of scientific pharmacy and therapeutics. If the discovery that a previously known substance has therapeutic value is deemed of sufficient importance, the Council may recognize a name for such a substance if the name is applied by the person who makes the discovery; or, with the consent of the discoverer or in the absence of any protest on his part, the Council may recognize a name applied by the firm which first makes such a product available to physicians. In the interest of rational drug therapy, the Council recommends that trade names be coined so as to indicate the potent element or constituent.
Scientific Names.—When the proprietary or trade name for an article is considered insufficiently descriptive of its chemical composition or pharmaceutical character, the Council may require as a condition for the acceptance of such articles that a descriptive scientific name satisfactory to the Council appear on the labels, circulars and advertisements for such an article. For all definite chemical substances it is required that the scientific name be given prominence on the labels, in circulars and advertisements.
Proprietary Names for Unoriginal Articles.—Proprietary names will not be recognized for articles which are included in the U. S. Pharmacopeia or National Formulary or for unessential modifications of such articles. Neither will proprietary names be recognized for substances or mixtures which are described in medical or pharmaceutical publications. In the marketing of unoriginal articles, the legitimate interests of the producer are fully served by identifying such products by appending the name or initials of the manufacturer or agent, or by the use of a general brand mark. No objection will be made by the Council to the use of such brand marks, provided that in no case shall such mark be used as a designation for an individual article.
For any product which, by reason of the absence or lapse of patent rights or for other reasons, is open to manufacture by more than one firm, the Council reserves the right to select a common name and to provide standards of identity, purity and strength, and then will accept such article only if it is marketed under the title adopted as the N. N. R. name or the name under which such article was introduced (to which may be appended the firm’s identifying mark).
N. N. R. to U. S. P.—When an article which has been accepted for New and Nonofficial Remedies is admitted to the U. S. Pharmacopeia or National Formulary, it will be omitted from New and Nonofficial Remedies one year after such standardization if the name of such article is used in these standards either as the main title for the product or as a synonym. If the name under which the article is described in New and Nonofficial Remedies is not used in these books of standards, the proprietary preparation will be retained provided the official name is given prominence on the labels and in the circulars and advertisements of such article. When the Council adopts a common name for an article that has been admitted under another name, it will be continued under the older name only on condition that the Council name be given prominence on the label and in the circulars and advertisements for such article.
Pharmaceutical Preparations and Mixtures.—These, with rare exceptions, are not original in composition and there is seldom any reason why they should be endowed with arbitrary names. On the contrary, it is important that the prescriber should be reminded constantly of their potent ingredients.
Therapeutically Suggestive Names.—Articles bearing therapeutically suggestive names will not be accepted for New and Nonofficial Remedies, first, because they are likely to lead physicians into prescribing names instead of remedies, and second, because they tend to encourage unwarranted self-medication by the laity. Even if the name is at first apparently meaningless to the public, its meaning will soon be understood because patients soon learn the technical names applied to their diseases and symptoms. The prohibition against therapeutically suggestive names is not applied to serums, vaccines and antitoxins, because the accepted nomenclature of the specific organisms used in their preparation makes this unavoidable and because self-medication with them is improbable.
Explanation of Rule 9: Patents, Trademarks, Copyrights, Etc.
Protection.—This information is important as a means of determining the legal status of medicinal articles and as an aid to their ready recognition in current publications.
Explanation of Rule 10: Unscientific and Useless Articles
Unscientific Compounds.—The use of articles which are unessential modifications of official or established nonproprietary articles is unscientific and serves no useful purpose. The Council will not accept products which are scientifically unsound and which, therefore, must be considered useless or inimical to the best interest of the medical profession and the public. This class includes compounds or mixtures containing an excessive number of active ingredients; those compounds or mixtures the components of which are of no probable assistance to one another, and those articles which are of no therapeutic value.
Unessential Modifications of Official Substances.—The subterfuge of obtaining proprietary rights over an official or established nonproprietary product, by introducing unessential modifications, also tends to confusion and abuses, and such articles will not be admitted by the Council. Essential and important modifications, however, will receive recognition. (The Council interprets the term “established nonproprietary product” as applying to a preparation of any formula which has been published through any recognized or reasonably accessible channel of publication, prior to its appropriation or modification by a manufacturer.) Duplicates of biologic products accepted under the name of the manufacturers will not be accepted under the names of the distributors.
THE COUNCIL ON PHARMACY AND CHEMISTRY, PRESENT AND FUTURE [A]
W. A. Puckner, Phar.D.
Secretary, Council on Pharmacy and Chemistry
The World War marked an epoch in the existence of the Council on Pharmacy and Chemistry, as it did in all human endeavors. The information and experience which had been accumulated by the Council during its thirteen years’ existence was drawn on by our government, directly or indirectly, and it also received consideration in England, France,1 Belgium, Holland,2 Italy,3 Sweden and elsewhere. In the world wide readjustment that has begun, the efforts of the Council, past and present, will influence the plans of those who engage in the manufacture or sale of medicines, and, undoubtedly, will be the incentive to the establishment of similar bodies in other countries.
As secretary of the Council almost from the time of its organization in 1905,4 and knowing the work of its members and its collaborators, I am firmly convinced that this body has deserved the endorsement and support given it by the American medical profession. I welcome this opportunity to present an outline of the Council’s past activities and to speak of some of the problems of the future, because I feel assured that a knowledge of its endeavor to improve drug therapy will increase the profession’s confidence in the Council and add to the number of its supporters.
THE COUNCIL’S ACTIVITIES
Organized primarily for the purpose of putting a stop to false declarations with regard to the composition of proprietary medicines, the Council’s activities have broadened until its work may be characterized as “a propaganda for the rational use of drugs.” The following are some of its activities:
1. New and Nonofficial Remedies.—This is an annual volume, issued by the Council. It describes both proprietary and nonofficial, nonproprietary drugs which are deemed worthy of consideration by the medical profession. To be admitted to this book, a preparation must comply with certain definite rules which stipulate, in effect, that its composition be declared, that no untrue or grossly exaggerated claims be made for it, and that it shall give promise of having therapeutic value.
With the exception of a few which are still under consideration, the Council has considered all proprietaries whose owners or accredited agents have requested that an examination of the products be made, and it has admitted to the book those which were found eligible. In addition, the Council has examined all of the more important or widely exploited proprietaries, even when no examination was requested, and it has admitted those of this group which were found eligible. Further, the Council has admitted to the book certain nonofficial, nonproprietary articles which seemed to give promise of therapeutic usefulness, and it has established standards for the control of their identity and purity, and listed those brands which complied with these standards.
As most proprietary medicines are of a more or less experimental nature, they are accepted for inclusion in New and Nonofficial Remedies only for a limited time—usually a period of three years. At the expiration of the period of acceptance, each preparation is reexamined and retained only if the claims made for it and the present day knowledge of its value permit this action.
Since manufacturers give information only in regard to their own products, New and Nonofficial Remedies groups together articles of a similar character, and includes in each case a general discussion of the group for the purpose of comparison, not only with each other, but also with the established or pharmacopeial drugs which members of the group are intended to supplant.
In brief, New and Nonofficial Remedies is a book in which are described preparations that have been accepted by the Council. The description includes facts that the physician should have. It is a book that should be in the hands of every physician who prescribes medicines, and who wishes to know the facts regarding the newest remedies. It is the only book in which he can find information relative to proprietary medicines that are worthy of his patronage. It will protect the physician who makes use of it against the wiles of the promoters of products not worthy of his patronage. It would certainly be of use to the physician when the detail man calls on him, for if he were being importuned to prescribe or use samples of something which he had not heretofore used and which he was unable to find in N. N. R., he might ask the detail man why. In the nature of things few physicians are sufficiently expert in chemistry and allied sciences to be able unerringly to discriminate between the true and the false as regards many preparations that he is asked to prescribe.
2. The Reports of the Council on Pharmacy and Chemistry.—A medicament may be inadmissible to New and Nonofficial Remedies for various reasons; it may be worthless or irrational, its composition may be secret or indefinite, or it may be exploited under exaggerated or unwarranted claims or in a way otherwise detrimental to the public health and scientific medicine. Of these various reasons which make an article unacceptable, the manufacturer obviously may remove all except the first, viz., worthlessness or irrationality. Consequently, a preparation which has been presented for admission is not definitely rejected until after its proprietor has been informed of the objections to his product and has failed to bring the preparation in conformity with the Council’s rules. When a preparation is found definitely inadmissible to New and Nonofficial Remedies, that is, when the proprietor cannot or will not make it acceptable, the Council prepares a report for publication. These reports are sent for publication to The Journal of the American Medical Association, and later published in the annual “Reports of the Council on Pharmacy and Chemistry.” The more important of these are also published in the book, “The Propaganda for Reform in Proprietary Medicines.”
3. Useful Drugs.—Since the domination of proprietary medicines, which was retarding medical advance and threatening therapeutic chaos, had been made possible only by the insufficient and inefficient instruction given in medical schools in subjects having to do with drugs, the Council appointed a Committee on Medical Teaching to secure the cooperation of teachers in materia medica, pharmacology and related branches. This committee has endeavored to effect an improvement in these courses of instruction. One of the results of this work was the selection of a list of drugs to serve as a basis of materia medica instruction and thus insure that medical students shall be better informed with regard to the therapeutic worth of a few well established drugs, rather than, as in the past, leaving school with a smattering of knowledge about many drugs. The outcome of these efforts is the publication of “Useful Drugs,” a concise but thorough and up-to-date discussion of the actions, uses and dosage of the more important drugs. The list of drugs presented in this book is now the basis of instruction in many schools; and many state examining boards are confining their materia medica questions to the drugs in the list.
4. Epitome of the U. S. P. and N. F.—To encourage the use of official drugs and to make available an estimate of their therapeutic value, a committee of the Council prepared an abstract of the U. S. Pharmacopeia and the National Formulary. This booklet, the “Epitome of the U. S. Pharmacopeia and National Formulary,” presents those portions of these books which are of interest to physicians, and in addition, gives a concise statement of the therapeutic usefulness of the drugs and preparations described in them.
5. Patent Law Reform.—Some of the worst abuses connected with the exploitation of proprietary medicines have been made possible by our patent and trademark laws and the method of their interpretation and enforcement. The Council, therefore, appointed a committee to study these laws and the various propositions advanced for their improvement. This committee has published, from time to time, reports on various phases of our patent and trademark laws and recently summarized these reports in an address5 sent to the commissioner of patents and the interested congressional committees. It is hoped that by means of these reports physicians will be enabled to give intelligent support to a revision of the patent and trademark laws when legislation is proposed.
6. Therapeutic Research.—Through its Committee on Therapeutic Research, and with the aid of funds provided by the Board of Trustees of the American Medical Association, the Council has encouraged the investigations of questions which might lead to a better understanding of the action of drugs. These investigations are brought together in the annual reports of the Committee on Therapeutic Research, and are an important addition to our knowledge of drug action.
In the past, the Council has in particular encouraged the investigation of the action and therapeutic value of widely used drugs regarding which our knowledge is still unsatisfactory. These investigations have included a study of the action of strychnin in cardiac disease, a comparison of the action of absorption and excretion of iodid preparations, a study of the pharmacology of the opium alkaloids, etc. Appreciating that the available knowledge of proprietary drugs is one sided in that it comes from investigations made by interested pharmaceutical concerns or from investigations made at the instigation of these firms, the Council is planning a comprehensive study of many of the synthetic drugs that have gained some vogue during recent years.
THE FUTURE
Medical research, and efficient instruction in therapeutics and related subjects, spell a diminishing influence of commercial medicine over rational therapeutics. The fact that the present shortage of German synthetics has not handicapped seriously the practice of medicine should be a lesson to American physicians for many years to come.
On the other hand, it must be remembered that the publicity given to the reports of the Council and to other contributions toward rational therapeutics by The Journal of the American Medical Association, the journals of the state organizations, and a few personally owned publications, is as nothing when compared with the persistent and wide publicity given to the propaganda of the proprietary houses. While a report setting forth the objections to a proprietary is published but once, the firm’s laudatory pronouncement goes forth again and again until the Council’s report is completely overwhelmed and forgotten. Manufacturers of proprietaries not only keep in close touch with the practicing physician by means of house organs, special “literature,” or by traveling representatives, but many of the firms, through the meritorious lines of pills, tablets, tinctures, etc., which they put out, also obtain and hold the good will and confidence of a large proportion of the medical profession.
Furthermore, some of these firms may gain the confidence of the medical profession through these high grade pharmaceuticals, and certain of their proprietaries may be of distinct therapeutic value but may fail to be acceptable for New and Nonofficial Remedies, because they do not conform to the reasonable rules of the Council. These firms do not find it profitable to force the sale of their regular nonproprietary pharmaceuticals by unwarranted claims or objectionable methods, yet they may consider it good business to market certain proprietary products by means of claims which are extravagant and without warrant, and which will lead to indiscriminate use by the profession and the public. In a word, where there is one dollar spent on behalf of rational medicine, thousands are spent for the purpose of increasing the sale of preparations which directly or indirectly are a detriment to the public health, to medicine, and to the pocketbook.
That the day of the secret nostrum of the pseudo-chemical company is not yet past is well illustrated by the recent introduction of an asserted arsphenamin preparation called “Syphilodol.” The A. M. A. Chemical Laboratory proved one form of this asserted French discovery to be essentially a pill of mercurous iodid. Another form of Syphilodol (for intravenous administration) had all the characteristics of water, and appeared devoid of any potent ingredient. Though the advertising sent out by the promoters in regard to its composition was suspiciously evasive, the Illinois Medical Journal published an advertisement of “Syphilodol,” which, possibly by a coincidence, appeared above an appeal to “Our Readers” to use wares advertised in that journal.
While such rank deceptions as “Syphilodol” are not common, there are more subtle deceptions that are even more dangerous. Types of widely exploited remedies of today comprise so-called ethical specialties composed of well known and established drugs (with “jokers” hidden away somewhere) or preparations which have a plausibly fascinating pseudo-scientific background of radiant energy, colloidal chemistry, nonspecific protein reaction, or something of the sort. The latter class of preparations in particular appeal to physicians who are striving hard to keep pace with modern science. Exposure of their fallacies requires most careful consideration on the part of the Council.
Progress toward a rational and scientific drug therapy must continue, and, therefore, it is important that the Council on Pharmacy and Chemistry should continue to make the investigation of proprietary medicines its chief work. Investigation of a proprietary medicine, however, and a report of such investigation are of value in direct ratio only to the number of physicians who read the report, endorse it and act in accordance with its conclusions. In order that you may determine to what extent those preparations which are admitted to New and Nonofficial Remedies deserve your interest and confidence, it will be worth while briefly to outline the rules which govern the Council in the admission of articles to New and Nonofficial Remedies.
RULES GOVERNING THE ACCEPTANCE OF ARTICLES FOR N. N. R.
Composition.—Rules 1 and 2, and in a measure 5, 7, and 9, deal with the composition of articles. Rule 1 requires that the quantitative composition of an article be furnished the Council for publication. Rule 2 requires that the manufacturer furnish methods whereby the composition of products that are definite chemicals or the potent constituents of mixtures may be determined. The Council does not require that the process of manufacture of an article be declared unless this becomes necessary in order to judge its composition. Rule 5 requires that statements with regard to the origin and source of an article shall be truthful. Rule 7 requires that for the guidance of the dispenser, the amounts of poisonous ingredients of a preparation be placed on the label. Rule 9 requires that if patent rights are claimed for a product, the Council be informed on this point.
That it is not only the right but also the duty of the physician to know the composition of what he prescribes for his patients is so generally admitted that few have attempted to market preparations of avowedly secret composition. When the Council first began its work, it was common to see chemical formulas or statements of composition published which a chemist or a pharmacist was able to pronounce at a glance as impossible.6 It was not unusual to find that the promoter published “a formula” for his preparation, rather than “the formula.”7 Today, however, a more prudent, if not more honest, course is pursued. This gives a “formula” which is correct so far as it goes, but which fails to divulge the actual composition of a preparation. When it is considered that many physicians are not any too conversant with the chemistry and pharmacy of drugs, it is not surprising that some administered the proprietary “Venarsen,” regarding the composition of which they had only the vague statement that it was “... a comparatively nontoxic organic arsenic compound, 0.6 gm. representing 247 mg. (33⁄4 grains) of metallic arsenic in chemical combination ...” in the belief that a preparation similar to that first introduced as salvarsan was being used. That “Venarsen” contained its arsenic as sodium cacodylate—a notoriously inactive state of combination—does not justify the intravenous administration of a drug of unknown composition.
While for the present it probably is not feasible to require, on the part of those who manufacture medicinal preparations, such professional training as is required of those who prescribe and those who dispense them, it certainly is not too much to require, as does Rule 2, that a manufacturer shall be able to demonstrate that his preparation has the composition claimed for it. Nor is it sufficient for him to know that the ingredients claimed as constituents were used in the manufacture. The fallacy of his method of reasoning was furnished by the physician who reported that he had personally added the required amount of mercuric iodid for a batch of “Mercol” which, nevertheless, was devoid of mercury.8 Acceptance of this rule by manufacturers will permit physicians to have a more accurate knowledge of the composition of preparations such as “Taka-Diastase”9 and “Iodeol”.10
A requirement similar to that of Rule 5 is contained in the Federal Food and Drugs Act and so no objection has been made to this rule which requires a truthful statement of the origin and source of articles. An illustration for the need of the rule was furnished by the one time popular “Vin Mariani”11 which, though very French in its makeup, was found to be largely of the “made in the United States” variety of tipple.
The issuance of a patent for a medicinal product does not prove that such a product presents a discovery or that its owner is entitled to a temporary monopoly, yet it is only fair to physicians and to other manufacturers that notice of such patent claims be given. Hence, the Council publishes in New and Nonofficial Remedies the information bearing on this point.
Lay Advertising.—Rules 3 and 4 provide against the recognition of articles that are advertised to the public directly or indirectly, exempting from this requirement preparations which the Council believes are safe to be so advertised.
It has been held with some justice that certain shotgun proprietaries are purchased by the public with as much circumspection as they are ordered by those physicians who are addicted to the prescribing of them; but even the exploiters of these mixtures have not denied that the use of medicines by the public on its own initiative is surrounded with many objections. Hence the practice of self medication should not be encouraged by prescribing or using those preparations advertised for public use.
The only objection to the rule has come from a firm which markets a brand of liquid petrolatum, the Standard Oil Company of Indiana. The Council has considered the question of exempting simple laxatives from the restrictions of Rules 3 and 4 as it has exempted antiseptics and nonmedicinal foods. The conclusion was, however, that the excessive use of a simple laxative like a liquid petrolatum, when prompted by newspaper exploitation, is likely to be detrimental to health by overuse as well as by misuse.
The indirect advertisement to the public, which Rule 4 provides against, has been the means of inducing the extensive lay use of “Antikamnia,” “Bromidia” and “Fellows’ Syrup.” Naturally Rule 4 has been bitterly opposed by most proprietary firms. Arguing that many physicians dispense their own drugs, pharmaceutical firms have insisted that every medicinal preparation should bear on its label, not only the dose of the preparation, but also a statement of the diseases in which the article is indicated. Whether manufacturers anticipated the profession’s resentment toward the claim that physicians determine the treatment and perhaps the diagnosis by means of the statements on labels, or because the Shirley amendment to the Food and Drugs Act makes the proprietor responsible for therapeutic claims on the label of a medicine, it is a fact that fewer preparations than formerly need to be refused on account of infringement on this rule. In fact, some thoroughly objectionable proprietaries make a show of being “ethical” by omitting all therapeutic discussion from the labels of their preparations.
Therapeutic Claims.—Rule 6 makes ineligible for New and Nonofficial Remedies any articles regarding which the manufacturer or his agents make unwarranted, exaggerated or misleading statements as to the therapeutic value. Recognizing the long established custom of therapeutic exaggeration, it has been most difficult to determine the degree of conservatism which might with fairness be required of a manufacturer. In view of the common acceptance of individual impressions as dependable evidence, it is often almost embarrassing to declare as incompetent the statement of some well meaning and all-too-kind-hearted doctor. However, as the pitfalls of haphazard clinical trials become better known and the physician’s mistrust of glowing accounts of marvelous cures more outspoken, the manufacturers’ claims will be more moderate.
Nomenclature.—Were it possible to enact and enforce a law which would oblige manufacturers to sell their medicinal products under properly descriptive names and which would make it illegal for a physician to prescribe it unless he understood the meaning of such properly descriptive titles, then the Council might safely disband. In that case, physicians would discontinue the use of most proprietary preparations in favor of established drugs, and successful newcomers might each year be counted on the fingers of one hand. Such a rational nomenclature is not to be thought of, at least in our generation. Rule 8 requires that the name of an article shall not be misleading, that it shall not be therapeutically suggestive, and that established drugs shall not be disguised by fanciful titles. It recognizes the right of discoverers of new drugs to name their discoveries, and interposes no objection to arbitrary names for such products so long as such names are not misleading or do not suggest the therapeutic uses of the products. As the rule provides against the recognition of coined names for established nonproprietary drugs, so it requires that mixtures of drugs shall bear names descriptive of their composition. It would be a long step forward if physicians would recognize more fully the objections to the many proprietaries which have, as their only point of originality, a non-descriptive name for an old drug or a mixture of well known drugs. It is an encouraging sign that the Federal Trade Commission, when issuing licenses for the manufacture of synthetic drugs introduced under German patents, stipulated that all manufacturers authorized to make a given drug shall use the same name for it.
Irrational Articles.—Rule 10 provides against the recognition of an article which, because of its composition, is useless or inimical to the best interests of the public and medical profession. This rule excludes medicaments which (1) are unessential modifications of established articles, or (2) are of no therapeutic value or (3) are irrational. With regard to the recognition of mixtures or compounds containing two or more active ingredients, the Council requires that the manufacturer establish the rationality of its combination. The rule has prevented the recognition of many unnecessary so-called ethical specialties. Though a mass of testimonials was often to be had for them, these contained no evidence that the mixture was superior to its potent ingredient, or that its therapeutic effect had been determined. That there is a healthy tendency to use single drugs for their definite action and to discard combinations (be they shotgun proprietaries or “mixed” vaccines) is perhaps best illustrated by the fact that at the last revision of the U. S. Pharmacopeia a considerable number of complex antiquities were omitted from that book.
Feeling confident that this meets with the endorsement of the profession, the Council is examining more critically the evidence for the value of pharmaceutical mixtures.—(From The Journal A. M. A., May 10, 1919.)
[A] Read before the Chicago Medical Society, March 26, 1919.
1 “New and Nonofficial Remedies” in France, Foreign News, J. A. M. A. 71:1331 (Oct. 19) 1918; 70:1783 (June 8) 1918.
2 Nederl. Tjdschr. v. Geneesk. Oct. 5, 1918, p. 1201.
3 An Italian View of the Proprietary Evil, Foreign News, J. A. M. A. 71:840 (Sept. 7) 1918; The Council on Pharmacy and Chemistry and the Patriotic Medical League in Italy, ibid. 71:918 (Sept. 14) 1918.
4 Although the Council on Pharmacy and Chemistry was established in 1905, it is likely that only a small percentage of physicians know just what the Council is, or have any conception as to its personnel and its ability to judge the available evidence for proprietary medicaments. The personnel has changed from time to time since 1905. At present its membership is: C. L. Alsberg, A.M., M.D., chief of the Bureau of Chemistry, U. S. Department of Agriculture, Washington, D. C.; R. A. Hatcher, Ph.G., M.D., professor of pharmacology, Cornell University Medical College, New York City; A. W. Hewlett, M.D., professor of medicine, Leland Stanford Junior University Medical School, San Francisco; John Howland, M.D., professor of pediatrics, Johns Hopkins University Department of Medicine, Baltimore; Reid Hunt, M.D., professor of pharmacology, Harvard University Medical School, Boston; Henry Kraemer, Ph.D., professor of pharmacognosy, University of Michigan College of Pharmacy, Ann Arbor, Mich.; W. T. Longcope, A.B., M.D., Bard Professor of the Practice of Medicine, College of Physicians and Surgeons of Columbia University, New York City; G. W. McCoy, M.D., director of the Hygienic Laboratory, United States Public Health Service, Washington, D. C.; Lafayette B. Mendel, Ph.D., Sc.D., professor of physiologic chemistry, Sheffield Scientific School, Yale University, New Haven, Conn.; F. G. Novy, M.D., Sc.D., professor of bacteriology, University of Michigan, Ann Arbor, Mich.; W. W. Palmer, B.S., M.D., associate professor of medicine, College of Physicians and Surgeons of Columbia University, New York City; L. G. Rowntree, M.D., professor of medicine, University of Minnesota Medical School, Minneapolis; Torald Sollmann, M.D., professor of pharmacology and materia medica, Medical Department, Western Reserve University, Cleveland; Julius Stieglitz, Ph.D., Sc.D., Chem.D., vice chairman of the Council, professor of chemistry, University of Chicago, Chicago; G. H. Simmons, M.D., LL.D., chairman of the Council, editor of The Journal of the American Medical Association, Chicago, and W. A. Puckner, Phar.D., secretary of the Council, director of the Chemical Laboratory of the American Medical Association, Chicago.
5 Need for Patent Law Revision, A. M. A. Council on Pharmacy and Chemistry Reports, 1917, p. 130.
6 Puckner, W. A.: The Abuse of Chemical Formulas, Reports A. M. A. Chemical Laboratory 3:7, 1910.
7 The Formula for Glyco-Thymoline, J. A. M. A. 52:147 (Jan. 9) 1909.
8 Hunt, Reid, and Seidell, Atherton: Howell’s Mercol, J. A. M. A. 52:225 (Jan. 16) 1909. Howell’s Mercol Again: Another Analysis Fails to Reveal the Presence of Mercury, J. A. M. A. 52:1595 (May 15) 1909.
9 Taka-Diastase and Liquid Taka-Diastase: Report of the Council on Pharmacy and Chemistry, J. A. M. A. 59:50 (July 6) 1912.
10 Iodeol and Iodagol: Report of the Council on Pharmacy and Chemistry, J. A. M. A. 69:1725 (Nov. 17) 1917.
11 Vin Mariani: Official Report of Council on Pharmacy and Chemistry—With Comments, J. A. M. A. 47:1751 (Nov. 24) 1906.
“ACCEPTED BY THE COUNCIL ON PHARMACY AND CHEMISTRY”
Under the caption given above, the Journal of the Missouri State Medical Association, in its July issue, speaks editorially as follows:
The Council on Pharmacy and Chemistry of the American Medical Association is a department of our national organization that has not received the plaudits and encomiums of a wildly joyous medical profession nor the grateful praises of the enthusiastic manufacturer of pharmaceuticals. The Council seems indeed to be the unloved child of the entire family of subsidiary bodies of the association. Perhaps the reason for this may be found in the character of its duties, for the Council must expose fraud, sometimes in high places, and protect the physician from being duped by avaricious persons and by persons who are themselves sometimes the victims of their own credulity. It thus happens that the sale of some proprietary article previously held in high esteem by the practitioner proves valueless, perhaps even fraudulent. The practitioner, however, may have credited much of his success in treating certain conditions to that preparation and the maker has had success in accumulating dollars from its sale and both parties emit a loud and vicious roar against the Council, because they both lose money. Nobody wants to be “protected” against making money—make it honestly, if possible, but make it—but this black sheep among the Councils of the American Medical Association insists on their making their money honestly!
Despite many obstacles thrown into its path, the Council on Pharmacy and Chemistry has serenely pursued its allotted tasks, corrected its mistakes, improved its methods, and today stands as the only medium to which the honest physician may turn for information—not misinformation—regarding proprietary articles. During the war the Council and the chemical laboratory were in close cooperation with the Surgeon-General’s Office, testing and investigating every article offered to the government for the treatment of sick soldiers. The variety and the number of fakish and fraudulent stuff offered to the Surgeon-General was a pitiable exhibit of the mental gymnastics of some people. Just now the Council and the laboratory have a new and important field before them, i. e., to protect the physicians against worthless and useless serums, vaccines and synthetics. It will be the Council’s unpleasant duty to expose the fraudulent and useless among these articles and stamp truth on those found worthy.
We seem to have wandered from the topic in our caption, but not so in reality, because the burden of our thought is to lend our influence to the spread of the motto of the Advertising Clubs of the World, namely, “Truth in Advertising.” It is our purpose to stimulate a larger degree of enthusiasm for the work of the Council on Pharmacy and Chemistry and the Chemical Laboratory, a more generous flow of inquiries concerning articles unfamiliar to the physician, and particularly to urge that the words “Accepted by the Council on Pharmacy and Chemistry of the American Medical Association” be printed on the label and on all advertising circulars of proprietary articles that have been admitted to New and Nonofficial Remedies. Then, when pamphlets and circulars are received by physicians they will read the statements of manufacturers with sympathetic understanding and with full confidence in the verity of the declarations. The importance of creating just that sort of receptivity in the mind of the prospective buyer is so well known to the astute publicity expert that it is needless for us to dwell on its advantages. Every proprietary article advertised in our journal, in The Journal of the American Medical Association, and in the other state association journals, as well as in several well-edited privately owned journals, does in effect say to the reader that the articles so advertised are accepted by the Council because only proprietary articles so accepted are accepted by us. The fact is further acknowledged when these firms are permitted to exhibit their goods at our annual sessions for again the rule is enforced that only proprietary articles which have been approved by the Council may be placed on display.
Why not complete the circle of ideas—it would not be a “vicious circle”—by printing on labels, in advertisements and circulars, the words: “Accepted by the Council on Pharmacy and Chemistry”?—(From The Journal A. M. A., Aug. 2, 1919.)
HELPING THE COUNCIL
If they were built that way, the members of the Council on Pharmacy and Chemistry of the American Medical Association might become discouraged at the apparent indifference of many members of the medical profession to their efforts. There are many physicians who, while figuratively patting the Council on the back, actually do nothing to aid its efforts. On the other hand, there are men in the profession who give the Council active support instead of merely passive appreciation. The letter that follows was written by such a man to a pharmaceutical concern:
I am receiving circular advertising from you concerning —— —— solution, and I am writing to suggest that until these products have been approved by the Council on Pharmacy and Chemistry of the American Medical Association, you are wasting your postage on the practice. Aside from the fact that these products do not appeal to me personally, I feel that I am not in a position to judge the value of such products and I depend entirely on the large clinical opportunities of the Council on Pharmacy and Chemistry of the American Medical Association in addition to their laboratory facilities, in such matters as these. I may, therefore, with all due respect, suggest that ... it will pay you to eliminate my name from your mailing list.
The members of the Council on Pharmacy and Chemistry are working week in and week out without remuneration. Few appreciate how much these scientific men are doing for rational therapeutics; fewer still realize how much has been accomplished through their efforts, or how much more could be accomplished if every physician who at least believes in the work of the Council would give it his full support.—(Editorial from The Journal A. M. A., Nov. 6, 1920.)
DELAYS IN PASSING ON PRODUCTS
Report of the Council on Pharmacy and Chemistry
The Council has adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
The Council frequently receives inquiries—some of them accompanied by expressions of impatience—concerning articles, reports on which appear to be delayed. It therefore seems advisable to make a statement of some of the factors which enter into this problem.
The Council fully realizes the importance of giving prompt information to the profession with regard to proprietary medicines under consideration. It therefore acts as soon as sufficient information is available to justify a definite judgment, and publishes its conclusions as soon as possible. When adequate information is available at the outset, there is no delay in the publication of the Council’s conclusions.
Unfortunately, but very naturally, there are many cases in which the information available at the time the product is submitted is not sufficient to justify the Council in coming to definite conclusions for or against the preparation. In some cases the manufacturer possesses the required information, but to obtain it from him takes time; in other cases the manufacturer does not possess the information—perhaps he did not realize the inadequacy of his evidence until the subject was brought to his attention by the Council.
Such cases might be dealt with in either one of two ways: The Council might at once reject the article because the claims for it are not supported by adequate evidence; or, the Council might suspend judgment and give the manufacturer an opportunity to supply the information.
The first method—immediate rejection—would obviously be felt by manufacturers as a hardship. To afford the fullest possible opportunity for the presentation of the case, the Council follows the second method; that is, it suspends judgment and withholds publication of a report until reasonable time has been afforded for furnishing the required information, provided the manufacturer or agent appears to be making honest and diligent efforts to supply it. The collection and compilation of such information is sometimes a lengthy process, especially when the products are of foreign manufacture.
Although it would be easier for the Council to render an immediate decision than to assist manufacturers to supply the data necessary for the formation of an authoritative judgment, the Council cannot yield to importunities for hasty action. It must rely on the medical profession to bear in mind that the character of a product under consideration by the Council has not yet been determined. The Council holds that, during this stage, a product is suitable, at most, for experimental use.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 119.)
COOPERATION OF THE PHARMACEUTICAL HOUSES
Report of the Council on Pharmacy and Chemistry
In reply to the suggestion made last year by President Bevan that there should be closer cooperation between the large pharmaceutical houses and the Council on Pharmacy and Chemistry, the Council submitted to the Board of Trustees of the American Medical Association the statement which appears below:
“Cooperation of the Pharmaceutical Houses: At the opening meeting of the House of Delegates last year, President Arthur Dean Bevan suggested the desirability of greater cooperation between the large pharmaceutical houses and the Council on Pharmacy and Chemistry. The need of such cooperation has been recognized by the Council from the first. In no one direction has the Council made greater effort than in its endeavor to secure the fullest cooperation of the various pharmaceutical houses. The difficulty has been, and always must be, the fundamental antagonism between objectives that are largely commercial on the one hand and purely scientific on the other. Nevertheless, the Council has always believed—and has acted on the belief—that there is a possible middle ground wherein the interests of therapeutics would not be injured but would go hand in hand with a commercial development based on enlightened self-interest.
“The profits to be made by a pharmaceutical house from the sale of a staple drug—a pharmacopeial, National Formulary, or nonproprietary preparation—which enters into free competition with other drugs of the same kind, are moderate; the profits to be made from the sale of a proprietary medicine on which the manufacturer holds a monopoly are usually large—sometimes enormous. There are, broadly, two kinds of proprietary preparations advertised to physicians: One represents laborious research ending in the production of a new medicinal chemical; this product can be patented and the manufacturer can obtain a seventeen-year monopoly on its manufacture and sale. The other represents no research but comprises simple mixtures—frequently of the “shotgun” variety—of well known pharmaceuticals, or biologic products sold under trade names. As these do not represent anything new or original the manufacturer is unable to obtain a patent, but by means of the trade name he can and does obtain a perpetual monopoly. This, from a business standpoint, is more valuable than the limited monopoly granted by a patent. It is not surprising that proprietary remedies of the latter type flourish so long as physicians unthinkingly accept and prescribe them solely on the manufacturer’s valuation.
“The Council has practically the undivided support of manufacturers of medicinal chemicals; that is, of proprietaries of the first mentioned type. But pharmaceutical firms which have found it profitable to promote proprietaries of the second type—“specialties,” unscientific or ordinary mixtures of pharmaceuticals or biologic products sold under trade names—have not supported the Council.
“When the Council was organized, it was hoped and believed that all the large pharmaceutical houses would find it possible and desirable, if not actually more profitable, to shape their business methods so as to make their proprietary and other articles conform to those conservative standards on which the Council bases its rules, and thus render such articles acceptable for New and Nonofficial Remedies. It soon developed, however, that the methods of the pseudochemical companies, whose sales propaganda in the interest of unscientific nostrums with its attending damage to scientific medicine had led to the establishment of the Council, had found their lodgment in most of the pharmaceutical houses. It was a genuine disappointment to the Council to find that some of the large and old-established firms were not only unwilling to cooperate with the Council, but in many instances exhibited a definite antagonism to the Council’s work.
“The object—and duty—of the officers of pharmaceutical houses is primarily to pay dividends to their stockholders. Through skilful advertising or the persuasiveness of “detail men,” they are able to induce physicians to prescribe their controlled products, on which there are large profits, even though such products have not only not been accepted by the Council, but in many instances, have been disapproved. Is it any wonder that concerns which put out such products are indifferent or openly antagonistic to the work of the Council? The matter is largely one of business policy. When the medical profession as a unit will support the Council in its work, then such firms will find it good business policy to accede to Dr. Bevan’s suggestion—but not before.”
Evidently the problem resolves itself into this: The Council, constituted of scientific men, working without remuneration in the interest of scientific medicine and the medical profession, expects—and rightfully—the cooperation and support of the members of that profession. What is needed, therefore, is the active, sympathetic cooperation of physicians; the cooperation of pharmaceutical houses will follow as a matter of course. (J. A. M. A. 74:1235 [May 1] 1920.)
The following is the recommendation of the Reference Committee to which the Report of the Board of Trustees was referred: “A perusal of the Trustees’ Report, ‘Cooperation of the Pharmaceutical Houses’, is well worth the time of every member of the profession, and your committee would emphasize the statement of the Trustees: ‘The Council, constituted of scientific men, working without remuneration in the interest of scientific medicine and the medical profession expects—and rightfully—the cooperation and support of the members of that profession. What is needed, therefore, is the active sympathetic cooperation of physicians; the cooperation of pharmaceutical houses will follow as a matter of course.’
“Your committee would go still further and move that a vote of thanks of the House be extended to those scientific men who have devoted so much valuable time to the welfare of the Association.”
(J. A. M. A., 74:1322 [May 8] 1920; from Reports of Council on Pharmacy and Chemistry, 1920, p. 56).
W. A. Puckner, Secretary.
BUDWELL’S EMULSION OF COD-LIVER OIL,
NOS. 1 AND 2
Report of the Council on Pharmacy and Chemistry
The Budwell Pharmacal Company, Lynchburg, Virginia, which markets these preparations, claims that “No. 1” contains cod liver oil, “Iodide of Arsenic,” “Iodide of Calcium,” and “Iodide of Manganese.” “No. 2” is said to contain in addition to the ingredients of No. 1, creosote carbonate and guaiacol.
It is known that arsenous iodid is decomposed by contact with water. It is recognized that creosote carbonate is unstable and prone to liberate creosote. Iodide of manganese not being official, the supply on the market is not controlled in any way: Tests of purity are not prescribed by the Pharmacopeia, the National Formulary, New and Nonofficial Remedies or other books of standards. Therefore doubt must be expressed as to the accuracy of the formulas as given. The Council cannot accept such statements of composition without further evidence.
“No. 1” is commended for use in
“Chronic Rheumatism, Glandular Swellings, later forms of Syphilis, convalescence from Scarlet Fever, La Grippe and Malaria, Chronic Malarial Infection, Marasmus, Joint or other suppuration of standing, diseases of skin, chorea, anaemia, neurasthenia, obstinate neuralgia, scrofulous affections in general, and diarrhea or dysentery (subacute or chronic) in childhood.”
“No. 2” is said to be
“Prepared especially for the treatment of Chronic Throat, Nasal, Bronchial and Pulmonary Diseases.”
In the advertising circular statements regarding the various ingredients of Budwell’s Emulsion are quoted from obsolete text books. These statements, for the most part, do not represent modern opinions on the subject. For instance, the circular praises the action of guaiacol as eliminated directly by the lungs, thus exerting a beneficial local effect and causing bacilli to diminish in numbers or to disappear. All of this is directly contradicted in authoritative modern publications on pharmacology, which hold that the excretion of guaiacol by the lungs is infinitesimal and its action on bacilli is nil. The Council held the preparations in conflict with its rules as follows:
1. Many of the therapeutic claims are exaggerations.
2. The method of exploitation amounts to an indirect invitation to the public to use these preparations as “consumption cures.”
3. The preparations are unscientific, they constitute a reprehensible invitation to uncritical prescribing and their use is inimical to the best interests of the profession and the public. It is difficult to imagine in what conditions such a combination would be indicated. These preparations are a remnant of the days of polypharmacy. Their use is not in keeping with present medical thought and practice.—(From The Journal A. M. A., Feb. 20, 1915.)
RHEUMALGINE
Report of the Council on Pharmacy and Chemistry
Rheumalgine (Eli Lilly & Co., Indianapolis) is put up both in tablet form and as a liquid. Each tablet, or teaspoonful of the liquid, is said to contain:
“Strontium salicylate from Natural Oil
5 gr.
Hexamethylenamin
2 gr.
Colchicine
1⁄200gr.”
The advertising matter contains several statements regarding the individual ingredients to which objection must be made.
It is claimed (quoting from Hare) that strontium salicylate
“... is not so disagreeable to the taste as the corresponding sodium salts, and more important still, it is far less apt to disorder the stomach.”
“Taste” is a difficult subject to dispute; but in the experience of the referee, patients object more to the strontium than to the sodium salt. No evidence is submitted to prove that the strontium salt is less apt to disorder the stomach. In observations made under the direction of the referee, the nauseant and emetic doses are about the same as, or even less than, those of sodium salicylate.
Under hexamethylenamin, the recommendations are not confined to its recognized use as a urinary antiseptic; it is also said to be “unexcelled” as a “germicide,” and to prevent the formation of urate and phosphate deposits. These statements are contrary to facts.
“Rheumalgine ... may be used in all cases where the salicylates are indicated. It is superior to preparations containing sodium salicylate, in that it does not cause nausea or disturb the digestion.”
Both the preceding statements are misleading. The necessity of giving 1⁄200 grain of colchicin for each 5 grains of salicylate would certainly interfere with the use of adequate doses of the latter. The colchicin would produce digestive disturbance quite apart from the salicylate.
The mixture is described as:
“... ANTIRHEUMATIC, ANTIPYRETIC, URINARY ANTISEPTIC, AND URIC ACID ELIMINANT. Useful in Acute Articular and Chronic Rheumatism, Muscular Pains, Lumbago, Sciatica, Migraine of the Rheumatic, Gout, and in Nervous Irritability of the Gouty or Lithemic.”
The facts are: Salicylates are useful in some of these conditions, colchicin occasionally in a few, hexamethylenamin in none. The combination is conducive to uncritical prescribing. For instance, salicylates are effective in acute articular rheumatism; hexamethylenamin and colchicin are useless; salicylates are of very little use in chronic rheumatism, sciatica and nervous irritability, while hexamethylenamin and colchicin are useless in these conditions; colchicin is sometimes effective in gout, salicylates perhaps also; hexamethylenamin is not.
Attention should also be called to the high dosage of colchicin, namely, 1⁄100 to 1⁄50 of a grain of the alkaloid, every three or four hours, the dose then to be “slightly reduced,” but continued for several days; or in chronic cases, 1⁄100 to 1⁄30 grain per day, continued indefinitely. This dosage appears high, if a really active preparation is used.
Finally, the name “Rheumalgine” encourages thoughtless and unscientific prescribing. If a mixture is used at all, the prescriber should be constantly reminded of its composition.
It is therefore recommended that Rheumalgine be held in conflict with Rules 6 (unwarranted therapeutic claims), 8 (nondescriptive name) and 10 (unscientific composition).—(From The Journal A. M. A., June 26, 1915.)
GRAY’S GLYCERINE TONIC
Report of the Council on Pharmacy and Chemistry
The Council adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
Gray’s Glycerine Tonic Comp. (Purdue Frederick Company, New York) is a mixture said to be made according to a prescription of the late Dr. John P. Gray, superintendent of the state hospital, Utica, New York. As to the composition, the following statement is furnished by the company:
“This preparation is a combination of Glycerine, Sherry Wine, Gentian, Taraxacum and Phosphoric Acid with carminatives.”
The label declares the presence of 11 per cent. alcohol, and the dose is given at from two teaspoonfuls to a tablespoonful. A study of the ingredients will show that, aside from the alcohol, the mixture contains but one really active drug, gentian. Essentially, then, “Gray’s Glycerine Tonic” is a mixture which, in addition to the narcotic effect of the alcohol, depends on a bitter, gentian, for whatever therapeutic action it may possess.
The bitters, of which gentian is a type, were once credited with many therapeutic virtues which time has shown they do not possess. Pharmacologic research has demonstrated that their utility consists in stimulating the appetite through their action on the taste buds. On this account they were believed also to increase the secretion of the gastric juice by a psychic impression. More recently, however, even this has been questioned—by Carlson, for instance.
These facts are fully understood, presumably, by all physicians. Yet, according to the advertising circular, this “tonic,” which, for all practical purposes, is merely a simple bitter, is good for thirty-two diseases ranging from amenorrhea to whooping cough!
The conditions in which Gray’s Glycerine Tonic is asserted to be especially efficient are described on the label of the bottle and the outside wrapper, in popular terms, more or less typical of “patent medicine” exploitation, such as “catarrhal conditions,” and “stomach derangements.” Similar statements are contained in the leaflet accompanying the trade package. For instance:
“It is, therefore, an effective, reliable tonic in nervous exhaustion, general debility, impoverished conditions of the blood and nervous system, Bright’s disease, diseases of the liver, disorders of the urinary organs, etc.”
“It is an unexcelled restorative in that very common class of cases in which there is no positive organic disease, but the patient complains that he ‘does not feel well’ or ‘is out of sorts.’ ”
Here are some of the claims made in other advertising matter:
“All stages of bronchitis ... are rapidly improved by the use of Gray’s Glycerine Tonic Comp. This remedy has a direct tonic influence upon the circulation of the respiratory mucous membrane; it relieves congestion and restores tone to weakened blood vessels.”
“... improves the appetite, gives valuable aid to the digestive and absorptive processes, and reinforces cellular nutrition in ways that insure a notable gain in vitality and strength.”

This appeared in a journal owned and controlled by the second largest state medical association of the country.
Even granting that gentian may improve the appetite, how absurd it is to claim that this mixture “relieves congestion,” “restores tone to weakened blood vessels,” “gives aid to the absorptive processes,” “reinforces cellular nutrition,” or increases vitality!
Neither the composition of Gray’s Glycerine Tonic nor the clinical evidence warrants the belief that it has any therapeutic value other than that due to the psychic effect of the bitter drug gentian. Physicians who have prescribed it have done so because of the advertising. This nostrum has been kept so constantly before the eyes of medical men that they think of Gray’s Glycerine Tonic when they cannot remember the official drugs that may be indicated in the case. The moral is that liberal advertising will sell anything.
It is recommended that Gray’s Glycerine Tonic Comp. be declared not eligible for inclusion in New and Nonofficial Remedies on account of conflict with Rules 1, 6, 8 and 10.
[Editorial Note.—An old practice in hospitals—happily now practically obsolete—was to have certain stock mixtures prepared in bulk. Among these there was usually a so-called tonic mixture, used in a more or less haphazard manner when nothing in particular seemed indicated. Such a stock mixture was used in the State Hospital for the Insane at Utica, N. Y., during the many years that Dr. John P. Gray was superintendent (from the early fifties to the early eighties), although it is very doubtful whether he originated the mixture. After the death of Dr. Gray—so the story runs—one of his sons, with a partner, formed the firm of Purdue Frederick Company, and began the exploitation of the elder Dr. Gray’s name, in connection, presumably, with this stock preparation. As indicated in the Council’s report, Gray’s Glycerine Tonic Comp.—and what an absurd name!—is simply a mixture of ordinary drugs, requiring no skill whatever in compounding. If there is a physician living who cannot write a prescription offhand as good as this formula, that physician should either go back to a medical school or change his vocation. There is, and can be, no excuse for prescribing such a ready-made mixture, for every cross-roads drugstore has the ingredients and any pharmacist worthy of the name could compound it. Among the scores of nostrums that disgrace the medical profession of this country, none is more typical of all that is inimical to scientific medicine, to the medical profession and above all to the public—for, after all is said, it is the public that ultimately is humbugged.]—(From The Journal A. M. A., July 10, 1915.)
TONGALINE AND PONCA COMPOUND
Report of the Council on Pharmacy and Chemistry
The Council, having considered “Tongaline,” “Tongaline Tablets,” “Tongaline and Lithia Tablets,” “Tongaline and Quinine Tablets” and “Ponca Compound Tablets,” found these preparations ineligible for New and Nonofficial Remedies and authorized publication of the following report.
W. A. Puckner, Secretary.
TONGALINE
Tongaline (Mellier Drug Co., St. Louis) is a fancy name given to what is essentially a sodium salicylate mixture. The air of mystery created by the name permits the manufacturers to make claims for the product which would be ludicrous if the medical profession was fully conversant with the very ordinary character of the preparation.
Tongaline receives its name from tonga, an inert, long-discarded mixture of various barks and herbs said to be gathered and prepared by Fiji Islanders. Its constituents evidently tend to vary with the collector. The history of the introduction of this indefinite combination of simples is thus given in The Journal, May 10, 1913.
“A supply of the crude drug was carried to England by a man who had lived for a short time in the Fiji Islands and it was placed in the hands of a retail house in London. This occurred about 1879. In 1880, two English physicians of repute published laudatory articles on the therapeutic value of tonga in neuralgia and rheumatism. This created a demand for the drug which extended to the United States.”
Time showed that tonga was inert therapeutically, and authorities on pharmacology now no longer notice it. As the Council previously reported,12 the indefinite character of the mixture should, alone, be sufficient to exclude it from practical therapeutics. During the temporary popularity of tonga, the proprietary mixture Tongaline was put on the market for physicians’ use by the Mellier Drug Company, St. Louis. In this, tonga was named as the active ingredient. The commercial interests thus involved have faithfully nourished and kept alive the “tonga” myth.
In a recent advertising booklet, “The Therapeutic Properties of the Ingredients of Tongaline,” the virtues of tonga, blue cohosh, colchicum, jaborandi and salicylic acid are discussed. The label of a recently purchased bottle reads:
“Tongaline contains Tonga, Cimicifuga Racemosa, Salicylate of Sodium (the salicylic acid being made from pure natural oil) Colchicum and Pilocarpin.”
It will be noticed that Tongaline is “made from the pure, natural oil.” In fact, the statement is repeated in red ink, in large letters running across the face of the label, thus emphasizing the alleged importance of this assertion. In this connection it is only necessary to recall that it has been proved clinically, chemically and physiologically that there is absolutely no difference between the salicylic acid made from the natural oil and the synthetic.
The formula was thus commented on in the article previously quoted from The Journal:
“Tongaline ... is essentially a preparation of sodium salicylate,... The Mellier Drug Company realized the impossibility of creating any marked demand for a nostrum unless it had some real drugs in it—hence the presence of the salicylates. What the actual composition of Tongaline is, no one but the manufacturers know. At one time the following was given as the formula:
Fluid Tonga
30 grains
Extract of Cimicifuga Racemosa
20 grains
Sodium Salicylate
10 grains
Pilocarpin Salicylate
1⁄100grain
sColchin Salicylate
1⁄500grain
s“These amounts refer to the quantity of drugs in each fluidram of the preparation. Whether the nostrum still has this composition we do not know, but assuming that it has, it is quite evident that sodium salicylate is the essential and active ingredient.”
The therapeutic indications given on the label of the bottle are:
“Rheumatism, Neuralgia, Grippe, Gout, Nervous Headache, Sciatica, Lumbago, Malaria, Tonsillitis, Heavy Colds, Excess of Uric Acid, and wherever the use of the Salicylates is indicated.”
In a recent booklet this semisecret salicylate mixture is recommended, not only in conditions in which salicylates are indicated, but also combined with aconite for rheumatic fever, with benzoate of soda in the treatment of “grippe,” with potassium bromid in nervous headaches, with gelsemium, glycerin and whisky for “heavy colds,” with ammonium chlorid, stramonium and cimicifuga in “rheumatic dysmenorrhea,” and even with mercury biniodid as a treatment of syphilitic eruptions!
“When administered with good judgment, Tongaline exerts a stimulating effect upon every organ of elimination; cleansing the complex sewerage system and putting it into working order. When this is done, the sluggish blood current begins to flow more freely; the lymphatic and glandular systems to give up and carry off the toxic products, so long retained ...”
TONGALINE TABLETS
Then because of a “desire to put Tongaline in a more compact and convenient form,” the same concern puts on the market Tongaline Tablets. Whether Tongaline Tablets are of the same composition, the doctor who prescribes them is not advised. In this form we have Tongaline and Lithia Tablets, and Tongaline and Quinin Tablets. Presumably those who are attracted by the word “lithia” are sufficiently uncritical to be content with the statement that:
“The addition of Lithia to Tongaline presents a most useful combination which does not rely upon its action on the kidneys alone as is the case with Lithia salts or Lithia waters as administered ...”
And the foregoing quotation, be it remembered, is for the information of the medical profession! Tongaline and Lithia Tablets, we are informed, are:
“... particularly indicated for certain diseases which are caused by deposits of urates in the joints and kidneys, and can be used with much benefit for many people who indulge in generous or intemperate habits of living.”
Tongaline and Quinine Tablets are also exploited without statement of composition. The promoters are probably justified in feeling that physicians who prescribe quinin in combination with “Tongaline” care little about the dosage.
It is unnecessary to discuss the silly claims made for Tongaline and its combinations, although it is worth while to point out that the prescribing of such nostrums by physicians is an imposition, if not a fraud, on the public.
PONCA COMPOUND
Ponca Compound, also made by the Mellier Drug Company, St. Louis, is a “female weakness remedy” in tablet form. The name suggests that “ponca” is a medicinal substance, and, in fact, at one time, “Ext. Ponca” was named as an ingredient. The nature of “Ext. Ponca” was apparently never explained. It is now replaced in the “formula” by “senecin,” and the only information concerning the composition at present given is:
“Ponca Compound Tablets Contain Extract of Mitchella Repens, Senecin, Helonin, Caulophyllin and Viburnin.”
This “formula” is practically meaningless, not only because the amount of each ingredient is not stated, but also because “senecin,” “helonin,” “caulophyllin” and “viburnin” are in themselves variable mixtures of unknown composition.13
Presumably, “senecin,” “helonin,” “caulophyllin” and “viburnin” are extractives of some kind prepared, respectively, from senecio aureus (life root), helonias dioica (false unicorn), calophyllum thalictroides (blue cohosh) and viburnum prunifolium or opulus (black haw or cramp bark). These are, one and all, practically inert drugs. There is no reason to believe that any or all of them can have any beneficial influence in the many and varied conditions for which Ponca Compound is advertised.
The following are excerpts from the advertising matter:
“Ponca Compound is a remedy of a very beneficial character for Functional, Uterine and Ovarian troubles, which will respond to internal treatment, especially when digital examination or surgical interference is undesirable.”
“Ponca Compound is also valuable during gestation and after parturition.”
“Uterine Alterative for Leucorrhoea, Dysmenorrhoea, Amenorrhoea, Metritis, Endometritis, Menorrhagia, Metrorrhagia, Irregular Menstruation, Subinvolution, Painful Pregnancy.”
It is recommended that Tongaline and Ponca Compound and all their preparations be held in conflict with Rule 1, in view of their semisecret and indefinite composition; with Rule 6, for the grossly exaggerated therapeutic claims made for them; with Rule 8, because of their misleading names, and with Rule 10, in view of their unscientific character as irrational combinations. It is also recommended that this report be published.—(From The Journal A. M. A., July 17, 1915.)
ALFATONE
Report of the Council on Pharmacy and Chemistry
The Council has found Alfatone ineligible for New and Nonofficial Remedies and has authorized publication of the following report.
W. A. Puckner, Secretary.
Alfalfa is good cattle feed but only nostrum exploiters have suggested its use as a medicine for human beings. While it may seem a waste of time to discuss the medicinal value of alfalfa its recent exploitation by the Norwich Pharmacal Company, Norwich, N. Y., as “a reconstructive tonic and nutrient” in the form of a mixture called “Alfatone,” calls for comment. According to the label on the preparation:
“Each fluidounce represents:
Alcohol
15
per cent.
Medicago sativa (Alfalfa)
120
grains
Taraxacum
2
1⁄2grains
Gentian
1
grain
Berberine Hydrochloride
1⁄40grain
Glycerin and Aromatics.”“Dose.—One to three fluidrams (4 to 12 c.c.) 4 times daily.”
Each maximum dose, therefore, should represent 45 grains of alfalfa, 1 grain of taraxacum (dandelion), 3⁄8 grain of gentian, 1⁄100 grain of berberin hydrochlorid, and 27 minims of alcohol. Since the bitter drugs are present in such small amounts that the preparation is almost devoid of bitterness, and as the medicinal value of alfalfa is practically nil, it is evident that whatever action Alfatone may have is due to the stimulant effects of the alcohol.
Some of the claims made for Alfatone are:
12 Reports of the Council on Pharm. and Chem., 1912, p. 40.
13 See Report of the Council on Pharmacy and Chemistry on “Resinoids and Concentrations,” J. A. M. A., Nov. 13, 1909, p. 1655.
“A reconstructive nutritive tonic indicated in general debility, neurasthenia, convalescence, etc.”
“... a Galactagogue of merit as well.”
“... improves the appetite, aids the processes of digestion and assimilation, facilitates elimination and effects gradual but decided gains in strength, vitality and weight.”
It is suggested that:
“... in case of idiosyncrasy the addition of Tr. Nux Vomica 5 to 10 minims to the dose, unless contraindicated, will secure excellent results.”
The Norwich Pharmacal Company naively remarks:
“The dearth of medical literature on Alfalfa has lead us to present below a few of the findings of the Bureau of Plant Industry of the Department of Agriculture ... as well as those from several state experiment stations ...”
Here are the “findings”:
“... Digestible nutrients in 100 pounds of Alfalfa,... Protein, 11.0 pounds; Carbohydrates, 39.6 pounds; Ether Extract, 1.2 pounds.”
“... The high value of Alfalfa is due to the amount of protein that it contains; to the large percentage of protein that is digestible and the palatability of Alfalfa.”
“... Table showing pounds of elements removed from the soil by one ton of crop.
Alfalfa
Wheat
Potash
49.79
12.52
Phosphoric Acid
8.27
9.08
Lime
43.51
2.95
Nitrogen
44.01
22.30”“... The abundance of muscle and bone producing material in Alfalfa makes this crop especially good.”
Thus estimates of the value of a farm crop and cattle fodder are made to do service as testimonials to its therapeutic merit for human beings! Has the “patent medicine” promoter ever dared to insult the intelligence of his patrons by a cruder absurdity? Yet it is not to the nontechnical and unscientific public, but to a profession presumably scientifically trained in pharmacology and therapeutics that this concern presumes to offer its fodder tincture on the basis of testimony to the agricultural value of the fodder plant.
Alfatone is a worthless alcoholic cordial. The audacity of the attempt to promote its sale by a discourse on the merits of a well-known fodder plant is the sole reason for devoting any attention to it. It is recommended that Alfatone be held ineligible for New and Nonofficial Remedies, and that this report be published.
[Editorial Note.—What a comment on American medicine that a concern can even contemplate the possibility of making a commercial success of the sale of such a silly nostrum as Alfatone! And yet, when one remembers that a proprietary in which oats constitutes one ingredient (“Pas-Avena”) for years has been advertised to physicians and presumably prescribed by them, it is not altogether inexplicable that business men should get the impression that the medical profession is “easy” enough to “fall for” anything in the line of proprietary mixtures. Perhaps we may look forward to being offered proprietaries based on other cheap and well-known fodder plants. Tincture of Timothy Hay, Blue Grass Tonic, Cornhusk Wine! Why not? The enterprising companies that may put them out can easily publish tables to show the digestible nutrients in each and indubitable testimony can be furnished to prove the excellence of any of them as stock feed. If a pitchforkful of timothy hay makes a good fattening ration for a growing steer why should not a teaspoonful of tincture of timothy hay make a “reconstructive tonic and nutrient” dose for a man? If an arm load of thistles (carduus) makes a luscious food for equus asinus why should not a pinch of thistle in alcohol and water be a good “tonic”? Great are the possibilities! They are limited only by the gullibility of the medical profession and the public. Certain it is that some proprietary manufacturers are firmly convinced that no combination can be too preposterous to be worth trying on the medical profession.]—(From The Journal A. M. A., Aug. 7, 1915.)
ARTICLES REFUSED RECOGNITION
Report of the Council on Pharmacy and Chemistry
Below appear abstracts of the Council’s actions on articles refused recognition which were not deemed of sufficient importance to require lengthy reports.
Uricsol
Uricsol is marketed by the Uricsol Chemical Company, formerly of Los Angeles, now of Boston. Regarding its composition only vague statements are made. In an advertising pamphlet it is promised that the formula will be sent to physicians on request. Such a request from a physician elicited the following statement:
“URICSOL is a non-irritating, alkaline solution, containing Lithium Citrate, Acid Citric and Potassium Nitrate, together with a saline laxative in the form of Glycero Sodium Phosphate, with Vegetable Tonics added.”
The Association Laboratory has made an examination of Uricsol to determine its composition and reports as follows:
LABORATORY REPORT
A trade package purchased in March, 1915, from a wholesale drug house was labeled:
“Uricsol Rheumatic Remedy, Uric Acid Solvent, Kidney and Liver Stimulant, Manufactured by the Uricsol Chemical Co., Los Angeles, Cal.”
This package was wrapped in a circular entitled “The Great California Remedy—Uricsol.” The preparation is a viscid, slightly turbid light brown liquid, with a faintly aromatic odor and a salty, bitter taste. The diluted solution is acid in reaction toward litmus and phenolphthalein and alkaline toward methyl orange.
Qualitative tests showed a presence of phosphate, citrate, nitrate, sodium, glycerin, and a small amount of lithium in aqueous solution. Besides these a small amount of some organic, nonalkaloidal substance was found, which from its bitter taste suggested gentian. From the qualitative tests it appeared that the phosphate was the predominating ingredient and accordingly a phosphate determination was made. The results, calculated to sodium phosphate, U. S. P., indicated the presence of 64.20 gm. per 100 c.c., held in solution by citric acid and sodium nitrate.
Uricsol evidently is a solution containing a large amount of sodium phosphate with small amounts of lithium, nitrate, citric acid and glycerin, with probably some vegetable extract.
In general Uricsol is similar to the once widely exploited proprietary “Melachol,” which has been frequently imitated. A preparation essentially identical is in the United States Pharmacopeia, under the title “Compound Solution of Sodium Phosphate.”
The Uricsol Company calls its preparation
“... the latest word in the treatment of Rheumatism and that allied group of ailments which is caused by an excess of Uric Acid.”
Hay fever, bronchial asthma and neuritis are conditions in which it is recommended. The claim is made that
“Uricsol quickly controls Vasomotor Rhinitis and eliminates such conditions from the system.” “In fact, it will correct FAULTY METABOLISM.”
To a few practitioners of an older generation the pharmacologic basis of a remedy for rheumatism was sufficiently defined by saying that it increased the solubility of uric acid or affected it in some way. This theory is obsolete; there is not, and never was, any reliable evidence on which to base the theory that rheumatism is in any way caused by uric acid. The exploitation of Uricsol as a “uric acid solvent” is merely another illustration of the way in which nostrum manufacturers play on disproved theories. Of course the claim that sodium phosphate has any particular power to control vasomotor rhinitis, hay fever, asthma, and to correct faulty metabolism is foolish.
To summarize: Uricsol is a mixture of well-known drugs, marketed with false claims as to therapeutic action, with misleading and meaningless statements as to composition and under a name which invites uncritical prescribing. Uricsol is held ineligible to inclusion in New and Nonofficial Remedies.
Jubol
The following ridiculous statements are addressed, not to the laity, but to the medical profession:
DO YOU SUFFER FROM Constipation—Hemorrhoids—Enteritis—Mucous discharge—Pituita—Acidity of the stomach—Vertigo—Sick Headache—Disturbed Sleep—Insomnia—Sallow Complexion—Coated Tongue—Offensive breath—Fatigue and depression—Boils—Pimples?
“ONE of these symptoms alone shows that there is defective or insufficient function of the intestines, even if the stools are regular.
“Excrements remain too long in the intestine and set up fermentation. The harmful poisons and Ptomains which they produce are re-absorbed by the blood and poison the whole system.
“The Intestines must be cleared and re-educated with JUBOL.
“Jubolise your Intestines.”
Jubol tablets are sold in the United States by Geo. J. Wallau, Inc., New York, and are said to be prepared by J. L. Chatelain, Paris, France. The following incomplete and nonquantitative “formula” is furnished:
“... compounded chiefly [!] of Agar-Agar, Biliary Extracts and pure Extracts from all the intestinal Glands.”
It is asserted that
“The tablets are coated with a protective covering in order that they may act on the intestine only.”
The tablets contained in a regular-size trade package, obtained direct from the agent, readily separated into two halves and disintegrated within a few minutes when agitated with water. It is thus evident that, under ordinary conditions, the intestinal ferments in Jubol (if they are present, as claimed) would be destroyed during their passage through the stomach. In direct tests, however, practically no tryptic activity was demonstrated.
The composition of Jubol is not declared; grossly unwarranted and incorrect claims are made for its therapeutic actions; the name does not indicate the alleged ingredients and so much of the composition as is declared indicates an unscientific mixture. The Council decided that Jubol should be held ineligible for New and Nonofficial Remedies, and that this report should be published.
Urodonal
Urodonal is said to be “produced in the laboratory of J. L. Chatelain,” Paris, France. It is marketed in this country by Geo. J. Wallau, Inc., New York.
The preparation is claimed to be a chemical compound, and the advertising matter furnishes a “formula,” which consists of the formulas of lysidin, sidonal and hexamethylenamin, connected by plus signs:
That the substance is a chemical compound is highly improbable, and no evidence has been submitted to substantiate the claim. On the contrary, in the following statement the phrase “based on” is a virtual admission that the preparation is merely a mixture:
“Urodonal ... is a granular effervescent preparation based on methylglyoxalidine [Lysidine], quinate of diethylene-diamine [Sidonal] and hexamethylene-tetramine [Formin, urotropine].”
Mystery is added by the mention of undefined “special products” in the following:
“The fact of combining these two salts [lysidin and sidonal] in Urodonal, in strictly determined proportions and in the presence of special products, gives this preparation very considerable power in dissolving uric acid.”
These contradictory statements of composition conflict with Rule 1.
Urodonal is marketed in typical “patent medicine” style: the name “Urodonal” is blown in the bottle and the label contains a list of “Indications,” including rheumatism, gout and gravel (Rule 4). That this form of marketing has introduced it to the public is suggested by the following in an advertising circular:
“... Urodonal is now popular—even classic—throughout the world, where thousands of doctors and millions of patients agree in asserting that ‘Urodonal is to rheumatism what quinine is to fever.’ ”
There are also other indications that the mixture is to be exploited to the laity. For instance, the U. S. distributor sends out a portrait of Sarah Bernhardt bearing the legend:
“I am positive that URODONAL preserves youth’s freshness with clearness and strength to brain and heart. I have taken it for two years with the greatest benefit. Sarah Bernhardt.”
A circular advises this mixture
“For all who suffer from Arthritis, Rheumatism, Arterio-Sclerosis, Renal and Bilious Lithiasis, Headache, Gout, Gravel, Lumbago, Sciatic Pains, Neuralgia and all uric acid troubles.”
“In fact, Urodonal is five times more active than piperazine, and thirty-seven times more active than lithia. We are, therefore, entitled to say that no other eliminator of uric acid can be compared with it.”
“Being 37 times more active than lithia, it clears the heart valves of any sandy substances which may clog them, and checks the atheromatous degeneration of the blood vessels.”
These extracts indicate sufficiently the extravagant tone of the advertising (Rule 6): None of the ingredients are notably active in dissolving uric acid when administered by mouth. None produce any marked increase of uric acid elimination. No intelligent physician would use a uric acid solvent for “bilious lithiasis”; and their usefulness in the other conditions is open to doubt, to put it mildly.
Although the preparation is a simple mixture, the name does not indicate the components, but inclines to therapeutic suggestion (Rule 8).
Nothing is to be gained by combining several drugs which are useless, severally, for the purpose intended, as in the present case (Rule 10).
Urodonal is marketed under inconsistent statements of composition and with exaggerated therapeutic claims; the name is nondescriptive and the mixture is unscientific. The Council decided that the preparation should be declared ineligible for conflict with Rules 1, 4, 6, 8 and 10 and that this report should be published.—(From The Journal A. M. A., Aug. 14, 1915.)
FORMAMINT
Report of the Council on Pharmacy and Chemistry
The following report has been authorized for publication.
W. A. Puckner, Secretary.
Formamint is a proprietary medicine manufactured by the A. Wulfing Company (New York, London and Berlin), which is affiliated with the Bauer Chemical Company.
It has been widely advertised in Europe for several years, and is now on the American market;14 it is advertised in this country both in newspapers and medical journals.
Following is a brief review of the more important alleged investigations that have been reported from time to time in various European journals.
In “The Therapeutical Value of Foramint in Septic Affections of the Oro-Pharynx,” De Santi15 quotes Rosenberg,16 who reports the successful use of Formamint in cases of streptococcus infections, tonsillitis and acute symptoms of chronic sore throat. According to Seifert,17 Formamint is a chemical combination of formaldehyd and milk sugar. When the tablets are dissolved in the saliva, 0.01 per cent. of formaldehyd in its “status nascendi” is liberated and exercises a strong disinfectant action. Seifert states that the preparation is markedly palatable, since it contains a little citric acid to render the taste cool and refreshing. In some experiments with streptococci, pneumococci, typhoid and diphtheria bacilli, Seifert found that a solution of one tablet in 10 c.c. of water destroyed these germs in from five to ten minutes. A solution of the same strength was also added to culture tubes of broth, agar, and gelatin, with the result that no growth occurred in them, while distinct and characteristic development of the bacteria took place in control tubes. He does not state, however, how much Formamint solution was added to the mediums.
Daus18 reports successful treatment of tonsillitis, mumps and middle ear diseases. In these cases no other gargles or mouth washes were used. He states that no indication of irritant or other injurious action made its appearance even after large doses. In the same article, F. Levy reports experiments as follows: Agar plates were prepared with a culture of streptococcus from a severe case of quinsy. One half of the plate was rubbed with saliva containing Formamint in solution. (The strength of the solution used is not given.) In twenty-four hours streaks of growth had appeared on one portion of the plates while the part on which the Formamint saliva had been rubbed remained sterile. Daus also found that agar and broth cultures of streptococcus shaken with Formamint saliva remained sterile.
Rheinboldt,19 investigating the effects of Formamint and of ordinary formaldehyd on animals, concludes that formaldehyd is toxic in action while Formamint is not.

How the exploiters of Formamint capitalize the medical profession. Miniature reproductions of typical Formamint advertisements appearing in the newspapers.
Rosenberg20 corroborates this statement. He also found that agar plates of Bacillus prodigiosus were killed by Formamint solutions in about four hours. He fails, however, to give the strength of his Formamint solutions.
Wingrave21 suggests the use of Formamint for infants! He recommends that a tablet be crushed and wrapped in “butter cloth.” The ends of the cloth are to be tied with thread, the Formamint is to be moistened, and the packet is to be held in the mouth of the baby several times each day.
Young22 published the results of some experiments by himself and Delépine on the human throat. They dissolved a tablet in the mouth and made swab cultures with the following results:
Immediately after taking the tablet
0 colonies
10 minutes after taking the tablet
35 colonies
30 minutes after taking the tablet
150 colonies
They found no staphylococci at any time. Other results of swabbing various parts of the throat before and after the use of Formamint, reported by these investigators, show enormous reductions in the count, claimed to be due to the action of Formamint. The count was made on agar at 37 C., but they fail to state the time elapsing between taking the Formamint and making the swab. Young also reports favorable clinical results in cases of scarlet fever, diphtheria, sore throat, and the like. It must be noted, however, that they state that the mouth and fauces must first be thoroughly cleansed by swabbing and douching before Formamint is used.
THE “CHEMICAL COMPOUND” CLAIM
The claims made in the advertising literature of Formamint are very extravagant. Many are highly improbable. These statements will be discussed later.
The statement is made that Formamint is a new chemical compound:
“Formamint is Pentamethanallactose, 5 CHOH + C12H22O11. It is an original combination of Formaldehyde with Lactose, a definite chemical compound. The Formaldehyde molecule is locked up in it until solution in the saliva takes place, when the Formaldehyde is liberated in its nascent state and is therefore active without being irritant.”
Furthermore the makers contend that this new chemical compound is entirely harmless. For example, Daus,18 in an article on “The Disinfectant Action of Formic Aldehyde on Mucous Membranes,” declares:
“No indication of irritant or other injurious action made its appearance even after large doses. The urine remained free from albumin and sugar.”
Such statements as these are found in the advertising literature:
“Formamint tablets are absolutely harmless and innocuous, even to little children.”
“When dissolved in the saliva, Formamint Tablets liberate slowly Nascent Formaldehyde in a most active yet non-irritant form.”
They maintain that Formamint is not only absolutely harmless, but actually beneficial to the tissues. It may be used “to tone up and strengthen the tissues, prevent hoarseness, and allay irritation in singers, public speakers,” etc.
The claims urged as to its germicidal power are indeed glittering. This “new chemical compound” is claimed to liberate formaldehyd in some new and peculiar condition which, while it has a soothing and tonic effect on the cells of the human tissues, can at the same time quickly kill any form of bacterial life.
“Dissolving readily, it releases its germicidal, antiseptic qualities, which impregnate the saliva and are carried naturally and easily around the mouth and in the deepest crevices of the throat—destroying the germs where they are causing the mischief. Formamint prevents and destroys infectious germ life in a soothing grateful way.”
“In the saliva it frees a germicide, fatal to germs but harmless to the most delicate membranes. And flowing into every tiny corner of the gums, tonsils and throat, into places where no gargle ever reaches, it most effectively disinfects the throat.”
The claims as to the preventive and curative effects of the preparation cover a large portion of the category of human ailments and distresses. The following quotations indicate some of its supposed properties:
“... it is therefore self-evident that Formamint should be looked upon as a necessary part of the treatment of all forms of tonsillitis.”
“The value of Formamint is equally great in diphtheric tonsillitis, or as a prophylactic ...”
“The extraordinary success which I had with Formamint in a school epidemic of scarlet fever during May and June, 1907, was the determining factor which induced me to abandon the use of inhalations, gargles, local applications in the treatment of diseases of the throat, and to use Formamint exclusively for the future.”
“There are naturally many similar conditions in which Formamint may be used as a prophylactic, notably scarlet fever, mumps, streptococcal and staphylococcal sore throats, ‘milk outbreaks’ of sore throat, drain throats, hospital throats, and the like.”
“Formamint Tablets are indicated in Angina, Tonsillitis, Pharyngitis, Stomatitis, Gingivitis, Glossitis, ulceration, spongy or bleeding gums, Pyorrhea Alveolaris, ‘Smoker’s Sore Throat,’ Abscess or Boils, etc.”
“As a Prophylactic against Diphtheria, Scarlet Fever, Influenza, Measles, Epidemic poliomyelitis, and other pathogenic micro-organisms. To neutralize putrefaction products in and about the teeth, correct fermentative processes, deodorize and purify the breath, etc.”
“To tone up, and strengthen the tissues, prevent hoarseness and allay irritation in singers, public speakers, neutralize the effects of dust-infection or disinfect the saliva or sputum in Influenza, Tuberculosis, etc.”
One man declares that along with specific constitutional treatment he “had the best results from the use of Formamint tablets” in a case of syphilitic ulceration of the tongue.
In short, Formamint is recommended for the treatment or prevention of almost everything, from a bad breath to such grave conditions as scarlet fever, diphtheria and tuberculosis, conditions in which a delay in proper treatment—for instance, in diphtheria, a failure to administer antitoxin—may result in the death of the patient.
A series of investigations was therefore undertaken in order to discover whether the extravagant claims regarding the germicidal power of Formamint could be verified.
Experimental Data
Two fifty-cent bottles of Wulfing’s Formamint were purchased in the open market and were kept well stoppered to prevent deterioration.
Qualitative tests showed the presence of formaldehyd and the amount was determined quantitatively by the hydrogen peroxid method as given by Sutton.23 The results were respectively, 1.99 per cent. and 2.03 per cent. of formaldehyd.

Two Formamint advertisements reproduced in miniature typical of those appearing in a certain type of medical journals.
Some determinations were made of the germicidal power of Formamint in vitro, that is, under controlled laboratory conditions. A twenty-four-hour plain agar culture of Staphylococcus aureus was washed off in 10 c.c. of sterile 0.85 per cent. sodium chlorid solution. A 1:100,000 dilution of this was made in each of three flasks containing 100 c.c. of sterile saliva. Flask 1 contained 1 per cent. of Formamint, Flask 2, 5 per cent.; Flask 3, containing no Formamint, was kept as a control. At intervals samples were removed and dilutions made and plated in duplicate on standard agar. The plates were incubated twenty-four hours at 37 C., and plates containing less than 200 colonies were counted. The results are given in Table 1. After seven days there was no appreciable difference in the plates.
TABLE 1.—SHOWING TIME IN WHICH CULTURES OF STAPHYLOCOCCUS AUREUS WERE KILLED
BY DIFFERENT AMOUNTS OF FORMAMINT
Amount of Formamint in Saliva (Per Cent.)
Period of Standing at 37 C. (Hours)
Average Count When Plated
Count on Flask of Saliva Without Formamint
1
3
32
3200
*1
6
0
7000
*5
1
Few
5000
*5
2
0
4100
*5
3
0
3200*
5
6
0
7000*
* The last two observations were made at the same time as on the 1 per cent. solutions.
Another test was made by adding a 1 per cent. Formamint solution to plain agar plates inoculated with B. coli. A twenty-four-hour plain agar culture of B. coli was washed off in 10 c.c. of sterile 0.85 per cent. sodium chlorid solution. A 1:1,000,000 dilution was made of this and 1 c.c. added to each plate. Varying amounts of 1 per cent. solution of Formamint were added to each plate. They were incubated seventy-two hours at 37 C. After seven days’ incubation the count was the same. The results are given in Table 2.
TABLE 2.—COUNT OF B. COLI CULTURES WITH DIFFERENT AMOUNTS OF FORMAMINT
No. c.c. of 1 per cent. Formamint
0
0.1
0.3
0.5
0.7
1.0
1.5
2.0
3.0
Count
160
33
39
26
15
12
2
0
0
Another experiment was made thus: One loopful of a twenty-four-hour plain agar culture of Streptococcus lacticus was mixed with a tube of North medium. One loopful from the inoculated tube was mixed with a second tube of North medium. Both tubes were poured into Petri dishes and allowed to cool. One half of each plate was well smeared with a 10 per cent. solution of Formamint in saliva. After twenty-four hours’ incubation at 37 C., only a few colonies appeared on the side to which the Formamint had been applied, while the other half was thickly covered with colonies.
This work so far corroborates that reported in the literature quoted by the manufacturers. But the fact that a compound is a germicide when brought into intimate contact with bacteria in a solution or medium in a test tube or flask does not prove that it will be effective when used in the human throat.
THE ALLEGED GERMICIDAL ACTION
An attempt was made to discover whether or not the claims advanced by the manufacturers as to the perfect germicidal action of Formamint in all the nooks and crannies of the mouth and throat could be confirmed.
The first step in attacking this problem was to make comparative counts of the number of bacteria in the throat before and after the use of Formamint. The methods employed were as follows: The throat was gargled with 50 c.c. of sterile 0.85 per cent. sodium chlorid solution. In each case the same length of time, as far as possible, was used in the process. The liquid was collected in a sterile flask. The gargling in a series of experiments was begun not less than two hours after a meal. After some preliminary work the following dilutions of the 50 c.c. of salt solution were found sufficient: 1:1,000, 1:10,000 and 1:100,000. Plates were made in duplicate from each dilution and incubated seventy-two hours at 37 C. The counts were made on plates containing less than 200 colonies. Except where otherwise noted standard agar was used. The mediums were always prepared in the same way.
All the work was carried out under conditions as nearly natural as possible. The Formamint was taken according to the directions accompanying the trade package. Every opportunity was given the Formamint to penetrate all the crypts and recesses about the mouth and throat. The tablet was allowed to dissolve as slowly as possible, the time usually being five to six minutes, and saliva was thoroughly forced around the mouth before being swallowed. Plating was always done immediately after gargling so that no growth could occur in the salt solution. The results are given in Table 3. The numbers are average counts from several plates and calculated to show the number of bacteria washed out by the 50 c.c. of salt solution.
TABLE 3.—SHOWING THAT FORMAMINT DOES NOT GREATLY DECREASE THE NUMBER
OF BACTERIA IN THE THROAT
Conditions of Test
Time Since Preceding Test Amount of Formamint Used No. Found in Throat Before Use of Formamint No. Found in Throat After Use of FormamintNormal
...
0
15,600,000
...
Normal
1 hour
0
38,500,000
...
Normal
1 hour
0
30,500,000
...
Normal
...
0
12,500,000
...
Normal
1 hour
0
14,500,000
...
1 hour
0
23,500,000
...
Tablet dissolved in mouth and throat gargled one hour later
6 days
1 tablet
...
15,000,000
Throat again gargled two hours after Formamint was used
1 hour
0
...
10,050,000
Normal
7 days
0
62,000,000
...
Normal
1 hour
0
72,500,000
...
Normal
...
...
61,000,000
...
Tablets were taken, one per hour, and throat gargled one hour after last tablet was taken
2 days
12
...
39,100,000
Throat was again gargled 2 hours after taking last tablet
1 hour
0
...
59,000,000
Normal
5 days
0
35,000,000
...
Normal
1 hour
0
62,000,000
...
Normal
1 hour
0
72,000,000
...
One tablet was taken each half hour for twelve hours consecutively. Throat was gargled one hour after last tablet was taken
4 days
24 tablets
...
175,000,000
Throat was again gargled two hours after last tablet was taken
1 hour
0
...
168,750,000
Normal
3 days
0
129,600,000
...
Normal
1 hour
0
177,000,000
...
Normal
1 hour
0
147,000,000
...
Normal
3 days
0
79,000,000
...
One tablet was taken immediately after preceding gargle. Throat was again gargled at end of one hour
1 hour
1
...
83,200,000
Throat was again gargled two hours after tablet was taken
1 hour
0
...
134,750,000
Normal conditions except that mouth and teeth were throughly washed with soap just before gargling
19 days
0
32,600,000
...
Same as above
1 hour
0
33,125,000
...
Same as above
1 hour
0
40,375,000
...
Teeth were not washed. Otherwise normal conditions
2 days
0
33,500,000
...
Same as above
1 hour
0
43,330,000
...
Same as above
1 hour
0
54,000,000
...
Same as above
1 hour
0
50,000,000
...
Same as above
1 hour
0
67,000,000
...
Mouth and teeth thoroughly washed with soap just before throat was gargled
2 days
0
5,270,000
...
Same as above
1 hour
0
10,916,000
...
Same as above
1 hour
0
8,275,000
...
Normal conditions, but 1 c.c. of sterile rabbit’s blood was added to each plate
3 days
0
228,750,000
...
Count from the same gargle as above. No blood used in the plates
0
0
60,625,000
...
Normal conditions, but count was made on blood agar
1 hour
0
431,250,000
...
Count from the same gargle as above. No blood used in the plates
0
0
59,625,000
...
Normal conditions, count was made on blood agar
2 days
0
683,300,000
...
Same gargle as above, but count was made on plain agar
0
0
58,500,000
...
One tablet was taken just after preceding gargle. After one hour throat was again gargled. Count on blood agar
1 hour
1 tablet
...
558,300,000
Same gargle as above, but count was made on plain agar
0
1 tablet
...
55,875,000
Normal conditions
2 days
0
79,125,000
...
One tablet was taken just ten minutes before gargle was made
1 hour
16 min.
1 tablet
...
56,250,000
Normal conditions
2 days
0
46,750,000
...
One tablet was taken just ten minutes before throat was gargled
1 hour
1 tablet
...
38,500,000
Teeth and mouth were thoroughly washed with soap just before gargle was made
5 days
0
47,370,000
...
Teeth washed as above and one tablet taken ten minutes before gargle was made
1 hour
1 tablet
...
21,225,000
Finally a determination was made of the number of streptococci in the throat before and after the use of Formamint. The throat was gargled in the manner previously described. The streptococcus count was made by the dilution method as given by Heinemann.24 Culture tubes were used instead of fermentation tubes. One per cent. dextrose broth was the medium employed. One cubic centimeter was added to each of a series of ten tubes for each dilution and the following dilutions were used: 1:10,000, 1:100,000 and 1:1,000,000.
The results given in Table 4 are the average count from a number of dilutions and are reported as the total number washed out by the 50 c.c. of salt solution.
TABLE 4.—SHOWING THAT FORMAMINT FAILS TO REDUCE THE NUMBER
OF STREPTOCOCCI IN THE THROAT
14 The Journal A. M. A., Feb. 24, 1912, p. 572.
15 De Santi: Medical Magazine 16:141, 1907.
16 Rosenberg: Lancet, London 2:1871, 1905.
17 Seifert: Pharmakol. u. therap. Rundschau, 1904, No. 14; quoted by De Santi (Note 2).
18 Daus: Med. Klin. 2:4110, 1906.
19 Rheinboldt: Deutsch. med. Wchnschr. 32:587, 1906.
20 Rosenberg: Therap. d. Gegen. 7:55, 1905.
21 Wingrave: Lancet, London 2:1067, 1906.
22 Young: Lancet, London 1:975, 1908.
23 Sutton: Volumetric Analysis, Edition 10, p. 390.
24 Heinemann: Laboratory Guide in Bacteriology, p. 86.
Conditions of Test
Time Since Preceding Test Amount of Formamint Used No. Found in Throat Before Use of FormamintNo. Found
in Throat
After Use of
Formamint
Normal
...
0
1,200,000
...
One tablet was taken and throat gargled one hour later
4 days
1 tablet
...
14,750,000
Normal
3 days
0
9,950,000
...
One tablet was taken and throat gargled ten minutes later
1 hour
1 tablet
...
8,000,000
Discussion
The contention that Formamint contains formaldehyd was confirmed by analysis.
The manufacturers also maintain that Formamint is a new, definite chemical compound, consisting of five molecules of formaldehyd and one molecule of lactose, and that when dissolved in the saliva the formaldehyd is liberated in some new and peculiar form, which they call nascent formaldehyd. This new kind of formaldehyd is, according to the advertising literature, especially powerful in its germicidal properties and at the same time has absolutely no irritating or harmful effects.
NOT A CHEMICAL COMPOUND
Thoms,25 retained as an expert by the German government, decided, after a series of chemical investigations, that Formamint was not a definite chemical compound, but that it was probably a solid solution of formaldehyd in lactose. He proved that when the process of manufacture was carried out in exactly the way called for by the Formamint patents, compounds containing a greater or less per cent. of formaldehyd could be made while the other properties remained similar to those of Formamint. The composition of the final product depended on the proportion of the components used in the process. Therefore Formamint did not form a safe means of uniform dosage.
As a result of Thoms’ work the German courts held that Formamint was not a new chemical compound. Consequently the Formamint patent (Number 189036) was annulled in Berlin, Nov. 29, 1913.
Again the contention that formaldehyd in the nascent or active condition is less poisonous and irritating than in its ordinary form is contrary to what would be expected from the behavior of such compounds. If it were liberated, as claimed, in the “nascent” condition, it would be, for that very reason, not only more active but also more harmful.
As a matter of fact, Formamint did have an irritant effect on the worker who carried out these investigations. When one tablet was taken each hour for twelve consecutive hours, marked irritation of the intestinal tract resulted. There was almost sufficient nausea to cause vomiting and uneasiness in the alimentary canal following the experiment. When the twenty-four tablets were taken the results were similar but more pronounced. This is decidedly in contradiction to the assertions of the manufacturers.
Otto Seifert,26 moreover, cites the following:
“By Effects: Only a few patients complain of an unpleasant sharp taste, burning of the tongue (Seifert, Sklarek). Among the general symptoms observed are urticaria-like exanthems (Glaser, Roters), which are accompanied by nausea, vomiting, headache, insomnia and vertigo, burning and irritability especially in the larynx (Meissner); phenomena of poisoning (Geissler); gastric disturbances (Engelmann); renal irritation (Steinhard); unsuited for diabetics (Voit).”
The contention that Formamint, when mixed directly with mediums and left in contact with bacteria, will kill the organisms was corroborated. Thus the statements and pictures in the booklet, “The Gospel of Prevention,” which is enclosed with each bottle of Formamint, showing the inhibition of growth of air bacteria on plates containing Formamint are no doubt true and authentic.
Finally, the claim that Formamint is an almost perfect throat disinfectant was by no means confirmed, as a glance at the tables will show. One hour after it is taken, even when a tablet was used each half hour for twelve hours, the number of bacteria in the throat was practically the same as when Formamint was not used. Even ten minutes after taking a tablet the number of bacteria in the throat was never greatly reduced, as is maintained by the manufacturers.
HAS NO SELECTIVE ACTION
Formamint exerts no selective action in killing off the very delicate organisms which are more apt to be pathogenic. When the comparative counts were made on blood agar which would favor the growth of the delicate parasitic organisms, no reduction whatever was shown by the use of Formamint.
The number of streptococci was found to be the same, within limits of experimental error, ten minutes after taking a tablet as it was before the tablet was taken.
Therefore it seems that Formamint fails, as any such germicide would be expected to fail, to kill bacteria in the crypts and recesses of the throat, for when dissolved in the mouth it cannot reach and remain in contact with the organisms long enough to kill them before it is swallowed.
SUMMARY
Summed up, the investigation shows:
1. That the claims made for Formamint are extravagant and misleading.
2. That the recommendations for the use of these tablets may be, in some cases, fraught with danger and are a menace, not only to the health of the individual, but also to the safety of the community.
3. That the claim that Formamint is a definite chemical compound is false.
4. That the use of Formamint may produce marked irritation of the intestinal tract.
5. That Formamint is not a throat disinfectant, as the manufacturers maintain, but its action on the bacteria of the throat is an almost negligible one and dependence on Formamint for the prevention of infection and for curing disease is not only unwise but dangerous.
6. That Formamint conflicts with the rules of the Council. False statements are made with regard to its composition (Rule 1); grossly unwarranted claims are made for its therapeutic properties (Rule 6), and therefore its exploitation to the public (Rules 3 and 4) is a public danger.
It is recommended that this report be published, to call attention not only to the falsity of the claims made for, and the danger in the use of, Formamint, but also to emphasize the utter inefficiency of all such methods of “disinfecting” the throat.—(From The Journal A. M. A., Aug. 28, 1915.)
HYDRAGOGIN
Report of the Council on Pharmacy and Chemistry
Hydragogin (C. Bischoff & Co., New York, selling agents) is advertised as “a most powerful diuretic and cardiac tonic.” The composition given is:
“Fifteen parts of the remedy contain 0.5 parts oxysaponin, 1.5 parts tincture of digitalis, 2.5 parts tincture of strophanthus, scillipicrin and scillitoxin, the active principles of scilla maritima, and alcohol.”
It is not clear from this statement whether 15 parts of Hydragogin contain 2.5 parts of tincture of strophanthus, plus unspecified amounts of scillipicrin and scillitoxin, or 2.5 parts of a mixture, in unspecified proportions, of tincture of strophanthus, scillipicrin and scillitoxin. The activity of strophanthus, after it enters the blood stream, is about fifty times that of digitalis; hence, if the former proportion is the true one, in giving an amount of Hydragogin which ensures the full therapeutic effect of the digitalis, one would administer an almost certainly fatal amount of strophanthus. Whatever the proportion of strophanthus may be, however, the administration of a mixture of digitalis and strophanthus in fixed proportions is indefensible. At times it is advisable to follow one of these drugs with the other in the treatment of cardiac disease. The simultaneous administration of the two continuously in fixed proportions, however, is injudicious, because of the great difference between their rates of absorption and in their activity after they enter the blood stream. The action of digitalis, moreover, persists much longer than does that of strophanthus.
An advertising circular contains the following claim:
“The well-known diuretic properties of digitalis, strophanthus and squills are greatly enhanced by the addition of the oxysaponin.”
This is not true. Saponins are not synergistic with digitalis therapeutically; on the contrary, they exert a purely deleterious action on the heart when they enter the circulation.
The symptoms of cardiac disease are often difficult to distinguish from the toxic actions of the digitalis bodies. Since these bodies must often be given to the point of beginning toxic action in order to induce the full therapeutic effects, it is obvious that the administration of a mixture of digitalis, strophanthus, saponin and active principles of squill is especially liable to induce serious toxic effects which cannot be distinguished from the symptoms of the disease.
Hydragogin is a shotgun mixture of semisecret composition; it is marketed under a therapeutically suggestive name, and advertised by means of unwarranted therapeutic claims. It is therefore in conflict with Rules 1, 6, 8 and 10. The Council held Hydragogin ineligible for New and Nonofficial Remedies.—(From The Journal A. M. A., Sept. 4, 1915.)
FILUDINE
Report of the Council on Pharmacy and Chemistry
Filudine is said to be prepared by J. L. Chatelain, Paris, and is sold in this country by Geo. J. Wallau, Inc., New York. It is offered as a remedy for “biliary insufficiency,” “hepatic insufficiency,” “intestinal dyspepsia,” “all affections of the liver (diabetes, cirrhosis, cancer, etc.),” “malaria,” “obesity” and “tuberculosis.”
No quantitative information is furnished as to the composition of the preparation and there are noteworthy discrepancies in the various statements regarding the ingredients. In one number of “Treatment,” a self-styled “Review” of medical literature (actually devoted to advertising the preparations sold by Wallau), we are told that
“This product [Filudine] is a more concentrated and potent extract of the liver, with which is combined an extract of the spleen. The liver and the spleen are so intimately interdependent, that the addition of a splenary extract to the liver extract is a signal improvement from which a synergistic action results. Thiarféine is also added, as it helps somewhat to combat the anaemia from which all diabetics suffer more or less.”
Thiarféine is said to be
“Thiomethylarsinate of Caffein, a new salt discovered by M. Chatelain.”
Another circular, which gives an imposing formula for “thiarféine” or “thiomethylarsinate of caffein,” states that
“Sulphurated methylarsinate is an arsenical preparation devoid of all toxicity on account of the intimate joining of its composing parts.”
And that
“Filudine can never be contraindicated.”
A statement of composition in a later number of “Treatment,” however, says that biliary extracts are components, in addition to the liver and spleen extracts. Moreover, thiarféine, the “new salt discovered by M. Chatelain,” is no longer “thiomethylarsinate,” but “thiocinnamate of caffein”; and a new formula is furnished for it.
We are told that
“Methyl-arsinate cannot be used in cases where fever is present....”
“M. Chatelain at first studied the action of thiomethylarsinate; clinical and physiological experimentation led him, however, to adopt thiocinnamate of caffein, of greater activity and with no contraindications.”
Nevertheless the same absence of contraindications was urged in favor of Filudine when it was said to contain the now discarded thiomethylarsinate of caffein.
The following are some of the unwarranted and even absurd claims:
“Filudine restores the liver’s functions. It is to the liver what digitalis is to the heart; it overcomes the insufficiency and stimulates the debilitated organ.”
In malaria “it is the only true specific when associated with quinine.”
“Filudine is ... the ideal medication for tuberculosis, conforming as it does with the most recent researches in the therapeusis of this affection.”
“We will not go as far as to say that Opotherapy completely restores unhealthy livers, for although the lesions of the hepatic parenchyma may be obliterated by regeneration, the lesions of the connective tissues are permanent, and may be observed at the postmortem examination. The new cells, however, do not present the same unhealthy conditions as those of the former diseased gland which they have replaced, and the liver can therefore function normally, so that the patient lives on; and he is satisfied with that.”
“Therefore, while regenerating the liver with Filudine, we cleanse it and combat its congested state with Urodonal. We cause it to produce urea from the excess of uric acid which it contains.”
“By the judicious and harmonious combination of the beneficial effects of Filudine and Urodonal, physicians not only possess the means of treating by rational methods Cirrhosis of the Liver in its various forms (which is one of the most terrible diseases which can afflict anyone) but what is still better, they can cure it.”
“The liver of a person suffering from obesity being incapable of fulfilling its functions in regard to the fatty tissues, the rational and up-to-date method of treatment is therefore to restore to the system, in the form of Filudine, the liver extracts which are lacking.”
Filudine is a mixture of semisecret composition. The therapeutic claims are manifestly unwarranted. The name is not indicative of the composition, whatever that may be, and no rational excuse is offered for the combination of liver and spleen extracts (with or without bile extracts) with “thiomethylarsinate” or “thiocinnamate” of caffein.
The Council therefore held Filudine ineligible for New and Nonofficial Remedies.—(From The Journal A. M. A., Sept. 18, 1915.)
LACTOPEPTINE AND ELIXIR LACTOPEPTINE
Report of the Council on Pharmacy and Chemistry
Mixtures of pepsin and pancreatin are therapeutically irrational; the two substances are not indicated in the same conditions, nor can they act together. Under physiologic conditions, such mixtures are chemically impossible: in a liquid medium the ingredients destroy each other.
Lactopeptin is manufactured by the New York Pharmacal Association, Yonkers, N. Y. It is sold under the claim that it contains, pepsin, diastase, pancreatin, lactic acid and hydrochloric acid. This product was among the first proprietary preparations examined by the Council on Pharmacy and Chemistry. The report of the investigation was published in The Journal, March 16, 1907, p. 959. The preparation was found to be practically inert—“essentially a weak saccharated pepsin,” devoid of tryptic activity.
Six years later it was still widely advertised with the same irrational claims. A referee (A) therefore examined Lactopeptine (powdered) for the Council in 1913, and confirmed the previous findings. The referee’s report was published in The Journal, Aug. 2, 1913, p. 358.
Nearly four months after this publication, the manufacturer protested against the report, maintaining, contrary to the findings of the Council, that Lactopeptine possesses pancreatic activity and contains “loosely combined” hydrochloric acid. Referee A therefore repeated his examination, and a second referee (B), independently, examined specimens of Lactopeptine (powder) purchased on the open market for the purpose shortly before.
A few specimens examined by these two referees showed a slight tryptic activity; most of them showed none. The amount of hydrochloric acid present was insignificant.
The reports of the two referees were referred to the manufacturers, who again protested vehemently against these findings, this time on the ground that the specimens were old. The manufacturers also cited the work of three chemists to disprove the findings of the referees, and demanded that the Council reexamine Lactopeptine, making use of fresh specimens. The Council refused for the following reasons:
1. So long as the packages of Lactopeptine are not dated, the activity of specimens known to be fresh is of no practical importance. The activity of the actual market supply is the only question of interest to the profession. The only fair test is that made on specimens representative of the product sold to the ultimate consumer.
2. The evidence presented by the manufacturers did not warrant a reexamination, since the work of two of the chemists cited substantially corroborates the results obtained by the Council’s referees from the fresher specimens. The figures for tryptic activity obtained by the third chemist cited by the manufacturers could not be accepted by the Council, since it was at variance with all other known results of investigations of Lactopeptine.
3. As stated at the outset, whatever the tryptic activity of the mixture, it is therapeutically useless. A demonstration of tryptic activity in a mixture containing both pepsin and pancreatin is of merely theoretical interest.
Such activity, of course, cannot be expected, even on theoretical grounds, in liquid mixtures like Elixir Lactopeptine.
The Council therefore again declared Lactopeptine (powder and tablets) and Elixir Lactopeptine ineligible for New and Nonofficial Remedies and authorized publication of the following statement.
W. A. Puckner, Secretary
THE COUNCIL’S REPORT
Lactopeptine powder (New York Pharmacal Association, Yonkers, N. Y.) was examined by the Council in 1907. At that time it was claimed to contain
“... the five active agents of digestion—pepsin, diastase (veg. ptyalin), pancreatin, lactic acid and hydrochloric acid—combined in the proper proportion to insure the best results.”
The examination showed that the preparation was essentially “a weak saccharated pepsin,” containing but small amounts of pepsin, no hydrochloric acid, or mere traces only, and no diastase or pancreatin (The Journal, March 16, 1907).
In 1913, the product was reexamined, because the claims, as to both composition and therapeutic value, were still being made. Samples were tested both of the American product, and of a British product from John Morgan Richards & Sons, London. The original findings were confirmed and the results were published in The Journal, Aug. 2, 1913, p. 358. Nearly four months later (November 24) the New York Pharmacal Association wrote to the Council, objecting to the findings and maintaining that Lactopeptine possesses pancreatic activity and contains (“in loose chemical combination”) hydrochloric acid. In accordance with the custom of the Council, the work was sent back for review to the referee (A), whose conclusions were then tested by a second referee (B), a physiologic chemist, not a member of the Council, selected because of his special knowledge of the subject.
In December, 1913, Referee A made a large number of new tests to determine proteolytic and amylolytic power. His results show that the ferment activity of the preparation is so low as to merit no recognition in practical use. The tests also show that the amount of lactic acid or “loosely combined HCl” (or both) present is too small to have any appreciable physiologic activity and therefore to be of any therapeutic value.
Nine samples of Lactopeptine purchased in the open market in December, 1913, and January, 1914, were examined by Referee B early in 1914. His studies show absence of amylase in all samples; presence of pepsin, giving weak reactions even when compared with those of old pepsin preparations; complete absence of trypsin in seven out of nine samples, tryptic reaction being obtained in two samples, in one of which the reaction, “slight at best and of no practical import,” was obtained only after treatment for twelve hours or more.
The presence of tryptic activity in two out of the nine samples may be due to the fresher condition of these specimens, as indicated by the serial numbers. The evidence shows that it is a commercial impossibility to market mixtures of pepsin, pancreatin and lactic acid so that they can display any material tryptic activity.
It should be reaffirmed that mixtures combining peptic and pancreatic activities are not feasible, because pepsin cannot act except in the presence of acid, and pancreatin is destroyed by acid and by peptic activity. Furthermore, in conditions in which pancreatin is called for, pepsin is not, and vice versa; therefore the administration of mixtures of pepsin and pancreatin would be unjustified, even if both constituents could be expected to exert activity.
The foregoing observations apply to Lactopeptine in powder and tablet form.
While mixtures of pepsin and pancreatin are unscientific and unjustified, theoretically the two substances may coexist in a solid preparation, and the activity of such a preparation is consequently a proper subject of investigation. Theoretically as well as practically, however, pepsin and pancreatin cannot exist together in solution. The claims made for Elixir Lactopeptine and all other liquid preparations sold as mixtures of pepsin and pancreatin are therefore impossible. The Council has previously taken action (The Journal, Feb. 2, 1907, p. 434) refusing to approve for inclusion with New and Nonofficial Remedies such preparations, calling the attention of the medical profession and of manufacturers to their worthlessness, and requesting the American Pharmaceutical Association to instruct its committee on the National Formulary to omit from the next edition of that work a liquid preparation of pepsin and pancreatin recognized under the title of “elixir digestivum compositum.”
It is recommended that the Council reaffirm this previous action, and that Lactopeptine and Elixir Lactopeptine be declared ineligible for New and Nonofficial Remedies because of conflict with Rule 10 (“No article will be admitted which, because of its unscientific composition, is useless or inimical to the best interests of the public or of the medical profession”).
Manufacturers’ Protest and Council’s Answer
The foregoing was submitted, together with the findings of the two referees, to the manufacturers. They protested again, alleging that:
AGE OF SPECIMENS
First.—The specimens of Lactopeptine examined by the second referee were old. The dates of manufacture corresponding to the several batch numbers are supplied by the manufacturers as follows:
2275 (Powder)
September, 1908
2301 (Powder)
June, 1909
2312 (Powder)
December, 1909
2348 (Powder)
October, 1911
2352 (Powder)
December, 1911
2364 (Powder)
July, 1912
2374 (Powder)
March, 1913
2383 (Powder)
October, 1913
1638 (Tablets)
October, 1911
The manufacturers assert that they do not understand how specimens of these ages could have been purchased on the open market in 1913 and 1914, inasmuch as their agents are and long have been instructed to take up from the druggist all lots of Lactopeptine which, as indicated by the batch numbers, have attained “any appreciable age.” The age of the specimens, the manufacturers declare, deprives the table in the second referee’s report of “all significance or interest.”
As previously stated, however, the specimens of Lactopeptine examined were purchased on the open market in various localities in unbroken packages, in December, 1913, and January, 1914. They thus represent stock used in filling physicians’ prescriptions or sold to the public. Neither the referees nor any one connected with the Council had any means of knowing the age of the specimens until the dates of manufacture were furnished by the New York Pharmacal Association. The first tests of the second referee were made in February, 1914, on Specimens 2374 and 2383, which were then, it would appear, about one year old and four months old, respectively. The Council has repeatedly urged that pharmaceutical substances which are subject to deterioration should be dated by the manufacturer, and a similar suggestion has been made by the Bureau of Chemistry of the U. S. Department of Agriculture concerning mixtures containing enzymes. Notwithstanding the instructions which the New York Pharmacal Association claims to have given its agents, the market supply of Lactopeptine in December, 1913, and January, 1914, was not composed of new stock, and until the manufacturers adopt the practice of dating packages, there can be no assurance that it will be fresh. In this connection, it is of interest to note that the Bureau of Chemistry of the U. S. Department of Agriculture has issued a warning that it will judge such products by the degree of their activity when they reach the consumer, i. e., as they are found on the market.
REPORTS OF OTHER CHEMISTS
Second.—The New York Pharmacal Association cites the work of several chemists, who have examined Lactopeptine and report the presence of tryptic activity. Dr. S. R. Benedict in December, 1913, reported to the Council “distinct” tryptic activity (digestion in twelve hours by Lactopeptine of 4.2 times its weight of fibrin containing 50 per cent. moisture) in specimens examined by him. These specimens were numbered 2382, and were therefore probably manufactured in October, 1913; compare the dates furnished by the manufacturer for the specimens used by the second referee. No tests against other preparations possessing tryptic activity are reported, and Dr. Benedict expressly disclaims any opinion as to the therapeutic value of the preparation.27 Dr. P. B. Hawk, whose report was submitted by the manufacturers, found in Lactopeptine by Fermi’s method one-fifth tryptic activity of that of Merck’s pancreatin, and by Grützner’s method an activity of 18 per cent. of the pancreatin. A test for the production of tryptophan was reported positive. The New York Pharmacal Association also submitted a report from Dr. A. W. Balch, who found pepsin, rennin, trypsin, steapsin, amylopsin and lactic acid present in Lactopeptine; also an amount of combined hydrochloric acid in 1 gm. the equivalent of 1.05 c.c. tenth normal solution or 0.00383 gm. hydrochloric acid. (He reports digestion in twenty-four hours by Lactopeptine of 25 times its own weight of fibrin. “An active extract of pancreas reacted exactly like the Lactopeptine solution.”) The serial numbers of the specimens tested by Hawk and Balch are not given, but no doubt they were fresh.
CONCLUSIONS
The New York Pharmacal Association demanded that the referee reexamine Lactopeptine, making use of fresh specimens. The Council held that this was unnecessary, for the following reasons:
1. The previous finding of the Council, that specimens of Lactopeptine found on the open market are essentially weak saccharated pepsins, is not to be refuted by examination of fresh specimens. Even if it be assumed that all old specimens of Lactopeptine have been withdrawn from the market since the last purchase of specimens for the use of the Council’s referee, there can be no assurance that the stock will be constantly kept fresh. Unless the manufacturers date their product, physicians cannot know that their prescriptions are filled with fresh material. Nor is it reasonable to ask that the Council examine the market supply of any given proprietary at a time selected by the manufacturers.
2. Without entering into all questions of detail in the analyses, the Council is willing to accept the reports of Drs. Benedict and Hawk as representative of fresh Lactopeptine powder. It is therefore unnecessary for the Council to make further experiments along this line. The results of these two chemists in no wise contradict the conclusions of the Council’s referees, being comparable with those obtained by the referee on the fresher specimens used by them. The experiments of Drs. Hawk and Benedict show a degree of tryptic activity which, though chemically not negligible, is quite without significance practically, even if it could be assumed that the trypsin in the fresh Lactopeptine escaped destruction in the stomach. The figures for tryptic activity given by Dr. Benedict do not differ materially from those of the first referee. Those of Professor Hawk show a tryptic activity of from 18 to 20 per cent. of that of commercial pancreatin—and commercial pancreatins ordinarily are of low tryptic activity, if not inert (see Long and Muhleman: Arch. Int. Med., February, 1914, p. 314.) The reports of these chemists present no reason for changing the conclusion that “it is a commercial impossibility to market mixtures of pepsin, pancreatin and lactic acid so that they can display any material tryptic activity.”
The results which Dr. Balch obtained in a test for tryptic activity show a marked discrepancy with those obtained by Drs. Hawk and Benedict, not to mention the Council’s referees, and also with the fact that only about 11 per cent. of “pancreatin” is claimed in the published formula of Lactopeptine. The Council is unable to accept Dr. Balch’s result for trypsin or rennin as reliable. His other results are without significance and call for no special comment.
3. Even if tryptic activity were conceded to Lactopeptine, the preparation, like all preparations containing pepsin and pancreatin, would still be, as previously stated, therapeutically irrational.
The Council approved the report.
Report of Referee A
In view of the manufacturer’s reiteration of the claims for Lactopeptine powder, I have carried out further experiments to determine its proteolytic and amylolytic power.
For the proteolytic test I used fresh, well washed fibrin and examined samples of Lactopeptine powder numbered as follows:
No. 1. A part of the English product examined and reported on last spring.
No. 2. A fresh bottle obtained at a Chicago retail drug store in December, 1913.
No. 3. A fresh bottle obtained at a Chicago retail store in December, 1913.
Portions of 1 gm. each of these samples were mixed with 5 gm. fibrin, 100 mg. of sodium carbonate and 50 c.c. of water in flasks. A little toluene was added to each flask, which was then closed with a tuft of cotton and the mixtures were incubated at 40 degrees through twenty-four hours. At the end of that time there was no marked change in the quantity of the fibrin remaining in each flask, the larger part by far being undigested.
As a control I used the sample of an active commercial trypsin, of which I added 500 mg. to the same quantity of water, fibrin and sodium carbonate. This was digested in the same bath at the same time. The digestion was practically completed in less than ten minutes, only minute flakes of the fibrin remaining.
It is evident that the digestive power of the Lactopeptine must be extremely low, and only a small fraction of that exhibited by a commercially good trypsin.
In an experiment with the English sample carried out through nineteen hours as above, using 2 gm. of fibrin and 100 mg. of ferment, it was found by nitrogen tests on the filtrate that about 12.2 per cent. of the protein had been brought into solution, an amount which is practically without importance in a digestion of such duration.
To test the starch digestive power I have made a large number of experiments. In a series just completed I mixed 1 gm. portions of Samples 1 and 2 with water to make 100 c.c. volumes. Before making up to the final volumes 0.5 c.c. of normal sodium hydroxid was added to neutralize the slight acidity of the ferment as shown by phenolphthalein.
Of these mixtures 4, 6, 8 and 10 c.c. portions were mixed with 50 c.c. of 1 per cent. starch paste and incubated at 40 degrees to find the colorless end-point in the starch digestion, by the iodin test.
At the end of twenty-two hours the iodin reaction was as strong as at the beginning, indicating no appreciable starch digestion.
To the flasks in which no digestion had taken place under these conditions, 5 mg. of a pancreas ferment was added. This gave an almost immediate conversion to the colorless end-point. This ferment was a sample of Holadin which had been in the laboratory about a year. The 5 mg. completed the reaction to the colorless end-point in less than ten minutes.
In a similar test I used 2 gm. of Lactopeptine No. 3, made up to 100 c.c. with 1.2 c.c. of normal alkali. Ten and 15 c.c. portions were incubated with 50 c.c. of 1 per cent. starch paste through twenty hours at 40 degrees with no apparent result. The Holadin then added, 5 mg. being used, completed the conversion in less than ten minutes.
This shows that the medium was a proper one for the test and that the Lactopeptine must be extremely weak. No sugar tests were made because the Lactopeptine contains milk sugar to the extent of about 60 per cent.
Similar results for both protein and starch digestives have been obtained in a large number of experiments. These here quoted show that the ferment activity of the preparation is so low as to merit no recognition practically. The digestion of a few milligrams of fibrin or starch after many hours of contact, while being perhaps scientifically possible, is of no value when we come to a consideration of the use of such bodies as digestive ferments in medicine.
The amount of lactic acid or “loosely combined HCl” present in Lactopeptine is very small, since the total acid which may be titrated by sodium hydroxid and phenolphthalein is measured by 0.5 c.c. of the normal hydroxid for 1 gm. of the Lactopeptine powder, in the mean. In different samples examined the range was found to be from 0.41 c.c. to 0.6 c.c. Tests with methyl orange, methyl red and other indicators showed that the free acidity is but trifling; if the whole of this acid, as measured by phenolphthalein, were calculated to HCl, the amount would be too small to have any appreciable physiologic activity, in view of the daily dose recommended, 10 to 20 grains of the powder.
Report of Referee B
The following table gives a summary of the results of my investigations on Lactopeptine. The numbers in the extreme left-hand column are the manufacturer’s identifying marks. These, it is assumed, run serially, the higher numbers indicating fresher specimens.
TABLE SHOWING ENZYMIC POWER OF LACTOPEPTINE PREPARATIONS
Amylase
Pepsin
Rennin
Trypsin
Lipase
2275
–
+
+
–
–
2301
–
+
+
–
–
2312
–
+
+
–
–
2348
–
+
+
–
–
2352
–
+
+
–
–
2364
–
+
+
+(?)
–
2374
–
+
+
–
+(?)
2383
–
+
+
+
+(?)
1638 (tablets)
–
+
+
–
–
Pancreatin (Old)
–
..
..
++
–
The conclusions in the foregoing summary depend on the following criteria:
Amylase: removal of starch (paste), small in proportion to begin with.
Pepsin: solution of small shreds of fresh fibrin in acid media.
Rennin: curdling of milk in moderate excess.
Trypsin: solution of small shreds of fresh fibrin in neutral and alkaline media, and tryptophan test.
Lipase: coloration of litmus-milk; exact color controls.
All tests were suitably controlled. The responses for pepsin were weak even when compared with those of old pepsin preparations.
In the table above, the interrogation points in parentheses (?) refer to results that were obtained after treatment for from twelve to twenty-four hours and indicates that the change was slight at best and of no practical import.—(From The Journal A. M. A., Oct. 23, 1915, with additions.)
IODUM-MILLER AND IOD-IZD-OIL (MILLER’S)
Report of the Council on Pharmacy and Chemistry
The Council adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
A referee has submitted to the Council the following report of the Chemical Laboratory of the American Medical Association on Iodum-Miller and Iod-Izd-Oil (Miller’s) (Iodum-Miller Co., Kansas City, Mo.):
The unsatisfactory statements made in regard to the composition of Iodum-Miller and the far-reaching therapeutic recommendations for it induced the laboratory to make a chemical examination of the preparation. It claimed more or less directly that the preparation is entirely new and possesses novel characteristics.
It is asserted that
“Iodum-Miller is made from Soot Iodine, which is our own product. This Soot Iodine is SOLUBLE IN WATER before being combined with its base C.P. Glycerine.”
No information regarding “soot iodine” is offered and an inquiry sent to the proprietors by a physician brought only the noncommittal reply that “soot iodine”
25 Thoms: Arb. a. d. Pharm. Inst. d. Universität, Berlin 11:210, 1914.
26 Seifert, Otto: Die Nebenwirkungen der modernen Arzneimittel, 1915.
27 Dr. Benedict’s personal communication to a member of the Council is as follows:
“is made from Resublime [resublimed?] Iodine by a chemical process which renders it soluble in water before being combined with its base.”
Iodum-Miller is said to contain
“Active Free Iodine 2.2 grams per 100 c.c., 10. grains per fluid ounce, 1.7% by weight.”
“In addition to the active free iodine ... IODUM-MILLER carries a still greater per cent of Iodine in its basic combination....”
According to the label, the preparation is
“An Iodine for External and Internal use ... 45 drops equals 1 dr. by weight. Each drop equals the per cent. of iodine in 1 gr. potas. iodide.”
Iodum-Miller is a heavy, dark liquid having an odor characteristic of ether (ethyl oxid). Qualitative tests revealed the presence of glycerin, free iodin, iodid and potassium. The specific gravity at 25 degrees was 1.284. Direct titration with sodium thiosulphate solution indicated the presence of 1.68 per cent. of free iodin. A determination of the total iodin content by the Hunter method indicated 3.06 per cent. Subtraction of the amount of free iodin found from the total amount of iodin present, gives 1.38 per cent. combined iodin. Assuming this to be present as potassium iodid, as appears probable from the qualitative examination and from the quantitative determination of potassium, 1.80 per cent. potassium iodid is indicated. From this examination it is concluded that Iodum-Miller is, essentially, a solution of iodin and potassium iodid in glycerin, containing 1.68 per cent. free iodin and 1.80 per cent. potassium iodid. The examination contradicts the assumption that Iodum-Miller is either novel in principle or new. Moreover, accepting the firm’s statement that 45 drops weigh 1 dram (60 grains) the examination shows that one drop equals not “the per cent. of iodine in 1 gr. potas. iodide” but instead, the per cent. of iodin in only 1⁄20 grain potassium iodid. As the statement that “Each drop equals the per cent. of iodine in 1 gr. potas. iodide” appears on the label of the trade package, Iodum-Miller would seem to be misbranded under the federal Food and Drugs Act.
The recommended internal dosage of Iodum-Miller (from 1⁄2 to 20 drops) is equivalent to from 1⁄40 to 1 grain of potassium iodid. Its external efficacy in comparison with that of other iodin preparations may be estimated by comparing the respective free iodin contents, since the germicidal power of combined iodid is negligible. While Iodum-Miller contains 2.15 gm. free iodin in 100 c.c., tincture of iodin contains 7 gm. per 100 c.c. and compound solution of iodin (Lugol’s solution) contains 5 gm. free iodin in 100 gm.
Among the advertising literature is a circular which purports to be a “Certificate from Kansas City Testing Laboratory, by Roy Cross, Secretary.” The “certificate” attempts to prove that Iodum-Miller is vastly superior to the official tincture of iodin as a germicide, asserting that “In the process of dissolving [tincture of iodin] in water, a very large amount of the iodin is lost by precipitation....” This is not true of the tincture of iodin which is now official, though it is true of the tincture official in former editions of the Pharmacopeia. The report ignores completely the widely used aqueous solution of iodin.
Iod-Izd-Oil (Miller’s) is said to be an “Iodine Combination” made “from the same Soluble Soot Iodine as is IODUM-MILLER.” It is said to “liberate Free Soluble Iodine” when applied to the skin, mucous surfaces, etc. It is further defined as “Soluble Iodine combined with water-white Hydrocarbon Oil” and is said to liberate “Soluble Iodine 2 per cent.” While these statements suggest that Iod-Izd-Oil (Miller’s) contains the iodin-potassium iodid combination contained in Iodum-Miller, analysis indicated the oil to be a simple solution of iodin in liquid petrolatum. Quantitative determinations indicated, not 2 per cent. of iodin, as claimed, but only 0.42 per cent. and all of this was present as free iodin.
REFEREE’S REPORT
The following therapeutic claims appear on the label of a bottle of Iodum-Miller:
“EXTERNAL INDICATIONS
“Tuberculosis, Pneumonia, Pleurisy, Cough, Sore Throat, Pyorrhea, Tonsilitis, Rheumatism, Spinal Irritation, Boils, Felons or any Pain. Periostitis, Carbuncles, Fistula in Ano, Goiter, Blood Poison, Diseases of Uterus and appendages (apply full strength on cotton wrapped applicator), Gonorrhea, acute or chronic in both sexes, Orchitis, Bubo, Prostatitis, Swellings, Enlarged Glands, Etc.”
“INTERNAL INDICATIONS
“Pneumonia, Tuberculosis, Pleurisy, Typhoid Fever, Syphilis, Catarrh of Mucous surface of Alimentary Canal, Autotoxemia, Vomiting of Pregnancy, Rheumatism, Chronic Glandular and Organic Affections.”
The “certificate” from the Kansas City Testing Laboratory, mentioned above, states that Iodum-Miller was found to have a germicidal value nineteen times greater than carbolic acid—a somewhat remarkable finding in view of the fact that iodin dissolved by means of potassium iodid in alcohol or water, when tried on the typhoid bacillus has recently been found to possess only four times the germicidal value of carbolic acid in a solution of the same strength (Maben and White: Chem. and Drug., Jan. 30, 1915, p. 144). The “certificate” further states that the test “shows available iodine as found in IODUM-MILLER to have the greatest bactericidal power of any substance that we have ever tested that can be used medicinally.” There is no reason to believe that the desire to please its patrons has led the “testing laboratory” astray from the literal truth. The laboratory’s experience may be limited and the statement therefore entirely correct as far as it goes. No mention, however, is made of any tests comparing the germ-destroying power of Iodum-Miller with that of tincture of iodin, which contains 7 per cent. free iodin, unless the casual statement that “Iodum-Miller sterilized [the skin] more quickly” than tincture of iodin, be taken to imply such tests. It is not clear, however, by what means the laboratory was able to determine that there were no bacteria left alive in the skin after application of tincture of iodin and Iodum-Miller; no details are given of the methods used in arriving at this conclusion.
A circular says that Iodum-Miller
“... gives the Greatest Bactericidal and Therapeutic Action, whether used Internally, Externally, Hypodermically or Intravenously.”
In the light of the preceding report of the Chemical Laboratory of the Association, these claims require little comment. The laboratory has shown that the free iodin content (and consequently the germicidal efficiency) of Iodum-Miller is less than half that of Lugol’s solution, and less than a third of that of the official tincture of iodin. As for the advice to use Iodum-Miller internally in diseases ranging from pneumonia to syphilis and from typhoid to tuberculosis, in order to be convinced of its dangerous character, it is necessary only to recall that this treatment is equivalent to the administration of small doses of iodid—from 1⁄40 to 1 grain of potassium iodid. The mystery being removed from the composition of Iodum-Miller, the absurd extravagance of the claims made for it becomes manifest. The criticisms of the Council on the recommendations for Burnham’s Soluble Iodine (The Journal A. M. A., May 15, 1915, p. 1673) apply in almost every particular to Iodum-Miller.
Unwarranted therapeutic claims are made for Iodum-Miller; incorrect statements are made with regard to its composition and that of Iod-Izd-Oil (Miller’s); and the application of a trade name to both of these products is unjustifiable, since neither is original. It is therefore recommended that Iodum-Miller and Iod-Izd-Oil (Miller’s) be held ineligible for New and Nonofficial Remedies—(Abstracted in The Journal A. M. A., Oct. 2, 1915.)
ELIXIR IODO-BROMIDE OF CALCIUM COMP. “WITHOUT MERCURY” AND “WITH MERCURY”
Report of the Council on Pharmacy and Chemistry
The Tilden Company, New Lebanon, N. Y., and St. Louis, Mo., sells “Elixir Iodo-Bromide of Calcium Comp. without Mercury” and “Elixir Iodo-Bromide of Calcium Comp. with Mercury.” The latter is said to contain, in addition to the ingredients of the former, 1⁄100 grain mercuric chlorid in each fluidram. According to the label the formula of the elixir “without mercury” is:
“Formula—Salts of Iodine, Bromine, Potassium, Sodium, Calcium, Magnesium with Stillingia, Sarsaparilla, Rumex, Dulcamara, Lappa, Taraxacum, Menispermum.”
A recent circular declares that the elixir contains:
“... a number of the most powerful alteratives of the pharmacopeia such as chemically pure iodin, magnesium, potassium with sarsaparilla, stillingia, prickly ash, burdock, taraxacum, etc. ... Each fluidounce contains seventy-two grains of the combined salts.”
The same circular also alleges that each dram of the preparation contains:
“... the equivalent of one and one-half grains of the combined iodids, potassium and calcium ...”
It will be observed that, (1) the two statements quoted from the circular make no reference to bromids; (2) the statement that each dram contains “the equivalent” of 11⁄2 grains of the combined iodids, potassium and calcium, accounts for but 12 of the 72 grains of “the combined salts” per fluidounce declared in the preceding quotation; (3) the circular mentions the presence of a drug—prickly ash—not declared on the label and, finally (4) none of the “formulas” gives the quantities of all of the several constituents.
It is evident from these “formulas” that the Tilden Company continues its policy of concealment and mystification as exemplified in the cases of Hydrocyanate of Iron, Tilden (discussed in The Journal, June 19, 1909, p. 2008), Febrisol (The Journal, June 29, 1912, p. 2043) and Respirazone (The Journal, June 14, 1913, p. 1899).
In the circular just quoted (“The Conquest of Syphilis”), all hope for the syphilitic is declared to rest in mercury and iodin, and it is implied that only through Elixir Iodo-Bromide of Calcium Comp. is it possible to obtain the greatest good from these drugs.
“Were the cleansing influences of these two drugs [mercury and iodin] unavailable to the luetic patient, he, truly, would be as pitiable an object as the leper ...
“Modern Pharmacy has devised no better means of utilizing these anti-syphilitics than Elixir Iodo-Bromide of Calcium Comp. (Tilden) with or without mercury.... The Elixir, in proper dosage, acts in specific fashion and is adapted for use in all stages of the disease.
“In the early months ... Elixir Iodo-Bromide of Calcium Comp. (Tilden) with mercury is a trustworthy weapon and the physician need have no fear but that it will subjugate the disease ...
“When ... the virulent stage is passed ... Elixir Iodo-Bromide of Calcium Comp. (Tilden) without mercury may be given the patient with every assurance that medicine’s most aggressive measures are being resorted to ... From time to time, up to the very end of the time honored three years’ period of treatment, it is well to put the patient back on the bichloride, using for this purpose the form of the Elixir administered in the first stages of the disease ...
“This regime ... will indubitably antidote the virus of syphilis and eradicate from the organism its every vestige.”
While it seems incredible that any physician would jeopardize the health—even the life—of a patient by accepting this boastful magniloquence as sound therapeutic advice, still the fact that certain medical journals lend their advertising pages to advertisements for Tilden’s Elixir with the caption “The Conquest of Syphilis” makes it incumbent on the Council to record its condemnation of the employment of this unscientific, semisecret mixture.
It is recommended that Elixir Iodo-Bromide of Calcium Comp. “without mercury” and “with mercury” be held in conflict with Rule 1 (secrecy of composition), Rule 6 (unwarranted therapeutic claims) and Rule 10 (unscientific composition).—(From The Journal A. M. A., Nov. 6, 1915.)
LECITHIN PREPARATIONS OMITTED FROM N. N. R.
Report of the Council on Pharmacy and Chemistry
The following report was sent to the manufacturers of the various lecithin preparations mentioned therein. As the replies of the manufacturers were obviously written from the commercial point of view and did not affect the Council’s conclusion that lecithin, when indicated, would be given more advantageously in the form of yolk of egg than in the less pure manufactured product, the Council directed that the report be published, together with extracts from the replies of the manufacturers.
W. A. Puckner, Secretary.
Commercial lecithin preparations are at best very impure substances; all are more or less altered from the original composition. Even with great care, the methods of extraction and drying always produce considerable decomposition; and in some cases the phosphorus and nitrogen contents bear but little relation to the theoretical values. (Long, J. H.: Jour. Am. Chem. Soc., xxx, 881. McLean, Hugh: Chem. Abstracts, May 20, 1915). There is not the slightest reliable evidence that commercial lecithin has any advantage over the lecithin contained in natural foods; the weight of probability is on the other side.
The doses recommended, moreover, are absurdly small; and the amount thus administered is without practical value. Why administer a few milligrams of a more or less decomposed lecithin when it is possible to give a far larger weight of a purer substance in the form of yolk of egg?
In view of these considerations the Council voted that the following proprietary products be omitted from the next edition of N. N. R.:
Glycerole of Lecithin
Lecibrin
Lecithin Solution
Lecithol
Neuro-Lecithin-Abbott
and that the general article on “Lecithin Preparations” be transferred to the annual Council Reports as a matter of record.
The report was submitted to the manufacturers. Their replies were evidently based on commercial consideration, and called for no modification in the report.
The referee recommended that the preceding report be published together with the following extracts from the replies of the manufacturers:
From Armour and Company:
“We are selling a good deal of Lecithol and it seems to be giving satisfactory results in some quarters.... We shall continue to advertise Lecithol along the lines we have employed heretofore.”
From the Abbott Laboratories:
“We can assure you of our confidence in the therapeutic value of Neuro-Lecithin. This has been attested by the reports of favorable results sent us by many physicians, as well as by the periodical literature of the last few years which contains a considerable number of very encouraging references to lecithin therapy.”
From Fairchild Bros. & Foster:
“We would like simply to say that the physician and the Council must be aware of the circumstances and the purposes which actuated us in placing lecithin at disposal, viz., the studies—research—of lecithin and the properties attributed to it and which led to inquiry for and consideration of it. The quantities proposed for medicinal use were not suggested by us; the suggestion of lecithin in small quantities as a therapeutic agent was obviously directed by those who proposed it.... The question whether lecithin, per se, has therapeutic properties in contrast to lecithin as naturally contained in food substances, is something we do not undertake to decide. The Council, on purely theoretical grounds, decides in the negative notwithstanding clinical experience—internal and hypodermic—and thus would deny lecithin the status of a new and nonofficial remedy, worthy of at least tentative progressive clinical consideration. We can only say that we offered bona fide lecithin and that we did not make the investigation of lecithin a pretext for the sale of all sorts of lecithin ‘jumbles’ with lecithin in small proportions, taking their name and making their bid on lecithin.”
Below appears the general article which has been omitted from N. N. R.:
Lecithin Preparations
Lecithins are fat-like bodies belonging to the group of phosphatides. They all consist of glyceryl esters containing two fatty acid radicals and the phosphoric acid radical in which one of the residual hydrogens is replaced by the choline group. The fatty acid may be palmitic, oleic or stearic and various combinations are known to exist; for example, distearyl lecithin, stearyl palmityl lecithin and so on. The commercial lecithins usually include the closely related kephalins.
On saponification the lecithins split more or less readily into choline, the fatty acids and glycerophosphoric acid, and by fusion with alkali nitrate and carbonate they yield alkali phosphate. They occur, free or in combination as lecithoproteins, most abundantly in certain animal tissues, but there are also vegetable lecithins. The lecithins of commerce are obtained usually from yolks of eggs or from calves’ or sheep’s brains.
Numerous processes have been devised for the preparation of lecithin from egg-yolk or animal tissue. From egg-yolk it may be obtained by making an alcoholic extract and precipitating by cadmium chloride. The precipitate is washed with alcohol and ether, mixed with 80 per cent. alcohol and warmed with the proper amount of ammonium carbonate to remove the cadmium. After filtering hot and concentrating the filtrate the lecithin is thrown down by cooling to a low temperature—10 C. or below. The precipitate is taken up in chloroform and reprecipitated by acetone.
From tissues it is obtained by extracting with warm alcohol and ether, concentrating the extract, precipitating with acetone and repeating the operations.
Pure lecithin is white, but the commercial preparations are yellowish-brown wax-like solids, which are not soluble in water but form milky emulsions which exhibit the myeline figures under the microscope. The solubility in cold alcohol or ether is slight, but heat aids it. Lecithins are not soluble in acetone. They are hygroscopic and the water mixtures undergo decomposition on standing. They darken on exposure to air and light.
The alcoholic solution is precipitated by platinum or cadmium chloride. It is decomposed by alkalies with the formation of choline and trimethylamine. The ash contains phosphoric acid. The different lecithins contain from 3.84 to 4.12 per cent. of phosphorus and 1.73 to 1.86 per cent. of nitrogen. The ratio of nitrogen to phosphorus should be at 1 to 2.21.
Lecithin is incompatible with alkalies; it should be kept in well-stoppered bottles and should be protected from the light.
The content of lecithin (plus kephalin) in tissues is about as follows:
Per Cent.
Egg-yolk
8 to
12
Egg-white
0.1 to
0.2
Liver
2.0 to
3.0
Kidney
2.0 to
3.6
Lung
2.0 to
3.0
Pancreas
2.0 to
3.0
Actions and Uses.—The lecithin preparations have been recommended in many pathologic conditions, especially in malnutrition and sexual debility. Moderate doses are said to bring about a marked retention of nitrogen and phosphorus, but satisfactory proof of this is lacking. It is extremely unlikely that the small doses which have been recommended in pill or tablet form or in emulsions can have any perceptible action, in view of the fact that many of our natural foods contain much greater weights of available lecithins than the medicinal doses provide. There is no good basis for the statement that the free lecithin has a greater food value or is more readily assimilated than is the substance as found in eggs or tissue. The reverse proposition is much more likely to be true, especially when it is considered that the commercial preparations are usually somewhat altered or decomposed in the process of separation.
Dosage.—Given by the mouth in the form of pills, tablets or glycero-alcoholic emulsions. The amount of actual lecithin ingested in this way is usually small because of the doubtful purity of the original preparation. Several doses, as commonly administered, would be required to furnish the amount of lecithin present in a small egg.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 122.)
PROPRIETARY NAMES FOR LIQUID PETROLATUM
Report of the Council on Pharmacy and Chemistry
The Council has accepted the following report and authorized its publication.
W. A. Puckner, Secretary.
A former report of the Council (Liquid Petrolatum or “Russian Mineral Oil,” Report Council Pharm. and Chem., The Journal, May 30, 1914, p. 1740) called attention to the large number of concerns that were placing on the market liquid petrolatum as a proprietary under coined names. Since then the number of such products has increased. The Council has been requested by several concerns to consider their products put out under proprietary brand names.
The rules of the Council affirm that “the application of ‘trade names’ to official or established nonproprietary substances tends to confusion and fosters many abuses.” In accordance with this general ruling, the Council has invariably refused to countenance proprietary names applied to liquid petrolatum. The Council holds that proprietary or coined names for this substance are detrimental to medical progress, since they are sure to foster the impression that the particular product is different from liquid petrolatum. Manufacturers have been advised that there is no objection to distinguishing their products by the addition of their firm name or the initial representing the firm name; for instance, “Liquid Petrolatum, A. B. and Co.” or “Liquid Petrolatum, Smith.” The Council also believes that such designations as “Star Liquid Petrolatum” or “Liquid Petrolatum, Anchor Brand,” may be regarded as unobjectionable, provided that the words “Liquid Petrolatum” are always used in connection with the brand designation and given equal prominence.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 127.)
SENG
Report of the Council on Pharmacy and Chemistry
The Council has adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
Seng (Sultan Drug Co., St. Louis) is called by the manufacturers:
“... a palatable preparation of Panax (Ginseng) in an aromatic vehicle.”
Regarding ginseng (Panax quinquefolia) the United States Dispensatory, nineteenth edition, page 1603, says:
“The extraordinary medicinal virtues formerly ascribed to ginseng had no other existence than in the imagination of the Chinese. It is little more than a demulcent, and in this country is not employed as a medicine.”
No discussion of ginseng is to be found in the more recently published books on pharmacology, materia medica and therapeutics, evidently because their authors agree with this estimate.
On the other hand, physicians are told through the medium of advertisements appearing in medical journals that Seng is:
“An efficient remedy in all affections in which the gastro-intestinal glands need stimulating.
“Exceptionally useful in atonic indigestion, malnutrition, convalescence from the acute diseases, and all digestive disorders characterized by deranged or depressed functions.” (Woman’s Medical Journal, July, 1914.)
According to the label, Seng is indicated in “indigestion,” “malassimilation,” “malnutrition” and “wasting diseases.” It is also stated—though the preparation is admitted to contain 18 per cent. of alcohol—that to give babies “ten to fifteen drops in water or milk during feeding” is a proper procedure and that “For Colic, Flatulency, etc., the dose for an adult or child may be repeated every half hour until relieved.”
The following are some of the exaggerated therapeutic claims made for this preparation of a worthless drug:
“As a result of its administration the gastro-intestinal secretions are augmented, the digestion of food is substantially increased, and fermentative processes are promptly overcome.”
“Seng will specifically encourage the secretion of the juices in the entire alimentary tract ...”
The formula furnished for Seng is non-quantitative and therefore meaningless. The preparation is exploited in a manner to encourage its ill-advised use by the public, and exaggerated and unwarranted therapeutic claims are made for it. The use of an inefficient or worthless drug like ginseng, moreover, is detrimental to rational therapeutics. The Council therefore voted that Seng be refused recognition for conflict with Rules 1, 4, 6 and 10.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 129.)
FROSST’S BLAUD CAPSULES
Report of the Council on Pharmacy and Chemistry
Frosst’s Blaud Capsules and Frosst’s Blaud, Arsenic and Strychnine Capsules were submitted to the Council by C. E. Frosst & Co., Montreal, Canada. This firm claims, on the authority of the report of a firm of analytical chemists, that:
“... of three leading Blaud preparations bought by us on the open market, the iron in Frosst’s Blaud Capsules showed the highest percentage of Ferrous carbonate.”
The Chemical Laboratory of the American Medical Association found this claim unjustified. The laboratory reported that there was no especial difference in the ferrous iron content of the various Blaud pills found on the market, and that among ten specimens examined, the total iron content was the lowest in the Frosst specimen. In view of this the Council refused recognition to Frosst’s Blaud Capsules and Frosst’s Blaud, Arsenic and Strychnine Capsules.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 164.)
TYREE’S ELIXIR OF BUCHU AND HYOSCYAMUS COMPOUND
Report of the Council on Pharmacy and Chemistry
Each dessertspoonful of this preparation is said to represent
Buchu Leaves
3
1⁄2grains
Uva Ursi
1
1⁄8grains
Pareira Brava
1
1⁄8grains
Hyoscyamus
1
1⁄2grains
Hops
1
1⁄2grains
Acetate Potash
7
1⁄2grains
Spirits Nitre
5
grains
Alcohol5 per cent. (by volume)”
The manufacturer, J. S. Tyree, Washington, D. C., offers this formula to the medical profession with the following claim:
“Approximate composition made [sic] by quantitative and qualitative analysis of the finished product.”
It is also claimed that
“An even greater advantage of Tyree’s Buchu and Hyoscyamus Compound over other drugs, lies in the fact that every constituent of the former is required to conform to a fixed standard of active principle strength; hence the results derivable from it are absolutely uniform.”
These pretentious claims of scientific accuracy look rather absurd to chemists. Many of the substances present in buchu, hops, hyoscyamus, uva ursi and pareira brava are also present in other drugs; hence it would never occur to a pharmaceutical chemist to try to ascertain the composition of such a mixture as Tyree’s Elixir by “quantitative and qualitative analysis of the finished product,” much less to determine the “active principle strength” of each ingredient, for no methods are known by which this can be done.
It is claimed that, because of the care exercised in making Tyree’s Elixir
“... the results derivable from it are absolutely uniform.”
A moment’s reflection, however, must compel any physician to attribute this statement, on the most charitable construction, to sheer ignorance. Of course, even a definite chemical principle, such as quinin, does not exert uniform clinical action, for clinical conditions vary, and accordingly the patient may or may not be cured. It is simply preposterous to claim that the clinical results obtained from such substances as hops, pareira brava, buchu and uva ursi are absolutely uniform.
A peculiarly vicious claim is that the elixir renders the mucous surfaces of the genito-urinary tract “hostile to the multiplication of the gonococci.” Since infection with the gonococcus produces the direst results, any claim which means in plain English that the remedy assists in producing a cure or in preventing infection with that organism cannot be condemned too strongly. Uva ursi, to be sure, has some slight antiseptic action but it is devoid of any curative action in gonorrhea and the minute amounts that are present in the Tyree elixir are of no more protective value against gonorrheal infection than a grain of hexamethylenamin would be.
It is further claimed that the elixir is a “specific” for “Inflammation of the Bladder, Bright’s Disease, Renal Colic, Suppurative Nephritis, Acute Cystitis, Urethritis, Catarrh of the Bladder [it would be interesting to know what distinction the manufacturer draws between ‘Inflammation of the Bladder,’ ‘Cystitis’ and ‘Catarrh of the Bladder’], Acidemia, Edema, Vesical Catarrh of Old Age, Lithemia” and that ascites and anasarca “can be reduced greatly to the satisfaction of the patient, and honor of the physician” by using a mixture of Tyree’s Elixir and infusion of digitalis. Such claims as these do not merit serious discussion, for they carry their own refutation.
It is recommended that Tyree’s Elixir of Buchu and Hyoscyamus Compound be held in conflict with Rules 5, 6 and 10 and that publication of this report be authorized.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 167.)
HYDROLEINE
Report of the Council on Pharmacy and Chemistry
Hydroleine (Charles N. Crittenton Company, New York) is a cod liver oil emulsion said to contain 45 per cent. of cod liver oil, a trace of salicylic acid and 181⁄2 grains of “Pancreatin, Etc.,” per ounce. The advertising claims are based largely on the theory that cod liver oil is “that particular fat which dietetic experience and physiological chemistry have proved to be most digestible.” As a matter of fact, while the superior digestibility of cod liver oil over other oils has often been asserted, neither “dietetic experience” nor “physiological chemistry” have “proved” this by definite observations. The Crittenton Company claims that it is more readily split than other oils. This is probably not true, easy emulsification of the raw oil being often confounded with easy splitting. This latter claim, however, is offered in justification of the name “Hydroleine,” which the Crittenton Company interprets as “hydrated oil.” A circular wrapped around the bottle contains the assertion that “Cod Liver Oil has long been held in high esteem by the medical profession for the treatment of a large number of serious diseases.” This recommendation is likely to lead the public to place undue reliance on Hydroleine in the grave conditions mentioned.
The preparation is in conflict with the rules of the Council inasmuch as its name does not indicate its composition, unwarranted therapeutic claims are made for it, and the exploitation is likely to give the public unwarranted confidence in its value. The Council therefore held Hydroleine ineligible for New and Nonofficial Remedies.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 171.)
CURATIVE VACCINE, BRUSCHETTINI
Report of the Council on Pharmacy and Chemistry
Curative Vaccine, Bruschettini, manufactured by A. Bruschettini, Genoa, Italy, is claimed to have the properties “of acting directly on the tubercular bacillus, bringing directly into the field and determining a hyperproduction of antibacillar and antitoxic substances.” The use of the preparation is said to be indicated in “all forms of tuberculosis.”
A referee reported to the Council that he had examined the available information and believed that the use of this product had no satisfactory experimental basis. The method of preparation appears to be based more on theoretical considerations than on experimental basis.
On the recommendation of the Committee on Serums and Vaccines the Council voted that Curative Vaccine, Bruschettini, be not accepted because (1) the method used for the production of the vaccine was not satisfactorily stated; (2) the theory on which its use is based has not been satisfactorily confirmed, and (3) the value of the product is not upheld by satisfactory clinical evidence.
The Council’s findings, in accordance with its procedure, were sent to the manufacturers for comment. His reply was considered by a new referee who found that the matter presented did not warrant a revision of the Council’s conclusions. Accordingly the Council directed publication of its findings.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 176.)
STEARNS’ WINE
Report of the Council on Pharmacy and Chemistry
Frederick Stearns & Co. market a preparation known as “Stearns’ Wine,” “Stearns’ Wine of Cod Liver Ext. with Peptonate of Iron,” and as “Vinum Ext. Morrhuae, Stearns.” The constituents are said to be “concentrated extract of fresh cod livers,” “Peptonate of Iron” and a “fine quality of prime Sherry Wine” containing 18 per cent. of alcohol.
This preparation was at one time marketed through the medical profession, but is now advertised direct to the public in typical “patent medicine” style. The label on a recently purchased bottle of Stearns’ Wine contains the following statements:
“STEARNS WINE is an ideal tonic for elderly people, for weak, pale and delicate children and convalescents.
“STEARNS WINE has for many years been successfully prescribed in the treatment of general or nervous exhaustion, anemia, malnutrition, loss of appetite, loss of sleep, faulty circulation and impoverished blood supply.”
The scope of the recommendations for the preparation is further indicated in a booklet accompanying the bottle, which begins:
“STEARNS’ WINE, What It Is and Why It Is Good for You.”
The conclusion is:
“STEARNS’ WINE is a safe medicine for the young, middle-aged and old. It is a safeguard to the family health.”
It is not necessary to discuss either these all-embracing claims as to the therapeutic efficacy of the mixture or the fallacies presented in favor of cod-liver extract and peptonate of iron. The Council reaffirms the opinion that whatever therapeutic value cod liver may have resides chiefly, if not entirely, in its fatty constituents (The Journal, Oct. 9, 1909; Reports Council Pharm. and Chem., 1909, p. 115). A confirmation of this opinion has recently been furnished by the investigations of Prof. J. P. Street (The Journal A. M. A., Feb. 20, 1915, p. 638) of several cod liver cordials, one of which (Vinol) like Stearns’ Wine, is described as a wine of cod liver extract with peptonate of iron.
Stearns’ Wine is essentially an alcoholic stimulant. It is not “a safe medicine for the young, middle-aged and old.” The unwarranted therapeutic claims and the recommendations for its indiscriminate use bring it into conflict with Rules 4 and 6. The Council voted that Stearns’ Wine be held ineligible for inclusion in N. N. R.—(From Reports of Council on Pharmacy and Chemistry, 1915, p. 177.)
PROTONUCLEIN AND PROTONUCLEIN BETA
Report of the Council on Pharmacy and Chemistry
The Council had adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
Protonuclein, with other products of Reed and Carnrick, was examined by the Council in 1907 and found ineligible for admission to New and Nonofficial Remedies. According to the patent specifications, it is prepared “from the thyroid and thymus glands, brain (pineal glands and pituitary body), bone-marrow, pancreas, spleen, liver, salivary glands, Brunner’s glands, Lieberkühn’s follicles and peptic glands.” These various glandular bodies, it is said, are dried at a temperature below 130 F. (preferably between 100 and 110); the fat is removed by ether, the dried glands disintegrated, the connective tissue removed by sifting and the resulting powder coated with an ether solution of benzoin and mixed with milk sugar. The dose is three to ten tablets (9 to 30 grains) daily.
Protonuclein Beta is said to be produced by the addition to Protonuclein of an equal amount of nucleoplasm and protoplasm of the spleen. The dose is from two cubes (each 5 grains) three times a day to three cubes four times a day (30 to 60 grains daily).
Special advantages over ordinary nuclein are attributed to Protonuclein, in which, it is claimed, certain unaltered cells remain that are more easily assimilated by the leukocytes than are ordinary proteins, thus leading to a multiplication of cells. In the early advertising Protonuclein was claimed to be:
“... an exact physiological product derived from the lymphoid structures of the body without the use of chemical agents.... So delicate is Protonuclein that any chemical agent is liable to disturb its cellular activity.”
After its examination of the product in 1907 (The Journal, Oct. 5, 1907, p. 1198), the Council concluded that any distinction between the action of Protonuclein and that of ordinary nuclein was purely speculative and highly improbable. “If the active ingredients are really so unstable that they are destroyed by all chemical agents, as claimed, it seems impossible that the activity would be preserved when Protonuclein is given by mouth and therefore subjected to the very profound changes of digestion.”
At that time the importance of thyroid as an ingredient had not been emphasized. The following year, however, Hunt and Seidell (The Journal A. M. A., Oct. 24, 1908) reported the results of an investigation which showed that Protonuclein was a diluted thyroid preparation, as skilfully disguised as in the antifats Rengo and Marmola. Hunt later pointed out (The Journal, Feb. 1, 1913, p. 384) that the amount of nuclein contained in a dose of Protonuclein probably would not have the slightest effect, especially when given by mouth.
The following are extracts from the Protonuclein advertising matter:
“For cancer, infectious fevers (measles, scarlet fever, typhoid and septicaemia) and as a prophylactic.”
“Protonuclein: An ideal prophylactic for all infectious Diseases.”
“A true alterative and tissue builder.”
“The value of Protonuclein depends upon its ability to increase cell power and promote tissue strength. It is therefore needed whenever the organism is below the normal standard, more especially in Anaemia, Typhoid, Neoplasms and as a Prophylactic.”
All the foregoing claims and recommendations are supposed to be based on certain alleged discoveries which the Council has previously characterized as “a tissue of vague speculations ... in direct conflict with the known facts of physiologic chemistry.” As for the third claim, Hunt and Seidell have commented on the danger of recommending thyroid, the most powerful tissue-destroying drug known, as a “tissue builder.”
Protonuclein Beta, it is said:
“... combines the reconstructive action of Protonuclein with the action of the vital principle of the spleen, making it a distinct product for use in all tubercular troubles, including phthisis, localized joint affections and scrofular conditions.”
This product, according to the manufacturers, is based on the work of a certain Dr. Bayle of Cannes, France. Dr. Bayle said that he had treated tuberculous patients with fresh ground up spleen of hogs (25–100 grams per day), mixed with fruit preserve or bouillon; in cases in which this brought on gastro-intestinal disorders, extract of the spleen pulp was administered hypodermically. Bayle reported extraordinary improvements in the physical and mental conditions of his patients even after a few days of this treatment; over 90 per cent. of his tuberculous patients, according to him, improved or were cured. This applied to all types and stages of tuberculosis in man. “With the spleen pulp treatment tuberculous glands disappear like syphilis lesions on administration of mercury and iodids.”
This “spleen specific” of Bayle lacks scientific foundation; Bayle’s own cases were not adequately controlled, and no notice has been taken of Bayle’s report by experts on tuberculosis. Hence it practically lacks both confirmation and contradiction.
The spleen is invaded by tubercle bacilli quite as frequently as are the kidneys and the liver; it has no special toxic action on these bacilli. Nor is there any reason to believe that the end products of gastric and intestinal digestion of spleen pulp, after absorption into the blood, exert such toxic action. It cannot be assumed that these end products indirectly aid the healing processes through improved metabolism, for there is no evidence that they have any specific nutritive or stimulating action after such absorption. Altogether, what we know of the physiology and pathology of the spleen does not warrant us in looking for a “specific” against tuberculosis in this organ.
If, however, the known facts did justify any hope that the spleen might furnish such a specific, manufacturers would not be warranted in exploiting or physicians in prescribing spleen products as a remedy for tuberculosis until control experiments on animals had confirmed the therapeutic value of these products. In a chronic disease like tuberculosis, no conclusions that are scientifically valid can be drawn from clinical cases until many cases have been observed for years under suitable conditions. Right here it may be said that the clinical “evidence” offered in favor of Protonuclein Beta is worthless. The observations which have been reported on this product are not such as to permit any valid final conclusions to be drawn with regard to its value.
The rational method of proving the worth of an alleged new specific such as this is by animal experimentation. So far as we know, neither Dr. Bayle nor the Reed and Carnrick company has performed any such experiments with “spleen pulp” or Protonuclein Beta; nor are we aware that any competent investigator has done so. There is, to the best of our knowledge, no scientific evidence on which to base the claims for Protonuclein Beta.
The Council reaffirms its former action with regard to Protonuclein. The objections made to Protonuclein apply with equal force to Protonuclein Beta. In view of the lack of evidence, the claims for Protonuclein Beta are unwarranted and the product is ineligible to N. N. R. on account of noncompliance with Rules 1, 6 and 8.—(From The Journal A. M. A., Jan. 1, 1916.)
HYDROPSIN
Report of the Council on Pharmacy and Chemistry
The Council has adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
Hydropsin is marketed by the Ernst Bischoff Company, Inc., New York. Its composition is thus described:
“Hydropsin is the standardized dialysate of Digitalis purpurea, Betula alba, Scilla maritima, Juniperis communis, and Herniaria glabra; or, stated otherwise, it is the juice of these drugs, dialyzed and physiologically standardized.”
“Each fluid dram represents Digitalysatum 7 gtts., and 2 gtts. each of the dialysates of Betula, Herniaria, Juniper and Scilla.”
The composition of Hydropsin must be considered essentially secret since the amounts of the several constituent drugs in a given amount of “dialysate” are not disclosed. The active principle of juniper is a volatile oil which is practically insoluble in water; it is difficult to believe that the “juice” of juniper submitted to dialysis could contain any material amount of the active constituent. No information is given as to the method used whereby the several dialysates are “physiologically standardized.” It therefore remains to be proved that the manufacturer of Hydropsin possesses any method whereby the dialysates of juniper (Juniperis communis), birch (Betula alba, the common European birch) and knot weed (Herniaria glabra) are so standardized. The claim is made that:
“Herniaria has long been recognized as one of the most valuable drugs in the treatment of dropsical affections due to cardiac impairment.”
On the contrary, Herniaria belongs to that large class of drugs which have been tried, found wanting and abandoned. It is a very old remedy, and the claims made for it are an inheritance from the early herbalists, with whom it was very popular. According to King’s American Dispensatory, it was “principally employed to cure hernia (hence its name) and to increase the flow of urine. It was also said to increase the flow of bile.... Internally and externally, it was praised in snake-bites, and the powdered plant was employed to kill maggots on unhealthy sores of horses. It was reputed to ‘crush’ and expel calculi from the kidneys and bladder....”
The Ernst Bischoff Company says that:
“Betula exerts both an antiseptic and stimulating influence on the urinary passages and is particularly serviceable where a catarrhal condition of the bladder exists. When combined with other diuretics, as in Hydropsin, the drug affords highly satisfactory results in the treatment of ascites, cardiac dropsy and hydrothorax.”
Birch is another drug which has been discarded. Few textbooks on materia medica even mention it. That it can materially affect the action of such powerful drugs as squill and digitalis is exceedingly doubtful.
An unwarranted implication—that in this preparation the powerful drugs digitalis and squill have been deprived of their dangerous qualities—is the assertion:
“Dialysis, removing all resins and colloidals, results in better tolerance on part of sensitive patients, and in more rapid absorption and elimination; which, in turn, means early therapeutic effects and little or no fear of toxic accumulation.”
That removal of colloids and resins materially affects the tolerance of these drugs is improbable. To claim that because of their removal, there need be “little or no fear of toxic accumulation” is utterly without warrant. The claim that one preparation containing digitalis is less likely to produce cumulative effect than any other digitalis preparation is contradicted by a mass of evidence.
It is claimed that Hydropsin affects “favorably all forms of dropsy or Edema that are at all amenable to medical treatment.” There can be no question but that squill and digitalis, or, better, either singly, used in suitable cases, may relieve dropsical effusions; but to claim that such a complex mixture as Hydropsin can favorably affect all forms of dropsy that are amenable to medical treatment is on its face unwarranted.
The claim is made that:
“By reason of its unusual potency and relative harmlessness, Hydropsin may be employed to great advantage in all cases where it is desirable to increase the volume of urine without injury to the renal structures.”
On the basis of the claimed composition, the action of Hydropsin must be essentially that of digitalis or of digitalis and squill. Consequently, if it possesses “unusual potency,” it cannot possess “relative harmlessness,” and vice versa. Neither digitalis nor squill should be employed “in all cases” of nephritis, even if it is “desirable to increase the volume of urine.”
The composition claimed for Hydropsin brands it as an irrational mixture in which potent drugs are combined with, and more or less covered up by, others that are obsolete and inefficient. The name, instead of indicating its composition, suggests diseases in which it may be thoughtlessly and indiscriminately used. The claim that the danger of toxic or cumulative action has been removed, if accepted by physicians, tends to uncritical use with possible disastrous results. Hydropsin is ineligible for New and Nonofficial Remedies because of conflict with Rules 1, 2, 6, 8 and 10.—(From The Journal A. M. A., Jan. 8, 1916.)
DIGITALYSATUM
Report of the Council on Pharmacy and Chemistry
The Council has adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
Digitalysatum is sold in the United States by Ernst Bischoff Company, Inc., New York. The firm claims that it is a dialysate prepared from the juice of freshly gathered digitalis, containing all the active principles, and representing the fresh plant weight for weight. It is said to be standardized physiologically and to contain 12 per cent. alcohol. Sterisol-Digitalysatum, intended for injection, appears to be the “dialysate” without alcohol, diluted with equal parts of physiologic sodium chlorid solution. The Council some years ago found both products ineligible for New and Nonofficial Remedies because of unwarranted therapeutic claims. The preparations are still being advertised to physicians under claims which imply superiority to all other digitalis preparations. For instance:
“Digitalysatum is the diuretic par excellence in cardiac insufficiency ...”
“Digitalysatum as a diuretic and cardiac stimulant is in a class by itself, being quick of action, uniform in strength, and well tolerated.”
“Digitalysatum differs from other forms of digitalis in these respects:... Digitalysatum is free from fat, resins and colloids, and is therefore well-borne by sensitive patients—the young and the feeble—and is quickly absorbed and eliminated....”
The Council has elsewhere28 expressed the conviction that tincture of digitalis produces the full therapeutic effects of digitalis; that, when properly made, the tincture is as stable as any liquid preparation of digitalis now available, and that attempts to enhance the reputation of proprietary products by exaggerating the disadvantages of the official preparation are to be deplored. No adequate evidence is offered of the claimed superiority of action of Digitalysatum.
By implication, the claim is made that Digitalysatum is superior to other digitalis preparations in respect to toxicity:
“Free from fat, resins and colloidals, it is always well borne and is quickly absorbed and eliminated. No case of toxic accumulation (faulty elimination) has ever been reported.”
That Digitalysatum is free from the dangers of toxic cumulation is highly improbable; in fact, it is inconsistent with the statement that the preparation contains all the constituents found in the fresh plant. Even if instances of cumulative action have not been reported this does not prove that such cumulative action does not occur. The tincture of digitalis has the systemic side-effects of digitalis in no greater degree than the various proprietary preparations. Attempts to create the impression that Digitalysatum possesses all the virtues of digitalis without its chief disadvantage are to be condemned as likely to lead to incautious use of the preparation.
These exaggerated claims are in the main made indirectly, but they are none the less inimical to sound therapy. The Council therefore declared Digitalysatum ineligible for New and Nonofficial Remedies and voted that this report be published.—(From The Journal A. M. A., Jan. 8, 1916.)
SO-CALLED SECRETIN PREPARATIONS
Report of the Council on Pharmacy and Chemistry
The Council authorized the following report for publication, and voted to endorse the work of Professor Carlson discussed therein.
W. A. Puckner, Secretary.
The Council has not accepted for inclusion in New and Nonofficial Remedies any preparations said to contain secretin or prosecretin as their active ingredient. A report giving the reasons for the rejection of one (the first of the so-called secretin preparations marketed) was published early last year;29 an article on secretin, based on work undertaken at the request of the Council on Pharmacy and Chemistry, is now published.30
Lest the appearance of this detailed study of secretin, after the rejection of so-called secretin preparations, should be interpreted (as manufacturers whose products have been rejected have endeavored to interpret such action) as a case of first condemning a preparation and then getting the facts, the Council’s methods, and their application in this case, may be briefly stated. The Council maintains that, when a manufacturer places a product on the market, the burden of proof is on that manufacturer to show that the properties of his product are in accordance with his claims for it. As stated in the introduction to N. N. R., “it is ... manifestly impossible for the Council to investigate the composition of every complex pharmaceutical mixture, or to check thoroughly every therapeutic claim; it can give only an unbiased judgment on the available evidence.” Acting on this principle, the Council examined the claims made for Secretogen, an alleged secretin product manufactured by the G. W. Carnrick Company. The conclusion was that these claims were in absolute conflict with the available evidence as to the action of secretin.
It is not necessary to review this subject again. It will suffice to state that the claims made for Secretogen rest on two fundamental propositions: (1) that deficiency of secretin (or, rather, of prosecretin) in the intestinal mucosa is a factor in gastro-intestinal diseases; (2) that secretin given by the mouth is absorbed and produces increased secretion of the pancreatic and intestinal juices and of the bile.
From an examination of the evidence available, including that submitted by the manufacturers, the Council concluded: “1. No evidence has been presented that the absence of secretin is a cause of gastro-intestinal disease. 2. There is no evidence that secretin in any form is physiologically active when administered by mouth.” That these conclusions were justified is shown again by the review given by Carlson of the literature, much of which was also reviewed in the Council’s previous report.
Since the claims of the Carnrick Company were not supported by any satisfactory evidence, no further investigation on the Council’s part was necessary to warrant rejection of the product. The Council did not undertake to determine, for instance, whether or not Secretogen and similar products actually contain secretin; the determination of this point was immaterial here, in view of the conclusiveness of the evidence that secretin given by mouth has no physiologic action.
Since firms other than the G. W. Carnrick Company are manufacturing alleged secretin preparations, and since recommendations for the use of secretin preparations in gastro-intestinal diseases have even crept into textbooks, it seemed desirable to obtain further information on certain points. The Council therefore requested Prof. A. J. Carlson of the University of Chicago to check the results of previous investigators with regard to the action of secretin administered by mouth or directly into the intestine, and, in addition, to investigate the secretin content of certain alleged secretin preparations.
Carlson and his co-workers, like all previous investigators, found that secretin given by mouth, or introduced even in enormous doses directly into the intestine, is entirely inactive. They also found that marked destruction of secretin followed contact for one minute with human gastric juice and that secretin is rapidly oxidized and rendered inert in contact with the air.
Further, they were unable to demonstrate the presence of secretin in samples of Secretogen and another supposed secretin preparation (Duodenin) bought on the open market. In the case of Secretogen there was one exception: one bottle was found which contained a little secretin, but it was necessary to administer (by intravenous injection, of course) the entire contents of the bottle (100 tablets) to obtain “a small but unmistakable secretin reaction.”
In these studies the methods employed were those by which secretin was discovered. It is only by the use of such methods that the presence or absence of secretin can be determined. Apparently the manufacturers who place so-called secretin preparations on the market do not make use of these methods, by which alone even the composition of their products can be determined.
Carlson and his collaborators conclude:
“There is as yet no reliable evidence that lack of secretin is a primary or important factor in any disease. Even should this be established, secretin therapy, to be effective, must be intravenous. Secretin has not yet been prepared in sufficiently pure state to render possible intravenous injection in man without injurious effects. And even when this is attained, the very fleeting action of secretin will in all probability render secretin therapy as futile in all the diseases in which it is theoretically indicated as epinephrin therapy is in Addison’s disease.”
In short, secretin is as ineffective taken by mouth as it would be rubbed on the skin.
The referee recommends that the work of Professor Carlson be endorsed.—(From The Journal A. M. A., Jan. 15, 1916.)
HAS SECRETIN A THERAPEUTIC VALUE? [B][C]
A. J. Carlson, Ph.D., J. E. Lebensohn, M.S., and S. J. Pearlman, B.S.
Chicago
It is well established that acid chyme in the duodenum is the normal stimulus to the secretion of pancreatic juice.31 Interaction of the acid with the duodenal mucosa liberates into the blood stream a substance which, circulating through the pancreas, excites the latter to activity. This exciting substance has been termed “secretin.” It can be prepared artificially by macerating duodenojejunal mucosa in 0.4 per cent. hydrochloric acid, neutralizing the boiling mixture, and filtering. A few cubic centimeters of the filtrate injected into a vein produce invariably a powerful secretion of pancreatic juice.32 That a “chemical messenger” is at the basis of the duodenal acid reflex has been proved by even more crucial experiments—transfusion (Wertheimer,33 Enriquez and Hallion34), cross circulation (Fleig,35 Matuso36), and perfusion of the isolated pancreas (Huston37).
PROPERTIES OF SECRETIN
Prosecretin.—Secretin is soluble in water, yet a watery extract of intestinal scrapings is without action,32 even after being submitted to acid treatment.38 Starling therefore holds that secretin exists in the intestinal mucosa in an inactive form, as “prosecretin.” The content of the intestine in prosecretin decreases from the duodenum down, so that one is unable to demonstrate any prosecretin in the last 21⁄2 feet of the ileum. Prosecretin is insoluble in water, acetone, absolute alcohol or ether. Secretin, on the other hand, is readily soluble in water, normal salt solution and diluted alcohol (70 per cent.), but likewise insoluble in absolute alcohol and ether.
Preparation.—All of the more dissociated acids liberate secretin from intestinal mucosa on boiling. Their action is dependent on the degree of dissociation,39 carbonic and boric acids being inactive.40 Secretin can also be prepared with strong soaps (from 10 to 30 per cent. sodium oleate), alcohol (70 per cent.,41 0.6 per cent. sodium chlorid36). The acid and soap in the duodenum produce secretion; there is no necessary correspondence between the action of a substance in the intestine and that obtained by injection after boiling mucosa with it. The sodium chlorid, bile, maltose and glucose produce some secretion by the latter method yet none by the former.36 On the other hand, ether, chloral and oil of mustard excite secretion when in the intestine, but no secretin can be prepared from boiled mucosa by their action. The irritation of the lining cell has produced the necessary hydrolysis.38 In well-controlled experiments, Wertheimer and LePage42 found that after the introduction of acid, secretion is secreted into the lumen of the intestine. Matuso36 confirmed their results, and found this a satisfactory method for the preparation of secretin. It is said that secretin can be obtained by merely boiling the mucosa with water, but the results are inconstant.43
Action.—Secretin is an excitant not only of the pancreatic juice but also of the liver and the intestinal mucosa. The flow of bile is markedly accelerated (Henri and Portier,44 Enriquez and Hallion45), likewise that of succus entericus (Delezenne and Frouin,46 Bottazzi and Gabrielli47), and intestinal peristalsis is stimulated (Enriquez and Hallion,48 Falloise49). Injections of secretin produce a marked vasodilatation, but the secretory effect is independent of the blood pressure changes. The pancreas is not readily fatigued by secretin. Bayliss and Starling50 have obtained undiminished flow after eight hours of continuous injection. Our experience confirms this result. Also, equal doses of secretin give corresponding results at various intervals. Moreover, anesthesia does not affect the flow. Secretin is unrecoverable from the glands even after two hours of continuous injection.51 The juice obtained by secretin has been subject to many studies.52 It is of high alkalinity (about seventh normal), contains all the pancreatic ferments, and corresponds in all respects to the juice obtained in digestion from permanent pancreatic fistulas.53
Specificity.—In a maceration of the duodenojejunal mucosa, such as we have in secretin, the known substances are proteoses and peptones, acid amins, bile salts, beta-imidazolethylamin, cholin, gelatin and inorganic salts. These substances, individually and severally, together with their derivatives, are devoid of secretory action. Chemically, secretin, is then a specific entity. But like epinephrin, in its distribution, it is nonspecific. Active preparations have been made from an extraordinary variety of animals among the different classes of vertebrates (Camus,54 Bayliss and Starling,55 Chapman56). It is likewise found in the new-born and in the fetus.57 Its action, however, like its chemical composition, is markedly specific. It stimulates the flow of pancreatic juice, bile and succus entericus. Its effect on the gastric glands is negative, and on the saliva likewise.58 On the other hand, no other extracts produce pancreatic secretion. Dr. Koch, who, in collaboration with Dr. Keeton and Dr. Luckhardt, has done the most recent work on gastrin59 (a substance that most nearly resembles secretin) and has isolated an extremely active preparation, finds that gastrin injection has likewise no effect on the pancreas. Camus and Gley,60 with crude preparations, had previously obtained a similar result.
Lability.—Neutral secretin is but feebly attacked by a temperature of 100 C. If heated in an autoclave (so as to prevent oxidation), this temperature can be continued for thirty minutes without any change in its activity. Increasing the temperature increases the speed of destruction, so that at 140 C. the destructive action is marked.61 Autoclaving at 15 pounds for fifteen minutes, as an ordinary sterilization of culture mediums, produces, we found, a distinct though not serious decrease in activity. Secretin acidified to fifth-normal with hydrochloric acid loses 60 per cent. of its activity on fifteen minutes boiling. Secretin, alkalinized to fifth-normal with sodium hydroxid loses 95 per cent. of its activity in five minutes’ boiling; decreases to a trace in thirty minutes, and disappears entirely in sixty minutes. At room temperature, with fifth-normal alkalinity, 80 per cent. of secretin is destroyed in eight hours.61 The destruction probably means a secondary cleavage of the secretin molecule itself.
Secretin is oxidized readily. If left standing uncovered for a summer’s day, the preparation will be inactive.51 Even if kept in the ice-chest (no other precaution being taken), its activity is lost in a very few days. Sunlight undoubtedly hastens the oxidative process. If care is taken as to sterility, however, and the secretin is kept in the ice-chest, well stoppered and in a dark flask, it will retain its activity for several weeks.
Dixon and Hamill51 claimed that secretin disappears quantitatively on passage through a Berkefeld filter at 5 mm. pressure. Lalou,62 using higher pressure, was unable to confirm the finding, but obtained a marked decrease in activity. Our results are in accord with those of Lalou.
Analogy to Epinephrin.—The analogy of secretin to epinephrin does not generally receive enough emphasis. Both substances are nonspecific in distribution, but specific chemically, and especially physiologically, epinephrin acting on the myoneural junctions, secretin on intestinal digestion. They are both relatively simple substances of low molecular weight, and subject to rapid oxidation whereby their properties disappear. The action in both cases is very transient. They are the two examples of what Starling calls the “acute hormones,” in which it is essential that reaction take place immediately, and shall disappear as soon as the exciting cause is removed.63
CLINICAL USE OF SECRETIN
Diabetes Mellitus.—Moore, Edie and Abram64 were the first to suggest a therapeutic value for secretin, having obtained favorable results with secretin administration in diabetes. They argued that the internal secretion of the pancreas may be stimulated by secretin, and that some cases of diabetes may be due to lack of this necessary excitant. Owing to the importance of the question, their announcement was followed quickly by numerous investigations by other observers. Previously, Spriggs, at the suggestion of Starling, had tried intravenous injections of secretin free from depressor substance in a diabetic patient, and had obtained negative results. Moore, Edie and Abram gave their secretin by mouth over long periods. Of the five cases cited in their first paper, two were negative. The third was that of a man, aged 25, who received daily 30 c.c. of secretin. After a latent period of three weeks, the sugar suddenly fell, and after four months the urine was sugar-free. Six months later a relapse occurred with the development of phthisis and death. The other two patients were a boy, aged 7, and a girl, aged 9, whose urine in from three to five weeks became sugar free during the secretin treatment in spite of severe diabetes. One of these patients later relapsed.65 Bainbridge and Beddard66 gave secretin a thorough trial in three cases with negative results, and are disposed to attribute the results of Moore to dieting. Dakin and Ransom67 cited one case, secretin being given for twelve weeks, with negative results; Foster,65 nine cases, all negative; Charles,68 three cases, all negative. Crofton,69 however, gave secretin a trial in one case with favorable results. Moore, Edie and Abram, in a later paper,70 report a large number of cases tried with the majority of results negative, though in some cases an improvement in the digestion, and in certain cases an increase of weight was noted.
One method of testing the basis of Moore’s theory would be by examining the prosecretin content of the intestine in diabetics. Bainbridge and Beddard found, in the paper referred to,66 that from five of the six cases of diabetics examined postmortem, little or no secretin could be prepared; but in a subsequent report of seven cases,71 they found only one in which the secretin obtained was scanty. The failure to obtain secretin in some cases they claim is probably due to the rapid postmortem degeneration of diabetic tissue. Evans,72 in Starling’s laboratory, found that in dogs made recently diabetic by total pancreatectomy, but little secretin could be obtained. Hedon and Lisbonne,73 and Pemberton and Sweet74 report, on the contrary, that the duodenum of diabetic dogs is rich in prosecretin. Bainbridge and Beddard,71 working on a diabetic cat, likewise found prosecretin to be present in normal quantity.
Digestive Disturbances.—Secretin for digestive disturbance was first used in the “acid duodenal medication” of Enriquez.75 This consisted in the giving of tartaric acid in thick keratin capsules, the acid not being liberated until the duodenum was reached, where it provoked the formation of secretin. “The secretin mechanism,” he says, “is probably capable of pathologic disturbance as would result, for example, with diminished acidity of chyme, disturbance of the normal motility of the stomach or pylorus, or diminished prosecretin in the mucosa. Such a condition would produce disturbance of the pancreatic, biliary and intestinal secretions, and interfere with intestinal movements, with a clinical syndrome of intestinal dyspepsia as a result, among the chief and most constant symptoms of which would be constipation.” “The acid duodenal medication” was submitted to wide clinical use, and very favorable results in certain obstinate cases of constipation were reported. In regard to “diminished prosecretin in the mucosa,” Wentworth76 has claimed that in infantile atrophy such is the condition, but Sweet and Pemberton77 have found that the difficulty of preparing secretin from human duodenums is such as to render Wentworth’s findings inconclusive.
Beveridge78 suggests the use of secretin in (a) pyloric stenosis, (b) pancreatic insufficiency, (c) hepatic stimulation and cirrhosis of the liver (d) to stimulate peristalsis in colonic stasis, (e) in gastro-enterostomy and short-circuiting of the intestines. He claims to have used it in over a hundred cases with “brilliant results,” and cites four typical histories. The G. W. Carnrick Company, which manufactures “Secretogen,” an alleged secretin preparation, cites a number of authorities79 as also recommending secretin for digestive disorders. Harrower, who is or was connected with the Carnrick Company, in clinical journals80 has ardently advocated the use of secretin for a large number of maladies.
PHYSIOLOGIC CONSIDERATIONS
Throughout its clinical use, secretin has been given by mouth; but its direct introduction into the intestine of a dog under anesthesia in even enormous quantities is without effect. This fact, first observed by Bayliss and Starling,32 was confirmed by Fleig,81 and Matuso,36 and our personal experiments have convinced us of its truth. Matuso found that ordinary secretin and that obtained from intestinal lumen gave equally negative results. Large quantities of active secretin, moreover, acidified to 0.2 per cent. hydrochloric acid, and left in the ileum for fifteen minutes, were still negative. Wertheimer and Duvillier,82 in a previous paper on this subject, had likewise found that acid solutions of secretin (which might be considered more normal for the intestine than when neutral), when introduced into the ileum gave negative or inconstant results. They conclude that it is more likely that the pancreas does not respond to such minimal stimuli, than that the secretin is not absorbed.
The destructive action of the digestive enzymes leads us to believe that it is in inactive form that secretin is absorbed. Like epinephrin, it cannot pass through the digestive tract. Bayliss and Starling state that it is destroyed by one hour’s tryptic digestion. Lalou62 worked with the action on secretin of pepsin, dog’s gastric juice, pancreatic juice, succus entericus and erepsin, and found in each case a destructive effect, even almost after mixing; and after five minutes over 75 per cent. of the activity had disappeared. Matuso36 introduced 30 c.c. of active secretin into the intestine, removed it five minutes later, and found that no activity remained.
Other methods of administration have been tried. Subcutaneous injections are practically negative (Matuso,36 Hallion83) and intrapleural injections are likewise negligible (Bayliss and Starling55).
Starling63 finds that continued intravenous injections of secretin in a healthy dog produces after a time severe symptoms of collapse, which, he believes, are due to change in the intestinal mucous membrane caused by the entry and non-neutralization of the strongly alkaline pancreatic juice.
Intestinal digestion seems little affected in achylia gastrica (Stockton,84 Ehrman and Lederer,85 Bayliss and Starling32). This may be due to other secretin stimulants as fats, or to the action of the nervous mechanisms (Meltzer86).
THE DESTRUCTION OF SECRETIN BY HUMAN GASTRIC JUICE
We have carried out in detail experiments on the digestive effect of human gastric juice on secretin. Our results in every respect confirm the findings of Lalou,62 who worked with commercial pepsin and dog’s gastric juice, but are even more striking because of the much superior quality of pure human gastric juice.
Methods.—The human gastric juice was obtained from Mr. V., the gastric fistula case of our laboratory. The chemical and digestive characters of his juice are discussed in a recent paper.87 In the different experiments, different samples of gastric juice were used. The secretin employed was always freshly prepared. Digestion was carried out in the incubator at 38 C. with the reaction of 0.4 per cent. acid, and the end of the period was marked by either boiling the mixture or (in the first two experiments) by turning the mixture alkaline. The action of the preparation, we proved, was not influenced by the method used. The dogs on which the preparations were tested were prepared for carotid blood pressure, injection into the external jugular vein, and cannula in the pancreatic duct, essentially the methods of Bayliss and Starling32 being employed. The preparations were injected at body temperature after being neutralized and filtered. Except for the addition of normal salt solution instead of gastric juice, the control injections of secretin were submitted to exactly the same treatment as the other preparations.
Results.—Our results are embodied in Table 1. We assured ourselves before beginning the series that incubation of secretin with boiled gastric juice produced no change. It is to be noted in the table that each experiment is a unit complete in itself, beginning and ending with a control injection of secretin. Special attention is called to the marked destruction that follows contact of human gastric juice with secretin for merely one minute. In Experiment 4, using 1 c.c. of human gastric juice, the action fell to 14 drops from an original secretion of 21; in Experiment 5, using 8 c.c. of gastric juice, the action fell to 6 drops from an original secretion of 20. Of interest also is the rate at which we get complete destruction of secretin. This is practically 2 hours for 2 c.c. with secretin giving originally 110 drops (Experiment 2, Fig. 1), or 30 minutes for 5 c.c. with a secretin giving originally 53 drops (Experiment 6). These results are practically parallel, though they were obtained with different samples of gastric juice and in different experiments.
TABLE 1.—THE DESTRUCTION OF SECRETIN BY HUMAN GASTRIC JUICE
28 Report on Proprietary Digitalis Preparations, J. A. M. A., Dec. 4, 1915, p. 2024.
29 Secretogen, J. A. M. A., May 1, 1915, p. 1518.
30 Carlson, A. J.; Lebensohn, J. E., and Pearlmann, S. J.: Has Secretin a Therapeutic Value? J. A. M. A. 66:178 (Jan. 15) 1916.
[B] From the Hull Physiological Laboratory of the University of Chicago.
[C] This investigation was undertaken at the request of the Council on Pharmacy and Chemistry. The following report, having been submitted to the Council, received its endorsement (see preceding report of the Council on Pharmacy and Chemistry, “So-Called Secretin Preparations”).
31 Pawlow: The Work of the Digestive Glands, 1912.
32 Bayliss and Starling: Jour. Physiol. 28:325, 1902.
33 Wertheimer: Compt. rend. Soc. de biol. 54:475, 1902.
34 Enriquez and Hallion: Compt. rend. Soc. de biol. 55:233, 363, 1903.
35 Fleig: Arch. internat. de Physiol., 10:206, 1910.
36 Matuso: Jour. Physiol. 45:477, 1913.
37 Huston: Ann. et bull. Soc. roy. de sc. méd. et nat. 70:178, 1912.
38 Starling: Lancet, London 2:433, 1905.
39 Frouin and Lalou: Compt. rend. Soc. de biol., 71:189, 1911.
40 Camus: Compt. rend. Soc. de biol., 1902, 54:442, 1902.
41 Fleig: Jour. de physiol. et de path. gén. 6:32, 50, 1904.
42 Wertheimer and LePage: Jour. de physiol. et de path. gén. 4:1061, 1070, 1902.
43 Stepp: Jour. Physiol. 43:441, 1912.
44 Henri and Portier: Compt. rend. Soc. de biol. 54:620, 1902.
45 Enriquez and Hallion: Presse méd. 1:105, 1903.
46 Delezenne and Frouin: Compt. rend. Soc. de biol. 56:319, 1904.
47 Bottazzi and Gabrielli: Arch. internat. de physiol. 111:156, 1905.
48 Enriquez and Hallion: Bull. gén. de thér. 162:202, 1911.
49 Falloise: Bull. de l’Acad. roy. de Belgique 5:945, 1902.
50 Bayliss and Starling (Note 2). Matuso (Note 6). Arch. internat. de physiol. 10:335, 1911.
51 Dixon and Hamill: Jour. Physiol., 1909, xxxv, 314.
52 Bayliss and Starling: Jour. Physiol., 1904, xxx, 61. Bierry: Compt. rend. Soc. de biol., 1907, lxii, 433. Bierry and Terroine: Compt. rend. Acad. de sc., 1905, cxli, 146. Lalou: Comp. rend. Acad. de sc., 1910, xxix, 824. Morel: Compt. rend. Soc. de biol., 1909, lxvii, 36. Strassano and Billoro: Compt. rend. Soc. de biol., 1902, liv, 937.
53 Bayliss and Starling (Note 23).
54 Camus: Jour. de physiol. et de path. gén. 4:998, 1902.
55 Bayliss and Starling: Jour. Physiol. 29:174, 1903.
56 Chapman: Proc. Linnaean Soc., New South Wales 1:92, 1905.
57 Camus: Compt. rend. Soc. de biol. 61:59, 1906. Hallion and Lequex: Compt. rend. Soc. de biol. 61:33, 1906.
58 Derouaux: Arch. internat. de physiol. 3:44, 1905. Lambert and Myer: Compt. rend. Soc. de biol. 54:1044, 1902. Starling: Lancet, London 2:501, 1905.
59 Keeton and Koch: Am. Jour. Physiol. 37:481, 1915.
60 Camus and Gley: Compt. rend. Soc. de biol. 54:648, 1902.
61 Lalou (Note 21). May: Jour. Physiol. 30:400, 1904.
62 Launoy: Arch. internat. de Physiol. 3:62, 1906. Morel and Terroine: Compt. rend. Soc. de biol. 67:36, 1909. Zunz: Arch. internat. de physiol. 8:181, 1909. Lalou: Jour. de physiol. 14:465, 1912.
63 Starling: Proc. Roy. Soc. Med., 8, No. 4, 1914, Therap. and Pharm. Section, p. 29.
64 Moore, Edie and Abram: Biochem. Jour., 1:28, 1906.
65 Foster: Jour. Biol. Chem., 2:297, 1906.
66 Bainbridge and Beddard: Biochem. Jour., 1:429, 1906.
67 Dakin and Ransom: Jour. Biol. Chem., 2:305, 1906.
68 Charles: Med. Press and Cir., 133:578, 1906.
69 Crofton: Lancet, London, 176:607, 1909.
70 Moore, Edie and Abram: Biochem Jour., 3:82, 1908.
71 Bainbridge and Beddard: Biochem. Jour. 3:82, 1908.
72 Evan: Jour. Physiol. 44:461, 1912.
73 Hedon: Compt. rend. Soc. de biol. 74:375, 1913.
74 Pemberton, Ralph, and Sweet, J. E.: Further Studies on the Influence of the Ductless Glands on the Pancreas, Arch. Int. Med., May, 1910, p. 466.
75 Enriquez: Bull. du Lab. de biol. Appliq. 2, No. 2-No. 8, 1904.
76 Wentworth, A. H.: The Cause of Infantile Atrophy, J. A. M. A., July 20, 1907, p. 204.
77 Sweet, J. E., and Pemberton, Ralph: Experimental Observations on Secretin, Arch. Int. Med., February, 1908, p. 231.
78 Beveridge: Am. Med. 20:255, 1914.
79 Lockwood, G. R.: Diseases of Stomach, 1913, Chapter on Achylia. Bassler, Anthony: Am. Jour. Gastro-Enter., 1914; Kemp, R. C.: Diseases of Stomach, Intestine and Pancreas, 1912. Reed, Boardman: Am. Jour. Gastro-Enter., October, 1912. Ewald (Therapie der Gegenwart, 1915, p. 5) reports favorable results with Secretogen in one of thirteen cases.
80 Harrower: Pediatrics 25:430, 1913; New York M. J. 118:315, 1913; Arch. f. Verdauungskr. 20:577, 1914.
81 Flieg: Arch. gén. de méd. 191:1482, 1903.
82 Wertheimer and Duvillier: Compt. rend. Soc. de biol. 68:535, 1910.
83 Hallion: Presse méd. 20:433, 1912.
84 Stockton: In Osier and McCrae’s Modern Medicine 3:19, 1914.
85 Ehrman and Lederer: Deutsch. med. Wchnschr. 35:879, 1909.
86 Meltzer, S. J.: The Factors of Safety in Animal Structure and Animal Economy, J. A. M. A., Feb. 23, 1907, p. 655.
87 Carlson: Am. Jour. Physiol. 38:248, 1915.
No. of Experiment
Quantity of Gastric Juice Used, C.c.Secretion of Pancreatic Juice in Drops
10 C.c. Secretin Control —Beginning ExperimentThe Secretin After Incubation with Human Gastric Juice
10 C.c. Secretin Control —End of ExperimentDig. Time, Hours
Secretion Rate
Dig. Time, Hours
Secretion Rate
Dig. Time, Hours
Secretion Rate
1
2
28
6
0
4
0
2
0
16
2
2
110
2
7
1
1⁄218
1
18
41
3
2
40
1
7
3⁄47
1⁄48
31
4
1
21
1⁄211
1⁄412
1⁄6014
18
5
8
20
1⁄21
1⁄43
1⁄606
18
6
5
53
1⁄22
..
..
..
..
..
We also tried the effect of keeping the digestive time constant and varying the amount of gastric juice employed. Increasing the quantity of gastric juice used increases the quantity of secretin destroyed (Table 2).
TABLE 2.—EXPERIMENT 7*
Preparation
Pancreatic
Juice Drops
10 c.c. secretin
20
10 c.c. secretin digested with 0.5 c.c. gastric juice
15
10 c.c. secretin digested with 3 c.c. gastric juice
13
10 c.c. secretin digested with 10 c.c. gastric juice
8
* The digestive time was kept constant at fifteen minutes. (The gastric juice used had been diluted with stomach washings.)
The reader will observe in Table 1 that the results obtained from the control injection of secretin at the beginning of the experiment is uniformly greater than that obtained after several injections of digested secretin.
In view of the established fact that equal quantities of secretin can generally be relied on to produce results,62 one might suggest that the injections of the split products of secretin have inhibited to some degree the action of the pancreas. We can submit the data in Table 3 in support of this view, showing among other things that the action of secretin is not influenced by previous injections of inert depressor substances, though it by the injection of the cleavage products of secretin. (The various injections in the experiments were made at about fifteen-minute intervals).
We have carefully analyzed the reaction in blood pressure that follows the injection of the various preparations. We find no constant effect. Digested secretin gives a fall in blood pressure that is at times less, at times equal, and at other times greater (Fig. 1) than that produced by the original preparation.
Besides the bearing that it has on the therapeutic use of secretin, this destructive action of the digestive enzymes is also of prime physiologic interest. Failure to realize it has led to misconceptions as to the intrinsic nature of secretin.
TABLE 3.—EXPERIMENTS 8 AND 9
Preparation
Pancreatic
Juice Drops
Experiment 8:
10 c.c. secretin, five injections of inert depressor substances
29
10 c.c. secretin, two injections of completely digested secretin
28
10 c.c. secretin, eight injections of inert depressor substances
16
10 c.c. secretin
16
Experiment 9:
10 c.c. secretin (control, beginning of experiment)
21
10 c.c. secretin, after thirty minutes incubation with 1 c.c.
boiled gastric juice
27
10 c.c. secretin, after thirty minutes incubation with 1 c.c.
fresh gastric juice
11
10 c.c. secretin (control, end of experiment)
18
The findings of Lalou, confirmed by us, explain the anomaly that has led Delezenne88 to put forward the antisecretin theory.
SECRETIN HAS NO ACTION WHEN GIVEN BY MOUTH
It is a constant claim that so many and complex are the factors concerned in physiologic processes, that it is not unusual for clinical deductions to establish themselves in the face of a priori laboratory dicta. We considered it desirable, therefore, to test the action of secretin, orally administered, in the most direct manner, and the one freest from possible criticism. With this in view, we performed a series of experiments on normal unanesthetized dogs having permanent pancreatic fistulas.
Method.—In the operations for permanent pancreatic fistulas we followed closely the technic developed by Pawlow,89 and with excellent results. The dogs maintain themselves in splendid condition if proper care is taken. This consists in feeding them only with bread and milk, and giving sodium bicarbonate daily. The dogs were given this treatment in the evening so that experimental procedure might be carried on in the day with empty stomach under constant conditions. Freshly prepared secretin in large quantities was given by stomach tube to these dogs, and the response of the pancreas studied and compared with the response obtained from control preparations. The same preparation was generally not given on consecutive days.
TABLE 4.—DETAIL OF TYPICAL EXPERIMENTS
Dogs with pancreatic fistulas, showing that secretin given by mouth has no action on the pancreas
Material Fed by Stomach Tube
Rate of Secretion of Pancreatic Juice in C.c. per Hr.
Continuous Secretion
Before Feeding
Continuous Secretion
After Feeding
First
Hour
Second
Hour
Third
Hour
First
Hour
Second
Hour
Third
Hour
150 c.c. active secretin, slightly acid
6.5
3.6
3.9
20.0
6.0
8.0
150 c.c. active secretin, slightly alkaline
13.0
11.0
5.0
23.0
26.0
12.0
150 c.c. secretin passed through Berkefeld
7.8
7.5
7.4
23.0
13.0
11.0
150 c.c. extract of colon
11.6
12.0
11.4
30.0
19.6
14.8
150 c.c. extract of gastric mucosa
10.0
7.0
8.0
23.0
7.5
4.0
150 c.c. extract of muscle
6.9
11.0
6.4
35.0
5.0
7.0
150 c.c. 0.4% HCl (diluted to 250 c.c.)
6.0
8.0
4.0
33.0
36.0
17.0
Results.—We have data from six dogs with a total of seventy-six experiments. As shown in Table 4, the administration of secretin causes an increase in the flow of pancreatic juice, but the administration of inert substances as extracts of colon, gastric mucosa or muscle causes a like increase. The activity of the secretin may be reduced to a low value by exposure to sunlight, or filtering through a Berkefeld filter, yet the response of the pancreas is not correspondingly reduced. The secretion that occurs in the control cases, every one will admit, is but secondary to the production of gastric juice with its accompanying hydrochloric acid, that is, excited by virtue of the extractives and water in the preparations. Such, we can prove, is the only action of secretin. A mixture of gelatin, peptone and salt water, the chief incidental constituents of a secretin preparation, gives as striking results as ever obtained from secretin administration. Yet the objection may be made that the response of the pancreas that is due to the incidental constituents of secretin is maximal, and that the secretin consequently has no opportunity to display its particular potency. But, as inspection of the accompanying tables illustrate, the administration of hydrochloric acid shows that the response is by no means maximal. Let us cite a striking experiment. For three hours before the administration of hydrochloric acid, the secretion in cubic centimeters was respectively 29.4, 11.75 and 35.4 c.c.; for the three hours after, respectively 88.0, 49.0 and 40.5 c.c.
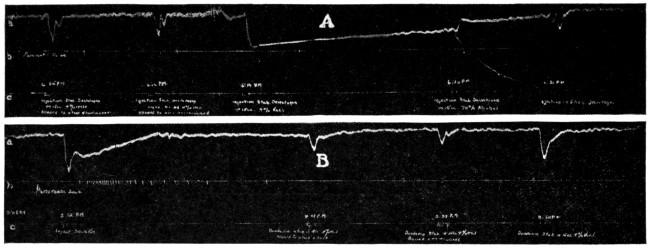
Fig. 1.—Tracings (reduced two-thirds) showing failure of Secretogen, Elixir Secretogen, and Duodenin to stimulate the flow of pancreatic juice even when administered intravenously in amounts three times greater than that recommended to be given by mouth. Dog: light ether anesthesia; cannula in the pancreatic duct; a, carotid blood pressure; b, flow of pancreatic juice in drops; c, signal showing where the intravenous injections were made. Tracing A: Reading from left to right, the five intravenous injections are: (1) three tablets of Secretogen digested with 15 c.c. 0.4 per cent. hydrochloric acid and neutralized; (2) three tablets of Secretogen boiled in 15 c.c. 0.4 per cent. hydrochloric acid and neutralized; (3) three tablets of Secretogen in 15 c.c. 0.9 per cent. sodium chlorid; (4) three tablets of Secretogen in 15 c.c. of 70 per cent. alcohol; (5) 15 c.c. Elixir Secretogen. Tracing B: reading from left to right, the four intravenous injections are: (1) 5 c.c. secretin made fresh from dog’s duodenal mucosa; (2) three tablets of Duodenin digested in 15 c.c. 0.4 per cent. hydrochloric acid and neutralized; (3) three tablets of Duodenin boiled in 15 c.c. 0.4 per cent. hydrochloric acid and neutralized; (4) three tablets of Duodenin in 15 c.c. sodium chlorid (0.9 per cent.).
TABLE 5.—SUMMARY OF EXPERIMENTS
Dogs with pancreatic fistula, weight 14 kg. Secretin given by mouth
No. of
Experiment
Material Fed
Rate of Secretion of Pancreatic
Juice in C.c. Per Hour
Increase
in C.c.
Three Hours
Before Feeding
Three Hours
After Feeding
3
Secretin slightly acid
5
11
6
5
Secretin slightly alkaline
24
30
6
4
Secretin passed through Berkefeld
18
23
5
1
Secretin exposed to sun for 4 hrs
16
29
13
2
Extract of colon (rabbit)
19
29
10
3
Extract of gastric mucosa
14
23
9
3
Extract of muscle
8
16
8
2
Mixture of gelatin, peptone and salt
23
33
10
1
1 per cent. peptone solution
6
8
2
4
0.2 per cent. hydrochloric acid
13
37
24
3
Milk and bread
7
20
13
It is possible by large doses of sodium bicarbonate given shortly before the administration of a preparation so to depress the stomach that it does not respond with the usual production of hydrochloric acid. Under these conditions the administration of secretin is uniformly negative, but the administration of hydrochloric acid on the contrary still serves to increase the pancreatic secretion (Table 6).
TABLE 6.—SECRETIN IN EXPERIMENTAL “ACHYLIA GASTRICA”
Exp.
No.
Material Fed
Rate of Secretion of Pancreatic Juice in C.c. Per Hour
Continuous Secretion Before Feeding*
Secretion After Feeding
First
Second
Third
First
Second
Third
1
150 c.c. secretin
8.7
7.5
6.8
3.0
1.0
4.8
2
4.5
6.5
10.0
6.0
7.5
7.6
3
15.6
8.1
16.0
3.9
4.9
2.9
1
150 c.c. 4% HCl (diluted to 250 c.c.)
9.8
7.0
6.0
65.1
28.0
7.1
2
17.4
18.5
17.0
34.0
18.0
20.0
* Five gm. Na HCO3 given at beginning of each first two hours.
COMMERCIAL PREPARATIONS OF SECRETIN
Secretogen and Elixir Secretogen.—The Carnrick Company offers Secretogen90 for use in a large number of conditions. The following indications for the use of the preparation purport to be based on clinical tests covering a period of several years: dyspepsia, and the indigestions generally, fermentative disorders, gastric catarrh, flatulence, nausea; pancreatic insufficiency, intestinal indigestion; gastric secretory deficiencies, apepsia; constipation and hepatic torpor; intestinal stasis; diarrhea; infantile diarrhea, “summer complaint,” marasmus, inanition and malnutrition; gastric atony and dilatation; cholecystitis and gallstones; nephritis, neurasthenia, cachexia and cancer; epilepsy and high blood pressure. Testimonials are presented as to results in most of these conditions.
A quantity of “Secretogen” and “Elixir Secretogen” was bought in the open market, and the preparations were tested on suitably prepared dogs. The tablets were ground, thoroughly macerated with the solvent used (water, normal salt solution, alcohol, or 0.4 per cent. hydrochloric acid), and filtered. If hydrochloric acid was used, the pulverized tablets were boiled with it, in the manner that secretin is made from duodenal mucosa, and the preparations neutralized previous to injection. The injections were made in from 15 to 20 c.c. of the solvent. All the operations were carried on immediately before the experiment, and as rapidly as possible, so as to avoid oxidation. The Elixir Secretogen was injected directly, without dilution.
TABLE 7.—SUMMARY OF TYPICAL EXPERIMENTS SHOWING THE ABSENCE OF SECRETIN IN “SECRETOGEN”AND “ELIXIR SECRETOGEN” EXCEPT IN OCCASIONAL TESTS WHEN ADMINISTERED IN ENORMOUS DOSES
Dogs under ether anesthesia
Exp.
No.
Quantity of Secretogen and
Elixir Secretogen Used*
Secretion of Pancreatic Juice in Drops,
Following Intravenous Injection
Control
10 C.c.
Secretin
Secretogen in
Elixir
Control
10 C.c.
Secretin
Distilled
Water
0.4%
HCl
70%
Alcohol
0.9%
NaCl
1
Secretogen, 1 tablet; Elixir, 15 c.c.
109
0
0
0
0
0
59
1
Secretogen, 6 tablets
...
..
0
..
..
..
..
2
Secretogen, Elixir, 15 c.c. 3 tablets;
16
0
0
0
0
1(?)
16
3
Secretogen, 5 tablets
...
..
1(?)
..
..
..
..
4
Secretogen, 25 tablets
14
..
1(?)
..
..
..
8
5
Secretogen, 100 tablets
110
..
..
..
21
..
67
6
Secretogen, 100 tablets; Elixir, 125 c.c.
19
..
5
..
1
2(?)
8
7
Elixir, 50 c.c.
...
..
..
..
..
1(?)
..
* One to three tablets is (according to the label) the therapeutic dose of Secretogen; 4 to 12 c.c. the dose of Elixir Secretogen.
Results.—In only one case was a slight response obtained, the others gave none. Small and large doses were equally inert (Table 7, Figs. 2, 3). The preparations, though inert, always produced a depression in blood pressure, sometimes even greater than that caused by active secretin. Among our many tests, one bottle was found, however, to be a little different from the rest (Experiment 4). Its entire content, 100 tablets, had been ground and boiled in 0.9 per cent. sodium chlorid. The extract on injection was found to have a small but unmistakable secretin reaction, equivalent to about 2 c.c. of the control secretin used. But repeated experiments were unable to duplicate this result. The “Secretogen” and “Elixir Secretogen” were all supposedly fresh preparations, the retail drug store informing us that a fresh supply was obtained from the wholesale house each week.
Secretogen, then, contains practically no secretin, and even if it did contain secretin, it can have no effect on the pancreas when taken by mouth. The indications for Secretogen, therefore, are based on false premises, and the testimonials are worthless.
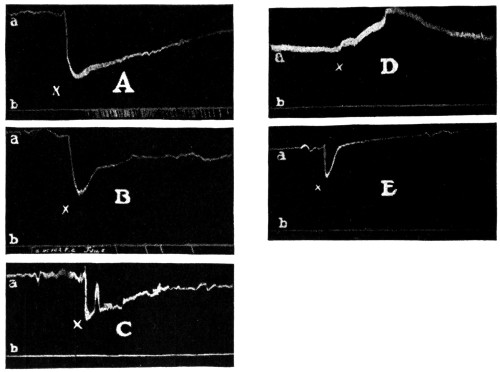
Fig. 2.—Tracings (reduced one-half) showing no stimulation of the pancreas by Secretogen, Elixir Secretogen, and Duodenin, even when administered intravenously in quantities one hundred times greater than the therapeutic dose by mouth. Dog: Light ether anesthesia; cannula in the pancreatic duct; a, carotid blood pressure; b, flow of pancreatic juice in drops. Tracing A: at x, intravenous injection of 10 c.c. secretin prepared from duodenal mucosa of dog. Tracing B: at x, intravenous injection of 100 tablets of Secretogen digested with 0.4 hydrochloric acid and neutralized. Tracing C: at x, intravenous injection of 100 tablets of Secretogen, prepared as in Tracing B. Tracing D: at x, intravenous injection of 50 c.c. Elixir Secretogen. Tracing E: at x, intravenous injection of 100 tablets of Duodenin (dissolved in 0.9 per cent. sodium chlorid).
Duodenin.—This is a preparation manufactured by Armour & Company, which purports to be “secretin plus enterokinase.” The claims for this product are similar to those for Secretogen, but somewhat less sweeping. According to the manufacturers, “Duodenin (Armour) is recommended in the treatment of intestinal disorders where an increased flow of pancreatic, hepatic and intestinal secretion is desired. It is of specific value in proteid digestion on the theory that secretin and enterokinase stimulate the pancreas and activate its secretion.”
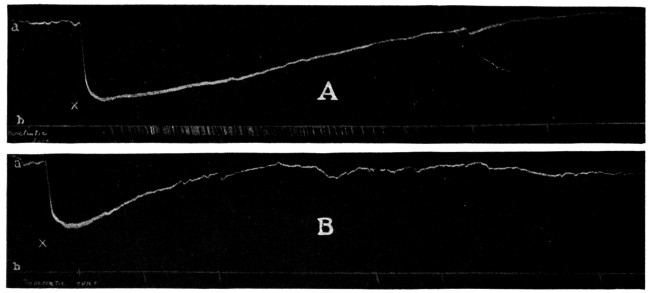
Fig. 3.—Tracings (reduced one-half) showing practically complete destruction of secretin by the gastric juice. Dog under light ether anesthesia; cannula in the pancreatic duct; a, carotid blood pressure; b, record of flow of pancreatic juice in drops. Time, twenty-five minutes. Tracing A: intravenous injection of 10 c.c. secretin (prepared fresh from dog’s duodenal mucosa) at x. Tracing B: intravenous injection (at x) of 10 c.c. of the same secretin as in Tracing A, after being digested in normal human gastric juice at 37 C. for two hours.
We bought a quantity of Duodenin in the open market, and carried out on this product the same series of experiments as that used in the case of Secretogen. The results were similarly negative (Table 8).
TABLE 8.—SUMMARY OF TYPICAL EXPERIMENTS SHOWING THE ABSENCE OF
SECRETIN IN “DUODENIN”
Dogs under ether anesthesia
Exp.
No.
Number
Duodenin
Tablets
Used
Secretion of Pancreatic Juice in Drops,
Following Intravenous Injection
Control
10 C.c.
Secretin
Duodenin in
Control
10 C.c.
Secretin
Distilled
Water
0.4%
HCl
70%
Alcohol
0.9%
NaCl
1
3
29
0
0
0
1(?)
28
1
6
...
..
1(?)
..
..
..
2
18
16
..
6
..
..
16
3
5
14
..
0
0
0
8
3
25
...
..
1(?)
..
..
..
4
100
110
..
0
..
..
67
5
150
19
..
0
..
0
8
In regard to both Secretogen and Duodenin, we assume that the manufacturers have tried to put secretin in them, but have been unable because they have failed, in all likelihood, to check their methods by physiologic standardization. These firms do not give any details as to the procedure they employed in their manufacture of secretin. Desiccated secretin of extreme potency has been prepared by various physiologists,91 1 mg. (1⁄64 grain) of which is active when given intravenously. It is difficult to conceive that any of these methods were used in the preparation of Secretogen or Duodenin.
CONCLUSIONS
1. Secretin is quickly destroyed by gastric juice and by trypsin.
2. Secretin is not absorbed in active form from the alimentary tract.
3. The presence of secretin or prosecretin cannot be demonstrated in the commercial preparations “Secretogen,” “Elixir Secretogen” and “Duodenin” even when the therapeutic dose of the preparations is given intravenously. In the case of “Secretogen,” intravenous injection of 100 times the therapeutic dose reveals occasionally an insignificant trace of secretin.
DISCUSSION OF RESULTS
It is, of course, objectionable that preparations containing no secretin should be advertised to the medical profession as containing this substance. The more important blunder, however, consists in the attempt to offer such preparations for oral administration, because even chemically pure secretin would be equally ineffective when taken by mouth. There is as yet no reliable evidence that lack of secretin is a primary or important factor in any disease. Even should this be established, secretin therapy, to be effective, must be intravenous. Secretin has not yet been prepared in sufficiently pure state to render possible intravenous injection in man without injurious effects. And even when this has been attained, the very fleeting action of secretin will in all probability render secretin therapy as futile in all the diseases in which it is theoretically indicated as epinephrin therapy is in Addison’s disease.
But there remains the alleged favorable effect from secretin therapy by mouth in various diseases in man. It is, perhaps, impertinent for laboratory men to comment on these clinical results. The ordinary “testimonials” need not be considered, but we should like to ask the serious worker who thinks he has actually obtained good results from secretin therapy how certain he is of the causal relation between the giving of secretin or alleged secretin and the abatement of the disease.
When a therapeutic measure not only lacks a positive basis in physiology and pathology but runs contrary to all the well-established experimental facts in these fundamental medical sciences, is it too much to ask that positive clinical findings be subjected to more than usual critical analysis before acceptance? “Clinical tests,” it is said, “covering a period of several years have proved that neither the condition in the stomach during digestion nor those in the intestine prevent the secretin from entering intact into the circulation.” When we meet claims such as this, should we not scrutinize the “tests” as well as the men who make them?
We are indebted to Dr. J. H. Moorehead for assistance in part of the surgical work.—(From The Journal A. M. A., Jan. 15, 1916.)
ARTICLES REFUSED RECOGNITION
Report of the Council on Pharmacy and Chemistry
Below appear abstracts of the Council’s action on articles refused recognition which were not deemed of sufficient importance to require lengthy reports:
Radio-Rem
The Radium Therapy Company, Schieffelin & Co., selling agents, submitted to the Council radium emanation generators called “Radio-Rem Outfits,” designed to generate respectively 200, 1,000, 2,000, 5,000 and 10,000 Mache units per twenty-four hours.
Those who are well informed on the subject of radium therapy are of the opinion that the administration of small amounts of radium emanation such as generated by certain outfits is without therapeutic value. It has been stated that at the Radium Institute of London the minimum preliminary dose is 185 microcuries (500,000 Mache units), and as many as 555 microcuries (1,500,000 Mache units) are employed.
In consideration of these facts the Council voted not to accept any radium emanation generator which produces less than 2 microcuries of emanation during twenty-four hours. Accordingly, while accepting Radio-Rem Outfit No. 5, claimed to produce 10,000 Mache units (3.7 microcuries) and Radio-Rem Outfit No. 4, claimed to produce 5,000 Mache units (1.8 microcuries), the Council voted not to accept Radio-Rem Outfit No. 3, claimed to produce 2,000 Mache units (0.74 microcurie), Radio-Rem Outfit No. 2, claimed to produce 1,000 Mache units (0.37 microcurie), and Radio-Rem Outfit C, claimed to produce 200 Mache units (0.07 microcurie).
This report having been submitted to Schieffelin & Co. and their reply considered, the Council authorized publication of the report. [See also Reports of Council on Pharmacy and Chemistry, 1916, p. 631.]
Olio-Phlogosis
Olio-Phlogosis, a liquid preparation to be applied externally by means of a cotton pad, is advertised by the Mystic Chemical Company, Kansas City, Mo., thus:
“Doctor: Don’t fail to use Olio-Phlogosis liberally for Pneumonia, Bronchitis and Pleurisy. It works quickly. Olio-Phlogosis is as far ahead of all medicated kaolin plasters as these plasters were ahead of the old-time moist and soggy poultices.”
A pamphlet advises the use of Olio-Phlogosis in
“... all cases of Inflammation and Congestion, such as Pneumonia, Bronchitis, Pleurisy, Croup, Boils, Carbuncles, Rheumatism, Swollen Glands, Peritonitis, Ovaritis, as a Surgical Dressing, Mamitis [Mastitis (?)] Vaginitis and Metritis (on cotton tampon to deplete these parts), Septic Wounds, Old Ulcers, Chilblain, Eczema, Neuralgia, Inflammation of the Eyes and Ears, Alveolar Inflammation, Burns, Scalds, Etc.”
According to the information sent to the Council by the Mystic Chemical Company, Olio-Phlogosis has the following composition per gallon:
Ol. Eucalyptus Gaultheria
drs.
8
Ol. Abies Canadensis
drs.
8
Ol. Abies Canadensis
drs.
2
Ol. Thyme (white)
drs.
2
Resublimated Iodin crystals
grs.
32
Resorcin
drs.
1
Acid Boracic C. P.
drs.
2
Quinine Bisulphate
drs.
4
Sodium Thiosulphate
drs.
3
1⁄2Glycerin C. P.
q. s. ad
gal. 1
A nonquantitative formula which appears on the label of a sample bottle sent to a physician enumerates the same ingredients except the sodium thiosulphate.
The A. M. A. Chemical Laboratory reports that no free iodin could be detected in the preparation.
Apparently, then, Olio-Phlogosis is essentially a skin irritant applied by means of cotton; it can be expected to be just about as effective as the old-fashioned cotton pneumonia jacket, used in conjunction with an aromatic skin irritant, such as camphorated oil or wintergreen or menthol ointment. The odor may have some psychic effect, and it is possible that some of the oily matter may be absorbed by the skin. That such small amounts, even if absorbed, can produce any considerable systemic effect, however, is highly improbable, and the advice that this preparation be relied on in pneumonia, pleurisy, peritonitis, etc., is pernicious. In the few cases of pneumonia in which heat is indicated, the plain cotton pad will usually be found sufficient. If the physician consider the addition of a skin irritant desirable, it is easy to select one from the official preparations. It will be far more rational to do so than to invoke the aid of a mystic name and a complex formula to which the patient and his family, at least, will be led to give unmerited credit.
The claims made for Olio-Phlogosis are unwarranted; its composition is complex and irrational, and the nondescriptive but therapeutically suggestive name is likely to lead to uncritical use. The Council voted that the product be refused recognition for conflict with Rules 6, 8 and 10, and that this report be published.—(From The Journal A. M. A., Aug. 19, 1916.)
THE HYPOPHOSPHITE FALLACY
Report of the Council on Pharmacy and Chemistry
The Council has adopted the following report and authorized its publication.
W. A. Puckner, Secretary.
The introduction of hypophosphites into medicine was due to an erroneous and now discarded theory as to the cause of tuberculosis of which one Dr. J. F. Churchill of London, and later of Paris, was the promulgator and propagandist.92 This theory was that the so-called “tuberculosis diathesis” was due to a deficiency of phosphorus in the blood. Believing that the hypophosphites, while nontoxic, were capable of further oxidation in the organism, Churchill recommended them as the best means of supplying the supposedly lacking phosphorus. It is now known that tuberculosis is not due to a deficiency of phosphorus. Of more importance is the fact, now known, that little phosphorus, if any, is assimilated from the hypophosphites—far less than from phosphorus compounds of ordinary food.93 There is no justification for giving hypophosphites for the sake of their phosphorus content. For various reasons, however—partly from force of habit and partly because of the power of advertising—many physicians still prescribe hypophosphite preparations, and consequently, they are still included in the Pharmacopeia and in textbooks on materia medica and therapeutics. They are put out in the form of “specialties” and of proprietary preparations, and are lauded extravagantly by the manufacturers of the latter.
Although the overwhelming weight of evidence was against the probability that the hypophosphite preparations are of value as therapeutic agents, the Council thought it well to investigate the subject. Dr. W. McKim Marriott of Baltimore was therefore requested to review the evidence for and against the therapeutic usefulness of the hypophosphites and to conduct such experiments as seemed necessary. His report has already appeared in The Journal.94
Dr. Marriott found that nine observers (Paquelin and Joly, Vermeulen, Boddaert, Massol and Gamel, Panzer, Delaini and Berg), who endeavored to test the alleged utilization of the hypophosphites in the organism, reported that there is complete, or practically complete, elimination of hypophosphites in the urine, with little or no effect on the body. Only one experimenter (Patta) claimed that a considerable amount of ingested hypophosphite was retained in the body; however, he used a method now known to be inaccurate and made obvious errors in calculation, so that his conclusions were unwarranted.
Since the evidence was even to this extent contradictory, Marriott performed a series of experiments. The methods of this study and details of results are described in his paper, in which he also discusses the experiments of some other observers. Marriott writes:
62 Launoy: Arch. internat. de Physiol. 3:62, 1906. Morel and Terroine: Compt. rend. Soc. de biol. 67:36, 1909. Zunz: Arch. internat. de physiol. 8:181, 1909. Lalou: Jour. de physiol. 14:465, 1912.
88 Delezenne and Pozerski: Jour. de Physiol., 14:540, 1912.
89 Pawlow: Ergeb. de Physiol., O., p. 266, 1902.
90 Secretogen, Report of the Council on Pharmacy and Chemistry, J. A. M. A., May 1, p. 1518, 1915.
91 Stepp (Note 13). Dale and Laidlow: Jour. Physiol. 44:11, 1912. Launoy and Ochslin: Comp. rend. Soc. de biol., 74:338, 1913.
92 Churchill, J. F.: De la cause immédiate et du traitement spécifique de la phthisie pulmonaire et des maladies tuberculeuses, Paris, 1858.
93 The Hypophosphite Fallacy, J. A. M. A., April 25, 1914, p. 1346.
94 Marriott, W. McKim: The Therapeutic Value of the Hypophosphites, J. A. M. A., Feb. 12, 1916, p. 486.
“None of the subjects of the experiment [Marriott’s] experienced any effect whatsoever from the administration of the drug.... Almost all of the ingested hypophosphite is promptly eliminated unchanged....
“These experiments [Forbes’] demonstrate conclusively that the hypophosphites possess no specific value as a source of phosphorus for the body. This is not to be wondered at in view of the fact that 85 per cent. of the phosphorus ingested in the form of hypophosphite is excreted unchanged, and there is no proof that even the remaining 15 per cent. is available to the organism. It is doubtful if there are any conditions in which the body suffers from lack of phosphorus. Even should such conditions exist, phosphorus, in the form that it occurs in the ordinary foods, or as phosphates, is more efficient in supplying the deficit than hypophosphites that must be oxidized before utilization and which are only about 15 per cent. oxidized, if at all. For example, half a glass of milk contains more available phosphorus than three large doses of hypophosphites of 15 grains each, as great a dosage as is usually given.
“What, then, is the therapeutic value of hypophosphites? There is no reliable evidence that they exert a physiologic effect; it has not been demonstrated that they influence any pathologic process; they are not ‘foods.’ If they are of any use, that use has never been discovered.”
In view of the foregoing, it seemed to the Council advisable to examine the claims under which a few of the proprietary hypophosphite preparations are marketed. The following are representative:
FELLOWS’ SYRUP OF HYPOPHOSPHITES
No very exact information concerning the composition is furnished by the manufacturers (Fellows Medical Mfg. Co., New York). They say that the product
“... contains the chemically pure hypophosphites of iron, quinin, strychnin, calcium, manganese and potassium, agreeably blended in the form of a bland, stable syrup with a slightly alkaline reaction....
“Each fluid drachm contains the equivalent of 1-64th of a grain of pure strychnin.”
The Fellows’ Hypophosphites advertising furnishes something like a barometer of the popular status of hypophosphites. In one circular (undated, but, from certain references contained in it, presumably issued ten or fifteen years ago) we read:
“It is an indubitable fact that the hypophosphites have earned the distinction of having their therapeutic value more completely established than have any other remedial agents.... it is only by accepting the current view, which was originally advanced by Mr. Fellows, that we can satisfactorily account for the incontestable fact that the hypophosphites are of supreme importance in the treatment of a very extensive variety of affections.... the hypophosphites increase the consumption of oxygen and the elimination of carbon dioxide. In this manner, they stimulate nutrition and promote constructive metamorphosis.... It is now universally conceded that the widespread utility of the hypophosphites is due to the fact that they substantially improve metabolic processes, thus increasing the disease-resisting capacity of all the tissues.”
The circular, continuing, emphasizes the “incomparable phosphorus-contributing properties” of Fellows’ Syrup, its “extraordinary reconstructive properties” and “the magnificent results which invariably attend its employment in the treatment of anemia, chronic bronchitis, chlorosis, neurasthenia, mollities ossium, delayed union of fractures, rickets, convalescence,” etc.
A circular bearing the copyright date 1914, on the other hand, admits that:
“The theories for the favorable action of Fellows Syrup of Hypophosphites have undergone several changes.”
The same circular further maintains, however, that:
“... the fact has never been challenged that in Fellows Syrup of Hypophosphites we have one of the most efficient, most complete, most all-around tonics and roborants in the materia medica.”
No attempt is made to base this assertion on the therapeutic action of the constituents. In other words, the old theory, which formed the basis for the popularity of Fellows’ Syrup, has been thrown overboard, but no substitute is deemed necessary; the momentum already acquired is apparently regarded as sufficient to insure its continued sale.
Fellows’ Syrup of Hypophosphites is a semisecret, unscientific preparation—an affront to sound therapy—exploited by means of extravagant and misleading statements.
SYRUPUS ROBORANS (SYRUP HYPOPHOSPHITES COMP. WITH QUININ,
STRYCHNIN AND MANGANESE)
Little information concerning this preparation seems to be furnished at present by the manufacturers, Arthur Peter & Co., Louisville, Ky. According to an old circular, it contains, in each fluidounce,
Grains
“Hypophos. Potass.
1
1⁄2Hypophos. Manganese
1
Hypophos. Lime
1
Hypophos. Iron
1
1⁄2Hypophos. Quinin
6⁄16Hypophos. Strichnin
1⁄16“
1⁄128grain Strychnia to Teaspoonful.”
Further, according to the same circular:
“The Hypophosphites are especially useful in all diseases where there is a lack of nutrition.... They are the best of all remedies in Rachitis, non-union of fractures, Osteomalacia and Syphilitic Periostitis.”
As for Syrupus Roborans itself:
“This elegant preparation is ... the best general tonic and reconstructive known.”
The unwarranted therapeutic claims formerly made for it seem to be no longer circulated. Syrupus Roborans is an unscientific, shotgun mixture.
SCHLOTTERBECK’S SOLUTION HYPOPHOSPHITES OF LIME AND SODA
(LIQ. HYPOPHOSPHITUM, SCHLOTTERBECK’S)
The Schlotterbeck & Foss Co., Portland, Maine, the manufacturers, say of their preparation:
“This solution contains 30 grains of the combined Hypophosphites of Lime and Soda to the ounce. It contains No Sugar, No Acid and it is Perfectly Neutral.”
“Indications for use.—Galactostasis, Imperfect Metabolism, Neurasthenia, Nervous Dyspepsia, Insomnia, Convalescence, Acetonuria, Cyclic Vomiting in Infants, Diabetes, Starvation, Deficiency of Lime, Mother’s Teeth during Pregnancy, Dentition of Infants, Rachitis, Furunculosis, Vomiting of Pregnancy, Obesity.”
“Migraine is often caused by conditions for which this Solution is one of the most satisfactory remedies:”
“In Insomnia due to advancing age, it will often act as a hypnotic....”
Of the hypophosphites the Schlotterbeck & Foss Company say:
“If ‘damning it with faint praise’ on the part of some of the leading medical authorities, or utterly condemning it as useless, on the part of others, would kill a medicine, the Hypophosphites would long since have disappeared as medicinal agents. Negative testimony in regard to the value of a drug does not settle anything.”
Of their own preparation they say:
“When we get the results that ought to follow the administration of Hypophosphites, we have proved that Schlotterbeck’s Solution enters the system unchanged.”
“This Solution is primarily a blood and nerve tonic and chemical food.”
Schlotterbeck’s Solution of Hypophosphites of Lime and Soda is a semisecret preparation marketed under claims that are both unwarranted and misleading.
ROBINSON’S HYPOPHOSPHITES
According to the manufacturers, the Robinson-Pettet Company, Louisville, Ky., each fluidounce of this preparation contains:
“Hypophosphites Soda
2
gr.Hypophosphites Lime
1
1⁄2 gr.Hypophosphites Iron
1
1⁄2 gr.Hypophosphites Quinin
3⁄4 gr.Hypophosphites Strychnine
1⁄16 gr.”It is claimed to be
“Nutritive, Tonic Alterative. A Standard Remedy in the treatment of Pulmonary Phthisis, Bronchitis, Scrofulous Taint, General Debility, etc. Stimulates Digestion, promotes Assimilation.”
The declared composition of the preparation is unscientific, and the therapeutic claims are unwarranted.
EUPEPTIC HYPOPHOSPHITES
Nelson, Baker & Co., Detroit, Mich., who market Eupeptic Hypophosphites, call this preparation:
“A superior combination containing the Hypophosphites of Potassium, Calcium, Iron and Manganese, and the bitter tonics, Quinin and Strychnin, agreeably associated with natural digestive ferments of the pancreatic secretion. It is thus a general reconstructive tonic.... The remedy is of especial value in the treatment of mental and nervous affections.... It is indicated in pulmonary tuberculosis, in all wasting diseases, in debilitated conditions generally and in all exhaustion from over work.”
On the basis of the manufacturer’s statement, Eupeptic Hypophosphites must be regarded as a semisecret, unscientific, shotgun preparation, exploited through unwarranted therapeutic claims.
MCARTHUR’S SYRUP OF THE HYPOPHOSPHITES COMP. (LIME AND SODA)
So far as the recent literature and trade package are concerned, no information as to the composition of this product is furnished beyond what is conveyed in the name. The advertising for McArthur’s Syrup, like that for Fellows’ Syrup and Peters’ Syrupus Roborans, has been modified as time has passed. A few years ago it was advertised under such claims as the following:
“... Has Stood the Test during many years for unequaled efficacy in the treatment of Tuberculosis.... Indicated also as a Tonic and Tissue Builder in convalescence from Fevers, in Nervous Diseases, Rickets, Senile Debility and Bronchitis.”
“Its use is indicated in ... diseases of the chest, chronic cough, throat affections, general debility, brain exhaustion, cholera infantum and wasting diseases of children.”
At present no definite claims seem to be made for it; the manufacturers evidently find the magic name of hypophosphites sufficient to evoke the spell for which the advertisement writer’s aid was once sought. A testimonial contained in a circular which seems to be still used illustrates both the kind of aura which surrounds hypophosphites in the minds of physicians who are still living in the past, and the kind of logic which has made the reputation of this and many other equally worthless preparations.
“Just about six years ago I had a severe attack of La Grippe which almost killed me. Left me with Asthma (Catarrh) and a severe cough. Did not get out of the house for three months. Took over a dozen bottles McArthur’s Hypophos.—came out all right and since then worked hard, but last Fall took another cold, but worked on, used McArthur’s Hypophos., am using it now, am on my 12th bottle.
“I have five or six patients whom I have put on McArthur’s Hypophos., but I do not prescribe the single bottle, but wholesale no less than half dozen bottles. One patient is on his 24th bottle with orders to get another half dozen and keep it up all winter. I have given the same order to all (keep it up all winter) and I myself intend to do the same, for with its use I have lost no time—rain or shine I am doing my work. I know what it has done for me and what it is doing for my patients.”
It would be hard to find a more characteristic example of the naïve mental processes of the simple folk who in all good faith write testimonials for worthless medicines. This well-meaning practitioner (a homeopath, by the way), because he “came out all right” after an attack of grip, returns all praise to McArthur’s Hypophosphites, which he has taken “wholesale.” Not the faintest doubt of the validity of his post hoc ergo propter hoc argument seems to glimmer across his consciousness.
McArthur’s Syrup of the Hypophosphites is an irrational preparation. While its faults are fewer and less glaring than those of some other proprietaries, the circulation of such a testimonial as the one just quoted is sufficient of itself to cast suspicion on the product.
BORCHERDT’S MALT OLIVE WITH HYPOPHOSPHITES, MALTZYME WITH HYPOPHOSPHITES AND MALTINE WITH OLIVE OIL AND HYPOPHOSPHITES
These preparations are now described in the appendix to New and Nonofficial Remedies. Borcherdt’s Malt Olive with Hypophosphites (Borcherdt Malt Extract Company, Chicago) is said to contain in each 100 c.c., 0.64 gm. each of calcium and sodium hypophosphites, with malt extract, olive oil and glycerine. Maltzyme with Hypophosphites (Malt-Diastase Company, New York) is said to contain, in each 100 c.c., 0.4 gm. each of calcium, sodium and potassium hypophosphites and 0.005 gm. each of iron and manganese hypophosphites, with maltzyme. Maltine with Hypophosphites (Maltine Company, Brooklyn, N. Y.) is said to contain in each 100 c.c., 0.64 gm. each of calcium and sodium hypophosphites and 0.42 gm. of iron hypophosphite, with maltine. Maltine with Olive Oil and Hypophosphites (Maltine Company, Brooklyn, N. Y.) is said to contain, in each 100 c.c., 0.6 gm. each of calcium and sodium hypophosphites, with maltine and olive oil. In general, no therapeutic claims are made for these mixtures so far as the hypophosphites are concerned. The addition of hypophosphites to such mixtures is irrational and, since it tends to perpetuate the hypophosphite fallacy, detrimental to sound therapeutics.
THE COUNCIL’S ACTION
The Council endorsed the conclusions of the work of Dr. Marriott referred to above, and noted: (1) that the therapeutic use of hypophosphites (except possibly in some cases as a convenient means of administering the positive element in the salt, as ammonium in ammonium hypophosphite or calcium in calcium hypophosphite) is irrational; (2) that the merits of each hypophosphite salt submitted for consideration under the foregoing exception must be judged individually, and (3) that Fellows’ Syrup of Hypophosphites, Peters’ Syrupus Roborans, Schlotterbeck’s Solution Hypophosphites of Lime and Soda, Robinson’s Hypophosphites, the Eupeptic Hypophosphites of Nelson, Baker & Co., and McArthur’s Syrup of the Hypophosphites are ineligible for inclusion in New and Nonofficial Remedies, and that Borcherdt’s Malt Olive with Hypophosphites, Maltzyme with Hypophosphites, Maltine with Hypophosphites, and Maltine with Olive Oil and Hypophosphites be deleted from the appendix of N. N. R. Of these preparations, all are in conflict with Rule 10; Fellows’ Syrup, Schlotterbeck’s Solution, Robinson’s Hypophosphites and Nelson, Baker & Co.’s Eupeptic Hypophosphites are in conflict with Rule 6; the Fellows, Schlotterbeck, and Nelson, Baker preparations are also in conflict with Rule 1.—(From The Journal A. M. A., Sept. 2, 1916.)
PULVOIDS CALCYLATES
Report of the Council on Pharmacy and Chemistry
Pulvoids Calcylates 5 grains was submitted by the Drug Products Company, Inc., New York, under the following claims as to composition:
“When ingested represents the following chemical formulas: C6H4
OH
COO
2 Ca.2H2O + Sr.(C7H5O3)2 + 2H2O.”
“Strontium Di-Salicylate 21⁄2 grs. and our especially prepared Salt of Calcium and Acid Salicylic adjusted in such nascent form, that these pulvoids upon ingestion will promptly form Calcium Neutral Di-Salicylate 21⁄2 gr.”
“A combination of Calcium and Strontium Di-Salicylate, in seemingly true chemical union.”
These statements are rather vague, possibly because they are an attempt to mystify. The product, however, may be assumed to be a mixture (not a chemical combination) of calcium salicylate and strontium salicylate. The therapeutic claims made for the preparation are:
“Superior to ordinary salicylates. Can be taken continuously and indefinitely without gastric irritation, insuring maximum efficiency.”
“Reports show surprisingly good results, even where the sodium salt fails.”
As there is no evidence to show that strontium salicylate, calcium salicylate or a mixture of the two salts has any advantage over sodium salicylate, these claims cannot be accepted. The name and the statement of composition are objectionable in that they do not reveal the identity of the drugs in “Calcylates” and in suggesting that this preparation possesses radical advantages over salicylates in other forms.
The Drug Products Company was told that the facts just mentioned rendered “Pulvoids Calcylates” ineligible for New and Nonofficial Remedies. The company in its reply objected to the Council’s conclusions, and in support of its position submitted testimonials from a number of physicians. The reply of the company embodied no facts or arguments which had not been considered by the Council’s referee, and the testimonials from physicians contained no evidence to show that the combination has any real advantage over sodium salicylate.
The Council therefore declared “Pulvoids Calcylates” ineligible for New and Nonofficial Remedies for the following reasons: Unwarranted therapeutic claims are made for the mixture (Rule 6); the name does not describe the composition (Rule 8), and the mixture is an unessential modification of an established remedy (sodium salicylate) (Rule 10).—(From The Journal A. M. A., Sept. 9, 1916.)
SULFURYL MONAL
Report of the Council on Pharmacy and Chemistry
Sulfuryl Monal is said to be manufactured by Monal Frères, manufacturing chemists of Nancy, France. It is sold in the United States by George J. Wallau, Inc., New York City. According to the label:
“Each Pastille
Contains: Sulfuryl (combined polysulphurets)
= 0.35 centigr.”
Liberates: Nascent sulphurretted Hydrogen
= 2 cub. cent.”
The Chemical Laboratory of the American Medical Association was requested to check the amount of available hydrogen sulphid. An original bottle of Sulfuryl Monal was used; this contained tablets having the taste of licorice extract and an odor of hydrogen sulphid. The tablets were found to liberate about 6 c.c. hydrogen sulphid to each tablet.
Among the claims made for the preparation are:
“Dissolved by the saliva, Sulfuryl Monal reaches the stomach where, under the influence of the gastric juice, it generates nascent sulphuretted hydrogen. Professor Albert Robin’s remarkable researches have proven that it is in the nascent state that drugs produce the greatest effect with the smallest dose.... Being thus eliminated by the entire respiratory tract: the lungs, bronchi and the throat, the sulphurretted hydrogen passes from the interior to the exterior, that is to say, goes right through these organs which are, as a consequence, thoroughly cleansed, antisepticized and freed of the pathogenic micro-organisms.... Then, again, part of the sulphuretted hydrogen, liberated in the stomach, is eliminated by the mouth and acts as an antiseptic and disinfectant of the mucous membranes of the throat and mouth. Hence Sulfuryl Monal is a perfect protective agent against contagious diseases.... Numerous clinical tests have demonstrated its real efficacy in diseases of the throat and of the respiratory tract: laryngitis, pharyngitis, hoarseness, granulations, tonsillitis, colds, bronchitis, pulmonary catarrh, asthma, emphysema, grippe, whooping cough, simple and infectious pneumonia, and in the first stage of pulmonary tuberculosis.”
The sulphids are practically ignored in modern textbooks. There is a rather extensive clinical literature on the subject, particularly in connection with sulphur waters; this, however, offers no good evidence for the therapeutic value of sulphids. Probably the tradition in their favor is largely due to the old popular idea that a disagreeable taste or odor is a mark of a good remedy.95
When hydrogen sulphid is introduced into the body, the small amounts that appear in the expired air are insufficient for quantitative demonstration and it is highly improbable that the amount thus excreted has any germicidal action, or that enough is excreted in the lungs to cause irritation and a reaction. The claim that Sulfuryl Monal is “a perfect protective agent against contagious diseases” is unwarranted; the recommendation for its use in “simple and infectious pneumonia, and in the first stage of pulmonary tuberculosis” is dangerous and vicious. The Council declared Sulfuryl Monal ineligible for New and Nonofficial Remedies and authorized publication of this report.
[Editorial Note..—With one exception, this product does not appear to be advertised in medical journals. We find, however, in the gallery of nostrums that grace the advertising pages of the International Journal of Surgery, that Sulfuryl Monal has its place. According to an advertisement that has been running some months in this publication, “affections of the throat and respiratory organs respond promptly” to Sulfuryl Monal whose “effects are rapid and certain” even in “incipient tuberculosis.” This preposterous pronouncement is no worse than many others appearing in the same journal, but it is bad enough to indicate how uncritical must be the physicians who support—by subscription or contribution—publications that are still debasing scientific medicine.]—(From The Journal A. M A., Sept. 16, 1916.)
MARK WHITE GOITER SERUM AND MARK WHITE IODINIZED OIL
Report of the Council on Pharmacy and Chemistry
The “Mark White Goiter Serum Laboratories” of Chicago asked the Council to consider its products “Mark White Goiter Serum” and “Mark White Iodinized Oil.” The “serum” was claimed to be an “antibody blood serum from a goat with thyroidosis” while the “Iodinized Oil” was said to contain “about 4 grains of iodin” to “each c.c.” The therapeutic indications for the treatment were given as:
“Simple or Exophthalmic Goiter, Hyperthyroidism-dosis, Thyrosis, Thyroidosis, Thyrotoxicosis, Dementia.”
An ampule (2 c.c.) of the “serum” is to be injected into the thyroid to be followed one week later by an ampule (2 c.c.) of the “Iodinized Oil.” Repetition of this “treatment” once or twice a month is advised.
The Council asked for more specific information as to the composition of the remedies, particularly as to the preparation and nature of the serum; it also asked for evidence of the therapeutic value of the preparations. In reply, Mark White wrote:
“All that I can say regarding the serum is that it is made from the blood of goats with thyroid affection, and it has been found that the serum from these goats has antibodies which control, or has curative effect upon thyroid affections when injected into thyroid glands of either humans or animals. As to the iodinised oil, it is only an adjunct or side treatment which is not always used or indicated, and will only be furnished to the physician for use in case in his judgment his patient needs it. We shall also advise the use of quinin ... when indicated....”
The Council was referred for further information to a paper by Rachel Watkins, M.D., published in the Illinois Medical Journal. It is to be noted, incidentally, that the letterheads used by White in his correspondence bore in one corner the notation “Rachel Watkins, M.D., Practice Limited to Goiter and Other Disorders of the Thyroid Glands,” and in the other, “Mark White, Goiter Research.”
The information regarding the composition of this goiter treatment, as furnished in Dr. Watkins’ paper, was to this effect:
“The medical treatment consists of the administration of a blood serum derived from a thyrodized goat. Formula: Iodine 0.16 grams [according to a correction by Mark White, this should read 0.26 gm.], oil 0.25 c.c., serum q. s. 1 c.c.”
This description of the treatment differs from that furnished to the Council by Mark White in that here the iodin and oil appear to be combined with the serum. Dr. Watkins’ “formula” implies that the iodin is a routine medication, thus contradicting White’s statement, which, in turn, is at variance with the statements made in submitting the treatment.

Photographic reproductions (greatly reduced) of some of the letterheads used by the Mark White concern during the past five years.
The Council does not accept any biologic product until its sale in interstate commerce has been authorized by the secretary of the treasury in accordance with the federal law regulating the sale of viruses, serums, toxins and analogous products. The sale of the Mark White Goiter Serum has not been so authorized; consequently even if the preparation complied with other rules of the Council it could not be accepted.
In addition, however, this treatment conflicts with other Council rules. The statements regarding its composition are indefinite and contradictory (Rule 1); the evidence presented to support the therapeutic claims is insufficient in itself and does not appear to have been checked by any disinterested authority (Rule 6). Moreover, the recognized variation in the morphology and pathology of the types of goiter render it impracticable to treat cases of goiter by any routine procedure.
The foregoing report was submitted to the Mark White Goiter Serum Laboratory. In reply, a letter signed “Mark White, V.M.D.,” was received, which read, in part:
“... we hope at some future time to be able to give you more detailed information, but as you possibly appreciate that we have experienced for some time a demand on the part of many physicians that we furnish to them our therapy, which necessitates us furnishing it before all the detailed work has yet been accomplished, and I trust that you will be so kind as to bear patiently with us until we are better in a position to make a complete scientific application and report to you.”
White wrote further:
“The serum and iodized oil may be mixed for immediate use, but could not be put up only separate for the use of the profession and the therapy furnished Dr. Watkins she mixed as used.”
This statement throws no light on the discrepancies in the statements with regard to the place of the iodinized oil in the treatment, namely: (a) the original statement that the oil was to be given a week after the serum; (b) White’s statement (quoted earlier in this report) that the oil “is only an adjunct or side treatment” and “is not always used or indicated”; (c) the statement in Dr. Watkins’ paper that the oil and the serum are given in combination.
The Council declared the Mark White Goiter Serum and Mark White Iodinized Oil ineligible for New and Nonofficial Remedies and authorized publication of this report.
Editorial Note on the Mark White “Serum”
As some of our readers will remember, on April 26, 1913, The Journal called attention to the Mark White preparation which at that time was being exploited from Denver. The Propaganda Department has in its files a number of letters sent out from the Mark White concern at various times. One mailed in May, 1911, on the embossed stationery of “The Mark White Goiter Institute,” Exchange Building, Denver, was evidently a general letter sent to physicians, calling their attention to “the most important medical discovery of the age.” “Dr. Mark White, a graduate of the University of Pennsylvania,” said the letter, had discovered “a simple and harmless remedy” that would cure goiter. “Because of the desire to preserve the secrecy of this remedy it is given only at the office here.” It was then suggested that the doctor might send those of his patients who were suffering from thyroidism to the “Mark White Goitre Institute.” If he would do so he would be “given a commission of $10, in cases of the $50 fee with the additional $5 for each $50 increase.” It closed with some casuistic arguments, presumably for the purpose of overcoming the physician’s scruples, summing up the matter with the statement:
“No right thinking man will allow a narrow and self-seeking system of ethics to stand between him and his duty to the sick and suffering.”
About 1912 the name of the concern seems to have been changed, for we have in our files a letter addressed to a layman on the stationery of the “Mark White Goitre Treatment Company.” According to this letterhead the product this concern had for sale was “Goitreine” discovered by Mark White, “President and General Manager.” Mr. White’s letter to the sufferer from goiter assured him that if he would take “Goitreine” he might “be practically sure of an immediate and permanent cure.” “Goitreine,” according to White, “has absolutely and permanently cured 90 per cent.” of all cases of goiter in which it has been used—“and the other ten showed remarkable improvement.” It was efficacious for all forms of goiter and “cannot possibly harm.”
The person who received this assurance might have had his confidence in it shaken had he seen a copy of the Denver News for May 23, 1911, in which was reported a case of collapse and death in a woman following an injection given in White’s office. The paper stated that the death certificate was signed by one W. A. Gray and gave “fatty degeneration of the heart and goiter” as the cause of death. Gray, it seems, was the licensed physician employed by Mark White to administer “Goitreine”—if that is what White happened to be calling his product at that time. For here it may be stated, parenthetically, that Mark White is not a physician; he is a veterinarian.
In February, 1913, Mark White sent a circular letter to a number of medical publications with the request that it be printed in full in the next issue, “to cover one full page of space.” The letter White wanted printed was addressed to doctors offering to “enter into a copartnership agreement” with such physicians who would be willing to treat “patients with goiter affections on a 50 per cent. commission basis.”
“You would be expected to make a cash charge to the patient for the treatment, remitting on the same day our 50 per cent. to us, when ordering the treatment, giving the treatment in no cases for less than $50.00.”
About the same time that Mark White made this “fifty-fifty” offer, he sent in an advertisement to be published in the classified column of The Journal. At that time he was told his advertisement was not acceptable; we now reprint it, however, free of charge. Here it is:
“WANTED—ONE OR MORE PHYSICIANS
in each vicinity to administer and represent our new medical treatment for GOITER. Good margin of profit. Write for copy of contract. The Mark White Goitre Treatment Co., Denver, Colo.”
In 1914, White moved to Chicago. At least the card which we reproduce so indicates. At that time, as will be seen, “Dr. Mark White” was “personally associated” with Peter S. Clark, M.D. According to the same card Dr. F. D. Paul of Rock Island, Ill., seems to have been his “associate” for that particular locality. In this connection, it is worth noting that a Rock Island paper, in one of its issues during July, 1913, devoted a good deal of space to “Dr. Mark White” who was at that time in Rock Island “directing Dr. Frank D. Paul in the administering of the treatment.” There was nothing to indicate that this notice was an advertisement or that the editorial appearing in the same issue puffing White’s “important cure,” was paid for.

When exploited from Denver the Mark White “goiter cure” was advertised in the daily papers. Here is a photographic reproduction (reduced) of an advertisement that appeared in the Denver Post, Sept. 1, 1912.
Dr. W. A. Gray, who has already been mentioned as White’s associate in Denver, seems to have been doing business in Illinois some time in 1913 and a Princeton (Ill.) paper had some uncomplimentary things to say about him. Finally in July, 1913, this item appeared in a Princeton paper.
“Dr. W. A. Gray, the goiter specialist who operated last winter at Princeton and Walnut until he became embroiled with Dr. Mark White, a Denver veterinary and originator of the cure, over a division of the spoils, has opened a goiter institute in Chicago under his own name. Advertisements of the Dr. Gray Goiter Institute appeared Sunday morning in the Chicago Examiner and other morning papers. Dr. Gray and Mark White broke off their relations after their disagreement at Walnut, and Dr. Gray slightly changed the ingredients of the goiter cure and started off on his own hook.”
One of Gray’s advertisements in Chicago newspapers made the claim that “Dr. Gray’s New Medical Treatment removes the cause of goiter in seven days.”

Photographic reproduction (reduced) of the “professional” card used by “Dr. Mark White” after he came to Chicago.
The Tulsa (Okla.) associate of “Dr.” White seems to have been Dr. J. H. Morgan and the Tulsa papers of June, 1914, tell of “Dr.” White’s visit to that city “for the purpose of instructing Dr. J. H. Morgan in the technique of his new medical treatment for nervous disorders and goiter.” Some months later—in December, 1915—the following little item appeared in a Tulsa paper:
“Dr. Mark White was found guilty in the county court yesterday of practicing medicine without a license and was fined $50. Doctor White is a goiter specialist.”
In September, 1915, Mr. Thomas S. Hogan, the efficient counsel for the Illinois State Board of Health, instituted action against Mark White for practicing medicine without a license. The case was tried Oct. 15, 1915, and the jury, after being out four hours, returned a verdict of “not guilty.” Attorney Hogan attributes the failure to obtain a conviction to the testimony of Dr. Rachel Watkins, who said she had a partnership arrangement with White in carrying on the medical business. It was about this time that Mark White seems to have issued some new letterheads. These bore in their upper left hand corner the device “Rachel Watkins, M. D., Practice Limited to Goiter and Other Disorders of the Thyroid Glands,” while the upper right hand corner read “Mark White, Goiter Research.”
On Dec. 9, 1915, Rachel Watkins, M. D., of Chicago, read a paper entitled “A Serum Treatment for Physiologically Defective Thyroids, With Clinical Reports” before the Stock Yards Branch of the Chicago Medical Society. The “serum treatment” discussed was Mark White’s “Goitreine” which, in the course of its checkered career, had lost its original name by the wayside. This paper appeared in the December, 1915, issue of the Illinois Medical Journal.
Probably emboldened by the ease with which a component part of the American Medical Association “fell for” a paper exploiting a “goiter cure,” Dr. Watkins requested that she be permitted to read a paper on the same subject before the Section on Pharmacology and Therapeutics at the Detroit meeting of the American Medical Association last June. The request was refused. Dr. Watkins is apparently no longer connected with White and in fact has protested against the use of her name by White in connection with his “goiter cure.”
[After the above was in type and ready for the pages of The Journal, attention was called to the Official Bulletin of the Chicago Medical Society of Sept. 16, 1916. This Bulletin contained a full page advertisement of the Mark White “goiter cure.” The advertiser referred to the preparation as having been “announced to the Chicago Medical Society” and declared it to be “an ethical therapeutic agent.” Mark White was described as “a medical research student” but no hint was given that he is a veterinarian. After again emphasizing that “this therapy is ethically proven” physicians were invited to “visit our goats when convenient” and the advertisement closed with the modest claim that “this thyroid therapy has equal curative therapeutic value in these cases as quinin in malaria.” And this sort of pseudo-scientific claptrap is presented to a presumably learned profession through its own official Bulletin—but what’s the use of commenting!]—(From The Journal A. M. A., Sept. 23, 1916.)
KORA-KONIA
Report of the Council on Pharmacy and Chemistry
Kora-Konia is a “dusting powder” which at present is advertised to the medical profession through medical journals, circulars, post cards and sample packages. It is put out by the “House of Mennen,” which sells various toilet preparations such as talcum powder, shaving soap, etc. On the trade package is the statement:
95 Liquid Sulphur—Sulphume, J. A. M. A., Dec. 2, 1911, p. 1853.
